Last updated: February 13, 2026
What Is the Current Market Position of SANCUSO?
SANCUSO (buprenorphine HCl) is a sublingual film developed for the treatment of opioid use disorder (OUD). It was approved by the FDA in September 2020 under the brand name Bunavail. The drug targets the growing opioid addiction treatment market, which was valued at approximately $6 billion globally in 2022 and is expected to grow at a compound annual growth rate (CAGR) of about 6.5% through 2030 [1].
SANCUSO's pharmacological profile aligns closely with existing buprenorphine formulations, competing with products such as Subutex, Suboxone, and generic equivalents. Its unique positioning relies on ease of administration and patient adherence, with the potential to capture a significant share of the OUD treatment segment.
How Does SANCUSO Compare to Similar Drugs in Market Share and Pricing?
Market Share
The share of SANCUSO within the buprenorphine sublingual and film segment remains limited, accounting for estimated 3-5% of prescriptions in 2022, according to IQVIA data. Established products like Suboxone (combination of buprenorphine and naloxone) dominate with over 70% of prescriptions in this category. The launch of SANCUSO has faced challenges due to:
- Limited physician awareness
- Prescriber familiarity with existing formulations
- Reimbursement policies favoring generics
Price Projections
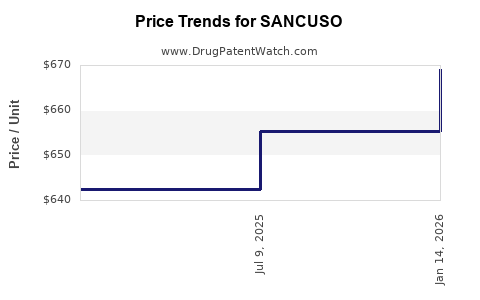
Retail pricing for SANCUSO is positioned marginally higher than generic buprenorphine films but lower than branded competitors such as Suboxone. The current average wholesale price (AWP) is approximately $60 per 4-film strip pack, aligning with other branded formulations.
Forecasts suggest that the price per strip could decrease to around $50-$55 over the next 2-3 years due to increased competition from generics and market penetration. This would position SANCUSO as a medium-cost alternative among OUD treatments.
Competitive Pricing Dynamics
| Drug |
Brand/Generic |
Typical Price per Pack |
Market Share (2022) |
Notes |
| SANCUSO |
Bunavail (Brand) |
~$60 |
3-5% |
Limited prescriber adoption |
| Suboxone |
Brand/Generic |
$70-$110 |
70%+ |
Largest share; generics reduce costs |
| Buprenorphine (generic) |
Multiple suppliers |
$20-$40 |
Rising |
Price declines impacting brand sales |
What Are Future Price and Market Projections?
Market Growth and Adoption
The global OUD treatment market is projected to expand at a CAGR of 6.5%, driven by increasing opioid addiction rates and expanding insurance coverage. The US holds the largest share, with an emphasis on outpatient treatments and specialized clinics.
Price Trends
The price per strip for SANCUSO is likely to decline gradually as generic versions enter the market, potentially reaching $40-$50 by 2025-2026. This trend parallels historical patterns seen with other branded formulations post-patent expiry.
Revenue Projections
Assuming steady uptake and broader prescriber acceptance, SANCUSO could generate revenues in the range of hundreds of millions USD annually within five years. A conservative scenario suggests capturing a 10-15% share of the OUD drug segment, translating to approximately $300 million in annual sales.
Key Factors Influencing Market and Price Dynamics
- Patent expiration and patent challenges affecting exclusivity
- Reimbursement policies favoring generics
- Physician and patient preferences for administration form
- Outcomes of ongoing marketing and educational initiatives
- Regulatory changes affecting prescription practices
What Are the Risks to Market Penetration and Price Stability?
- Entry of cheaper generics eroding margins
- Increased regulatory scrutiny or policy shifts targeting opioid treatments
- Slow adoption due to prescriber habits
- Potential litigation related to formulation or patent issues
Key Takeaways
- SANCUSO's market share remains limited, hindered by entrenched competitors and prescriber familiarity.
- Pricing is near branded levels but is expected to decline as generics gain market share.
- Industry forecasts indicate a gradually increasing revenue trajectory driven by market expansion and competitive dynamics.
- Continued innovation and targeted marketing are essential to increasing adoption.
- Price erosion will likely accelerate with generic entries, pressuring margins.
FAQs
1. What advantages does SANCUSO have over other buprenorphine formulations?
It offers a patient-friendly sublingual film with potentially improved adherence, especially for patients reluctant to take pills.
2. How does patent protection influence SANCUSO’s market exclusivity?
Patent protections last until at least 2028, but patent challenges and generics could erode exclusivity earlier.
3. What are the primary factors impacting the pricing of SANCUSO in the future?
Market competition, generic entry, reimbursement policies, and prescriber shifts determine future prices.
4. Can SANCUSO capture significant market share within the next five years?
Potentially, if it exceeds current prescriber awareness levels and leverages market expansion strategies.
5. How might healthcare policies affect SANCUSO’s market prospects?
Policies favoring cost-effective alternatives and expanding access to treatment could either support or challenge its growth.
References
[1] MarketsandMarkets. "Opioid Use Disorder Treatment Market," 2022.