QTERN Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Qtern, and when can generic versions of Qtern launch?
Qtern is a drug marketed by Astrazeneca Ab and is included in two NDAs. There are eight patents protecting this drug and two Paragraph IV challenges.
This drug has three hundred and ten patent family members in forty-eight countries.
The generic ingredient in QTERN is dapagliflozin; metformin hydrochloride; saxagliptin hydrochloride. There are twenty-six drug master file entries for this compound. Additional details are available on the dapagliflozin; metformin hydrochloride; saxagliptin hydrochloride profile page.
DrugPatentWatch® Generic Entry Outlook for Qtern
Qtern was eligible for patent challenges on January 8, 2018.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be December 16, 2029. This may change due to patent challenges or generic licensing.
There have been sixteen patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
Summary for QTERN
| International Patents: | 310 |
| US Patents: | 7 |
| Applicants: | 1 |
| NDAs: | 2 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 1 |
| Formulation / Manufacturing: | see details |
| Drug Prices: | Drug price information for QTERN |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for QTERN |
| What excipients (inactive ingredients) are in QTERN? | QTERN excipients list |
| DailyMed Link: | QTERN at DailyMed |
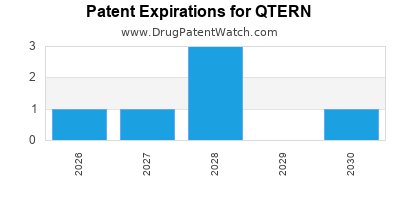

DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for QTERN
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Pharmacology for QTERN
Anatomical Therapeutic Chemical (ATC) Classes for QTERN
Paragraph IV (Patent) Challenges for QTERN
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| QTERN | Tablets | dapagliflozin; saxagliptin hydrochloride | 5 mg/5 mg | 209091 | 1 | 2020-07-29 |
| QTERN | Tablets | dapagliflozin; saxagliptin hydrochloride | 10 mg/5 mg | 209091 | 5 | 2018-01-08 |
US Patents and Regulatory Information for QTERN
QTERN is protected by eleven US patents.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of QTERN is ⤷ Sign Up.
This potential generic entry date is based on patent ⤷ Sign Up.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
Patents protecting QTERN
C-aryl glucoside SGLT2 inhibitors and method
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF TYPE 2 DIABETES MELLITUS
Crystal structures of SGLT2 inhibitors and processes for preparing same
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Pharmaceutical formulations containing an SGLT2 inhibitor
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Pharmaceutical formulations containing an SGLT2 inhibitor
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF TYPE 2 DIABETES MELLITUS
Pharmaceutical formulations containing an SGLT2 inhibitor
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: METHOD FOR TREATING TYPE 2 DIABETES MELLITUS (T2DM) IN PATIENTS WHO ARE ALREADY TREATED WITH DAPAGLIFLOZIN AND SAXAGLIPTIN
Pharmaceutical formulations containing an SGLT2 inhibitor
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: METHOD FOR TREATING TYPE 2 DIABETES MELLITUS (T2DM) IN PATIENTS WHO HAVE INADEQUATE CONTROL WITH DAPAGLIFLOZIN
Crystal structures of SGLT2 inhibitors and processes for preparing same
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF TYPE 2 DIABETES MELLITUS
Crystal structures of SGLT2 inhibitors and processes for preparing same
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: METHOD FOR TREATING TYPE 2 DIABETES MELLITUS (T2DM) IN PATIENTS WHO HAVE INADEQUATE CONTROL WITH DAPAGLIFLOZIN
Crystal structures of SGLT2 inhibitors and processes for preparing same
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: METHOD FOR TREATING TYPE 2 DIABETES MELLITUS (T2DM) IN PATIENTS WHO ARE ALREADY TREATED WITH DAPAGLIFLOZIN AND SAXAGLIPTIN
Coated tablet formulation and method
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Pharmaceutical formulations containing an SGLT2 inhibitor
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Astrazeneca Ab | QTERNMET XR | dapagliflozin; metformin hydrochloride; saxagliptin hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 210874-004 | May 2, 2019 | DISCN | Yes | No | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Astrazeneca Ab | QTERNMET XR | dapagliflozin; metformin hydrochloride; saxagliptin hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 210874-003 | May 2, 2019 | DISCN | Yes | No | ⤷ Sign Up | ⤷ Sign Up | Y | Y | ⤷ Sign Up | ||
| Astrazeneca Ab | QTERNMET XR | dapagliflozin; metformin hydrochloride; saxagliptin hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 210874-002 | May 2, 2019 | DISCN | Yes | No | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Astrazeneca Ab | QTERNMET XR | dapagliflozin; metformin hydrochloride; saxagliptin hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 210874-003 | May 2, 2019 | DISCN | Yes | No | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Astrazeneca Ab | QTERNMET XR | dapagliflozin; metformin hydrochloride; saxagliptin hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 210874-002 | May 2, 2019 | DISCN | Yes | No | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Astrazeneca Ab | QTERN | dapagliflozin; saxagliptin hydrochloride | TABLET;ORAL | 209091-002 | May 2, 2019 | RX | Yes | No | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for QTERN
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Astrazeneca Ab | QTERN | dapagliflozin; saxagliptin hydrochloride | TABLET;ORAL | 209091-001 | Feb 27, 2017 | ⤷ Sign Up | ⤷ Sign Up |
| Astrazeneca Ab | QTERN | dapagliflozin; saxagliptin hydrochloride | TABLET;ORAL | 209091-002 | May 2, 2019 | ⤷ Sign Up | ⤷ Sign Up |
| Astrazeneca Ab | QTERN | dapagliflozin; saxagliptin hydrochloride | TABLET;ORAL | 209091-002 | May 2, 2019 | ⤷ Sign Up | ⤷ Sign Up |
| Astrazeneca Ab | QTERN | dapagliflozin; saxagliptin hydrochloride | TABLET;ORAL | 209091-001 | Feb 27, 2017 | ⤷ Sign Up | ⤷ Sign Up |
| Astrazeneca Ab | QTERN | dapagliflozin; saxagliptin hydrochloride | TABLET;ORAL | 209091-001 | Feb 27, 2017 | ⤷ Sign Up | ⤷ Sign Up |
| Astrazeneca Ab | QTERN | dapagliflozin; saxagliptin hydrochloride | TABLET;ORAL | 209091-001 | Feb 27, 2017 | ⤷ Sign Up | ⤷ Sign Up |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for QTERN
When does loss-of-exclusivity occur for QTERN?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 1730
Estimated Expiration: ⤷ Sign Up
Australia
Patent: 07265246
Estimated Expiration: ⤷ Sign Up
Brazil
Patent: 0713544
Estimated Expiration: ⤷ Sign Up
Patent: 2017015106
Estimated Expiration: ⤷ Sign Up
Patent: 2017021516
Estimated Expiration: ⤷ Sign Up
Canada
Patent: 53344
Estimated Expiration: ⤷ Sign Up
Patent: 24318
Estimated Expiration: ⤷ Sign Up
Patent: 85797
Estimated Expiration: ⤷ Sign Up
Chile
Patent: 07001915
Estimated Expiration: ⤷ Sign Up
China
Patent: 1479287
Estimated Expiration: ⤷ Sign Up
Patent: 3145773
Estimated Expiration: ⤷ Sign Up
Colombia
Patent: 60299
Estimated Expiration: ⤷ Sign Up
Croatia
Patent: 0141007
Estimated Expiration: ⤷ Sign Up
Cyprus
Patent: 15738
Estimated Expiration: ⤷ Sign Up
Denmark
Patent: 69374
Estimated Expiration: ⤷ Sign Up
Eurasian Patent Organization
Patent: 8229
Estimated Expiration: ⤷ Sign Up
Patent: 0428
Estimated Expiration: ⤷ Sign Up
Patent: 8259
Estimated Expiration: ⤷ Sign Up
Patent: 5999
Estimated Expiration: ⤷ Sign Up
Patent: 0900066
Estimated Expiration: ⤷ Sign Up
Patent: 1171333
Estimated Expiration: ⤷ Sign Up
Patent: 1490902
Estimated Expiration: ⤷ Sign Up
Patent: 1791254
Estimated Expiration: ⤷ Sign Up
Patent: 2091391
Estimated Expiration: ⤷ Sign Up
European Patent Office
Patent: 69374
Estimated Expiration: ⤷ Sign Up
Patent: 57918
Estimated Expiration: ⤷ Sign Up
Patent: 45466
Estimated Expiration: ⤷ Sign Up
Patent: 63807
Estimated Expiration: ⤷ Sign Up
Hong Kong
Patent: 27359
Estimated Expiration: ⤷ Sign Up
Israel
Patent: 5882
Estimated Expiration: ⤷ Sign Up
Patent: 4180
Estimated Expiration: ⤷ Sign Up
Patent: 4181
Estimated Expiration: ⤷ Sign Up
Patent: 4182
Estimated Expiration: ⤷ Sign Up
Japan
Patent: 13889
Estimated Expiration: ⤷ Sign Up
Patent: 66651
Estimated Expiration: ⤷ Sign Up
Patent: 37187
Estimated Expiration: ⤷ Sign Up
Patent: 09545525
Estimated Expiration: ⤷ Sign Up
Patent: 13209394
Estimated Expiration: ⤷ Sign Up
Patent: 15071636
Estimated Expiration: ⤷ Sign Up
Patent: 16172758
Estimated Expiration: ⤷ Sign Up
Patent: 17222681
Estimated Expiration: ⤷ Sign Up
Patent: 19059779
Estimated Expiration: ⤷ Sign Up
Malaysia
Patent: 8566
Estimated Expiration: ⤷ Sign Up
Patent: 3930
Estimated Expiration: ⤷ Sign Up
Mexico
Patent: 9143
Estimated Expiration: ⤷ Sign Up
Patent: 7155
Estimated Expiration: ⤷ Sign Up
Patent: 08015377
Estimated Expiration: ⤷ Sign Up
New Zealand
Patent: 4346
Estimated Expiration: ⤷ Sign Up
Patent: 9190
Estimated Expiration: ⤷ Sign Up
Patent: 9195
Estimated Expiration: ⤷ Sign Up
Patent: 9202
Estimated Expiration: ⤷ Sign Up
Norway
Patent: 6828
Estimated Expiration: ⤷ Sign Up
Patent: 085169
Estimated Expiration: ⤷ Sign Up
Patent: 221233
Estimated Expiration: ⤷ Sign Up
Peru
Patent: 080349
Estimated Expiration: ⤷ Sign Up
Patent: 120776
Estimated Expiration: ⤷ Sign Up
Philippines
Patent: 012500168
Estimated Expiration: ⤷ Sign Up
Poland
Patent: 69374
Estimated Expiration: ⤷ Sign Up
Portugal
Patent: 69374
Estimated Expiration: ⤷ Sign Up
Serbia
Patent: 638
Estimated Expiration: ⤷ Sign Up
Singapore
Patent: 2741
Estimated Expiration: ⤷ Sign Up
Patent: 201402181S
Estimated Expiration: ⤷ Sign Up
Slovenia
Patent: 69374
Estimated Expiration: ⤷ Sign Up
South Africa
Patent: 0810475
Estimated Expiration: ⤷ Sign Up
South Korea
Patent: 1493102
Estimated Expiration: ⤷ Sign Up
Patent: 090023643
Estimated Expiration: ⤷ Sign Up
Spain
Patent: 21665
Estimated Expiration: ⤷ Sign Up
Patent: 59862
Estimated Expiration: ⤷ Sign Up
Patent: 69130
Estimated Expiration: ⤷ Sign Up
Taiwan
Patent: 21245
Estimated Expiration: ⤷ Sign Up
Patent: 66876
Estimated Expiration: ⤷ Sign Up
Patent: 19528
Estimated Expiration: ⤷ Sign Up
Patent: 0811127
Estimated Expiration: ⤷ Sign Up
Patent: 1406743
Estimated Expiration: ⤷ Sign Up
Patent: 1509927
Estimated Expiration: ⤷ Sign Up
Patent: 1546054
Estimated Expiration: ⤷ Sign Up
Ukraine
Patent: 765
Estimated Expiration: ⤷ Sign Up
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering QTERN around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Hong Kong | 1205120 | -芳基葡糖苷 抑制劑和方法 (C-ARYL GLUCOSIDE SGLT2 INHIBITORS AND METHOD C- SGLT2) | ⤷ Sign Up |
| South Korea | 20140124874 | 다파글리플로진 프로필렌 글리콜 수화물을 함유하는 제약 제형 (PHARMACEUTICAL FORMULATIONS CONTAINING DAPAGLIFLOZIN PROPYLENE GLYCOL HYDRATE) | ⤷ Sign Up |
| Argentina | 065809 | FORMULACIONES FARMACEUTICAS QUE CONTIENEN UN INHIBIDOR SGLT2 | ⤷ Sign Up |
| Czech Republic | 307784 | Inhibitory dipeptidylpeptidázy IV na bázi cyklopropylu kondenzovaného s pyrrolidinem, způsoby jeho přípravy a jejich použití (Cyclopropyl-coupled cyclopropyl-dipeptidyl peptidase IV inhibitors, how to prepare and use them.) | ⤷ Sign Up |
| Denmark | 1261586 | ⤷ Sign Up | |
| Turkey | 200200986 | ⤷ Sign Up | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for QTERN
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1506211 | SPC/GB13/021 | United Kingdom | ⤷ Sign Up | PRODUCT NAME: DAPAGLIFLOZIN AND PHARMACEUTICALLY ACCEPTABLE SALTS THEREOF; REGISTERED: UK EU/1/12/795/001 20121114; UK EU/1/12/795/002 20121114; UK EU/1/12/795/003 20121114; UK EU/1/12/795/004 20121114; UK EU/1/12/795/005 20121114; UK EU/1/12/795/006 20121114; UK EU/1/12/795/007 20121114; UK EU/1/12/795/008 20121114; UK EU/1/12/795/009 20121114; UK EU/1/12/795/010 20121114 |
| 1506211 | C01506211/01 | Switzerland | ⤷ Sign Up | PRODUCT NAME: DAPAGLIFLOZIN; REGISTRATION NO/DATE: SWISSMEDIC 65176 19.08.2014 |
| 1506211 | 179 5017-2014 | Slovakia | ⤷ Sign Up | PRODUCT NAME: KOMBINACIA DAPAGLIFLOZINU ALEBO JEHO FARMACEUTICKY PRIJATELNYCH SOLI A METFORMINU ALEBO JEHO FARMACEUTICKY PRIJATELNYCH SOLI; REGISTRATION NO/DATE: EU/1/13/900/001 - EU/1/13/900/012 20140116 |
| 1261586 | 91650 | Luxembourg | ⤷ Sign Up | 91650, EXPIRES: 20241001 |
| 1506211 | C01506211/02 | Switzerland | ⤷ Sign Up | PRODUCT NAME: DAPAGLIFLOZIN + METFORMIN; REGISTRATION NO/DATE: SWISSMEDIC 65377 16.07.2015 |
| 1261586 | C300524 | Netherlands | ⤷ Sign Up | PRODUCT NAME: COMBINATIE VAN SAXAGLIPTINE EN METFORMINE, DESGEWENST IN DE VORM VAN FARMACEUTISCH AANVAARDBARE AFGELEIDEN DAARVAN; NAT. REGISTRATION NO/DATE: EU/1/11/731/001-012 20111124; FIRST REGISTRATION: |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


