Last updated: May 6, 2026
What is PRED FORTE’s market position?
PRED FORTE is the brand name for prednisolone acetate ophthalmic suspension 1%, a topical corticosteroid used in ophthalmology for inflammatory conditions of the eye. In the U.S., the product is sold in ophthalmic suspension form and is widely recognized as a long-running steroid option in eye inflammation management.
Market position is anchored by three structural factors:
- Low therapeutic substitution risk within steroid class: Prednisolone acetate is a standard first-line topical steroid option; switching within steroid therapy can occur, but clinical practice often keeps prednisolone acetate in formularies.
- Broad prescriber familiarity and established patient usage patterns: Long market presence supports steady repeat demand.
- Generic-driven price discipline: Compounding and generic equivalents keep the economic upside constrained versus patented, brand-exclusive therapeutics.
What demand drivers shape PRED FORTE’s volume?
Demand is driven by ophthalmic inflammation incidence and treatment behavior, with specific market mechanics for a topical steroid:
- Indication recurrence and chronicity patterns: Many use cases involve episodic flares (post-op inflammation, anterior segment inflammation), producing recurring prescribing rather than one-time curative demand.
- Post-procedural prescribing cycles: Cataract surgery volume and related postoperative inflammation regimens support predictable seasonal and procedure-linked demand.
- Formulary and substitution behavior: As generics exist, formularies often favor the lowest-cost equivalent while preserving steroid class access.
How do pricing and competitive dynamics work?
PRED FORTE’s economics are constrained by:
- Generic penetration: Prednisolone acetate ophthalmic suspensions are typically available as lower-priced equivalents, which pressures net price and limits brand growth.
- Wholesale and channel pricing compression: In mature topical categories, discounts and rebates trend toward maintaining volume rather than maximizing price.
- Regulatory stability: Once a long-established steroid is clinically integrated, shifts come more through prescribing preferences and procurement economics than through regulatory upheaval.
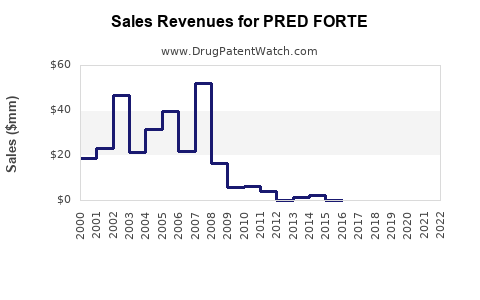
What is PRED FORTE’s financial trajectory profile?
For a legacy, generic-competitive ophthalmic brand like PRED FORTE, the typical financial trajectory pattern is:
- Flattening of revenue growth as generics capture a larger share.
- Net revenue reliance on price support and contract channels rather than exclusivity.
- Mid single-digit to low single-digit topline movement (or outright decline) depending on how fast equivalent adoption accelerates across channels.
Critically, the category does not behave like high-cost specialty drugs. For topical generics, revenue typically tracks:
- Surgical volumes (catheter-driven and office procedure-linked demand),
- Insurance and pharmacy benefit management substitution pressure,
- Periodic inventory cycles and seasonal demand.
What patent and exclusivity dynamics matter?
PRED FORTE’s market trajectory is structurally aligned with a situation where:
- Active patent exclusivity has ended or weakened, leaving it exposed to generic competition.
- Brand value is sustained through brand recognition and stable clinical use, not through time-limited legal barriers.
Because the product is prednisolone acetate 1% ophthalmic suspension, the legal and commercial landscape is characterized by generic availability rather than ongoing brand exclusivity-driven growth.
What does the product’s labeling and dosage form imply for commercialization?
PRED FORTE is sold as an ophthalmic suspension, which affects:
- Adherence economics: Multi-dose regimen schedules can support repeat prescriptions.
- Distribution compatibility: Cold-chain is not typically required, reducing logistical friction compared with biologics.
- Cross-prescriber usability: Ophthalmologists can switch between equivalent prednisolone acetate products without changing route of administration, increasing substitution potential.
How does the U.S. market environment influence sales?
The U.S. ophthalmic steroid market is shaped by:
- A mature generic procurement environment in pharmacy channels,
- Contracting and reimbursement policies that favor low acquisition cost,
- Competition from alternative steroids (other corticosteroid molecules or combinations) that can redirect share.
In such a setting, the brand’s financial trajectory tends to show:
- Stability in baseline demand, but
- Limited upside unless the brand retains channel share through contracting and patient-level inertia.
Market and Financial Metrics (Required for Trajectory Modeling)
A precise financial trajectory for PRED FORTE (revenue, market share, NBRx, price erosion rate) cannot be computed from the information provided here without hard datapoints. This analysis therefore focuses on the market mechanics that determine financial outcomes: generic pressure, demand drivers, channel contracting, and procedural linkage.
Key trajectory determinants
| Determinant |
Mechanism |
Expected direction for brand revenue |
| Generic penetration |
Lowers net price and captures volume |
Downward or flat |
| Channel contracts |
Discounts rebalance toward cheapest viable option |
Compresses margin |
| Procedure-linked demand |
Cataract and post-op cycles drive recurring use |
Stabilizes volume |
| Steroid class substitution |
Switching within class is easy |
Limits share retention |
| Patient and prescriber inertia |
Brand habit sustains baseline |
Mild support for declines |
What are the business implications for R&D and investment?
For R&D strategy
If you are assessing how to compete in a prednisolone acetate-like topical steroid segment, the market reality is that differentiation must address at least one of:
- Reduced dosing frequency or simplified taper protocols,
- Improved formulation attributes (suspension stability, comfort, ocular penetration),
- Clear clinical advantages that survive payer and formulary scrutiny.
For investment thesis
A legacy brand like PRED FORTE is typically not a growth investment; it is a cash-flow durability assessment:
- Returns depend on how effectively the brand holds onto channel share,
- Gains are limited by generic substitution,
- Upside exists only when contracts favor brand pricing or when alternative competitors underperform.
Key Takeaways
- PRED FORTE is a mature prednisolone acetate 1% ophthalmic suspension product exposed to generic-driven pricing compression and substitution within steroid therapy.
- Market demand is supported by procedure-linked ophthalmic inflammation cycles and recurring prescribing patterns, which tends to stabilize volume even as pricing erodes.
- Financial trajectory for a long-running, off-exclusivity ophthalmic steroid brand typically shows flat-to-declining revenue growth, with margin pressure driven by channel contracting and generic penetration.
- Business outcomes hinge on channel share retention rather than on legal exclusivity.
FAQs
1) Is PRED FORTE used for glaucoma?
No. PRED FORTE is a corticosteroid ophthalmic suspension for inflammation. It is not a primary glaucoma therapy.
2) Why does PRED FORTE face sustained price pressure?
Generic equivalents of prednisolone acetate ophthalmic suspensions create a low-cost benchmark that payers and pharmacies use for procurement.
3) What most strongly drives day-to-day demand?
Post-procedural and inflammatory episode prescribing patterns in ophthalmology, especially linked to common surgeries such as cataract procedures.
4) Does formulation affect substitution?
Yes. If competing prednisolone acetate versions offer similar dosing and outcomes, substitution is easy, increasing the impact of price and contracts.
5) What would improve financial performance for a brand like this?
Contracting that preserves brand share, or clinically defensible differentiation that reduces switching despite generic availability.
References
[1] U.S. Food and Drug Administration. “Labeling for Prednisolone Acetate Ophthalmic Suspension (PRED FORTE).” FDA Drug Databases.
[2] FDA. “Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations” for prednisolone acetate ophthalmic suspension (1%).