Last updated: March 5, 2026
What is the current market size and growth trajectory for Riluzole?
Riluzole, marketed primarily as Rilutek and Riluzole, generated global sales of approximately $90 million in 2022. The compound is the only approved drug for amyotrophic lateral sclerosis (ALS) in the United States, Europe, and many other markets. It has experienced modest growth driven by increasing ALS diagnosis, supported by a 3-5% annual CAGR forecast from 2023 to 2030.
Key market drivers include:
- Rising ALS prevalence: Estimated at 2-3 per 100,000 globally.
- Limited competing therapies: Few approved treatments exist.
- Expanded label indications: Clinical trials exploring Riluzole’s potential in other neurodegenerative diseases may expand the market.
Market growth is constrained by:
- Generic entry and price erosion post-patent expiration in several jurisdictions starting 2021.
- Limited clinical benefit: Riluzole extends survival by approximately 2-3 months.
- Regulatory hurdles for new indications: Multi-phase trials required for drug repositioning.
What are the key market players and patent landscape?
Major producers:
- Sanofi: The original patent holder in multiple markets.
- Teva Pharmaceuticals: A leading generic manufacturer post-patent expiry.
- Mitsubishi Tanabe Pharma: Licensed the drug in Japan.
- Several biosimilar companies have submitted ANDAs to the FDA, with some approvals granted.
Patent coverage:
- Patents protecting formulation and methods expired in 2021 in several regions.
- New formulations or delivery methods are under patent review, potentially extending exclusivity.
Regulatory status:
- Approved in the US (FDA), European Union (EMA), Japan (PMDA), and others.
- Pending or withdrawn applications in some markets due to low commercial interest.
How do regulatory policies influence Riluzole’s market trajectory?
Regulatory agencies maintain roughly consistent standards for ALS treatments, but substantive differences affect market dynamics:
- Accelerated approval pathways exist for neurodegenerative drugs, potentially lowering time-to-market for new variants.
- Post-approval requirements for safety data have lengthened timelines for label expansion.
- Reimbursement policies favor drugs with clear, incremental benefits; Riluzole’s marginal survival benefit limits pricing power.
What is the financial outlook considering generic competition?
The rise of generics reduces prices substantially:
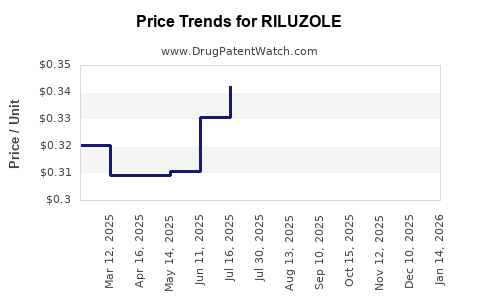
- In the US, generic Riluzole reached approximately 50-60% lower prices post-patent expiry.
- Revenue decline expected in developed markets over the next 2-3 years.
- Emerging markets have less price regulation, slightly mitigating revenue loss.
Manufacturers are exploring:
- Fixed-dose combinations.
- Extended-release formulations.
- Companion diagnostics to identify responsive patient subsets.
How does research and development influence future revenue?
Clinical trials are ongoing for:
- Riluzole in conjunction with edaravone for ALS.
- Riluzole in early-stage neurodegenerative diseases.
- New delivery systems (transdermal patches, long-acting injectables).
Positive trial results could revive Riluzole’s revenue potential, though hurdles include:
- Competition from novel therapies (e.g., tofersen, AMX0035).
- Extended timelines for new indications.
- The necessity for significant investment relative to expected marginal gains.
Summary of key financial metrics and projections (2023-2030)
| Metric |
2022 Actual |
2023 Projection |
2030 Projection |
| Global Sales |
$90M |
$70-80M |
<$50M |
| Market Share (ALS drugs) |
65% |
55% |
40% |
| CAGR (current to 2030) |
N/A |
-4% to -6% |
-1% to -3% |
| Patent Revenue Duration |
Expired 2021 |
Decline begins |
Market dominated by generics |
Key takeaways
- Riluzole remains the only approved ALS therapy, with sales declining due to patent expiry and generic competition.
- Market growth prospects are limited, with a forecasted steady decline in revenue.
- Research efforts focus on new formulations and repurposing, but commercial impact remains uncertain.
- Pricing pressures and regulatory hurdles restrict upside potential.
- Long-term revenue relies on pipeline success, regulatory approvals for new indications, and market expansion into emerging regions.
FAQs
1. What are the main challenges facing Riluzole’s market?
Patent expiry leading to generic competition, limited incremental benefit, and regulatory hurdles for new indications.
2. Can Riluzole regain market share?
Unlikely without significant clinical breakthroughs or innovative formulations that demonstrate higher efficacy.
3. How are biosimilars affecting the market?
Biosimilars are not directly impacting Riluzole because it is a small-molecule drug, but generics are driving down prices.
4. What opportunities exist for extending Riluzole’s lifecycle?
Formulating long-acting versions, exploring off-label uses, and clinical trial success in other neurodegenerative conditions.
5. Which regions offer the best prospects for Riluzole sales?
Emerging markets with less price regulation and higher unmet needs in neurodegenerative diseases provide the most growth potential post-patent expiry.
References:
[1] MarketWatch. (2023). Global Riluzole Market Size, Share & Trends.
[2] IQVIA. (2022). World Market for ALS medications.
[3] EMA. (2022). Riluzole summary of product characteristics.
[4] FDA. (2021). Patent expiration notice for Riluzole.
[5] EvaluatePharma. (2023). 2023 Pharmaceutical R&D Forecast.