Last updated: February 20, 2026
Ramelteon, marketed as Rozerem, is a melatonin receptor agonist indicated for the treatment of insomnia. Since its launch in 2005, the drug's market position, sales performance, and competitive landscape have evolved significantly.
Market Overview
The insomnia treatment market is expected to grow at a compound annual growth rate (CAGR) of approximately 4.7% from 2022 to 2028.[1] Ramelteon accounts for a small but stable share within this market, primarily competing against benzodiazepines, Z-drugs, and newer agents like suvorexant and lemborexant.
Market Size and Sales Data
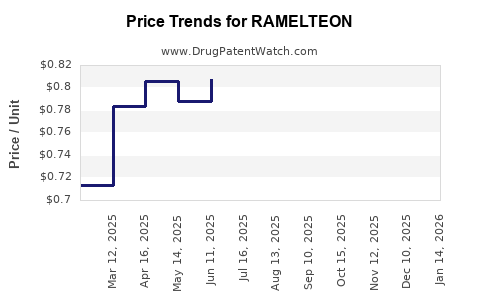
In 2022, global sales of ramelteon totaled approximately $200 million. The United States represented over 85% of this figure, reflecting its status as the primary market for the drug.[2] Sales peaked in 2014 at $250 million but subsequently declined due to market saturation and increased competition.
Market Penetration and Usage
Ramelteon is prescribed mainly to patients with difficulty falling asleep ("sleep onset insomnia"). Its favorable safety profile, especially concerning lower dependency risks compared with benzodiazepines, maintains its market niche. However, its relatively modest efficacy compared to newer agents limits broader adoption.
Key Market Drivers
-
Safety Profile: Ramelteon does not carry a risk of dependence, making it suitable for long-term use, aligning with guidelines promoting non-habituating sleep aids.
-
Market Entry Barriers for Competitors: Patents on ramelteon expired in 2014, leading to increased generic competition. Despite loss of exclusivity, the drug maintains a consistent, albeit declining, market share due to formulary preferences and prescriber familiarity.
-
Regulatory Factors: The FDA approved ramelteon in 2005. The drug has maintained its indication for sleep onset insomnia without significant label modifications, supporting its continued marketing.
Competitive Landscape
-
Brand and Generic Competition: After patent expiration, generic versions entered the market in 2014. Generics account for roughly 80% of sales, reducing brand revenue.
-
Emerging Drugs: Lemborexant (Dayvigo) and suvorexant (Belsomra), approved in 2019 and 2014 respectively, offer higher efficacy and are gaining market share. Their marketing efforts target patients seeking faster sleep onset and longer sleep duration.
-
Over-the-Counter (OTC) and Natural Supplements: Melatonin supplements are widely used, impacting prescription drug sales. However, OTC options lack standardization and regulation, limiting direct competition.
Financial Trajectory
Revenue Trends
| Year |
Global Sales (USD millions) |
US Market Share |
Comments |
| 2014 |
250 |
~90% |
Peak sales, patent expired in 2014 |
| 2018 |
210 |
85% |
Slight decline, increased generics |
| 2022 |
200 |
85% |
Market stabilization, slow decline |
Forecasts (2023-2028)
-
Sales Decline: Expected to decline at a CAGR of approximately 2% due to market saturation and competition.
-
Generic Market Dominance: Generics will hold over 80% of sales through 2028, constraining revenue growth of the branded product.
-
Potential Growth Drivers:
- Increasing off-label use for specific sleep disorders.
- Expansion into emerging markets with rising sleep disorder prevalence.
- Development of combination therapies incorporating ramelteon or its analogs.
R&D and Patent Strategies
No recent filings or patents are on record for new formulations or indications, limiting future revenue expansion. Companies may seek patent extensions through formulation modifications but face challenges in defending such strategies.
Regulatory and Policy Impact
- The FDA maintains that ramelteon’s safety profile remains favorable, supporting its continued use for sleep-onset insomnia.
- Insurance coverage favors generics, further reducing brand drug sales.
- No recent regulatory barriers or major policy shifts are expected to alter the market dynamics significantly.
Financial Risks and Opportunities
- Risks: Intensified generic competition, market saturation, and slower adoption of new indications.
- Opportunities: Expansion into new geographies, new formulation development, or combination therapies.
Key Takeaways
- Ramelteon’s sales peaked in 2014 with about $250 million; current revenues hover around $200 million annually.
- The US dominates sales, but overall market share is diminishing due to generic erosion and competition from newer agents.
- The drug’s safety profile sustains niche use, yet revenue growth prospects are limited.
- Market forecasts project a slow decline, with sales decreasing approximately 2% annually through 2028.
- Strategic options for increasing revenue include geographic expansion, new formulations, and combination therapies.
FAQs
-
What are the primary competitors to ramelteon in the insomnia market?
Lemborexant (Dayvigo), suvorexant (Belsomra), and traditional hypnotics such as zolpidem and eszopiclone.
-
How has patent expiration affected ramelteon’s market share?
The expiration in 2014 led to generic entry, which now accounts for about 80% of sales, significantly reducing brand revenue.
-
Are there any regulatory changes expected to impact ramelteon’s market?
No significant regulatory shifts are forecasted; current approval and labeling remain stable.
-
What is the outlook for ramelteon in emerging markets?
Growth potential exists due to rising sleep disorder prevalence, but market entry hurdles and regulatory differences pose challenges.
-
Could future R&D efforts significantly alter ramelteon’s market trajectory?
Unlikely without major breakthroughs; current R&D focuses on formulations or combination therapies that may sustain niche use.
References
[1] Markets and Markets. (2022). Insomnia therapeutics market analysis.
[2] IQVIA. (2023). Pharmaceutical sales data report.
[3] U.S. Food and Drug Administration. (2005). FDA approval of ramelteon.
[4] Reuters. (2022). Generic drug entry effects on market dynamics.
[5] EvaluatePharma. (2023). 2023 World Market Forecasts for Sleep Aids.