Share This Page
Drug Price Trends for PRED FORTE
✉ Email this page to a colleague
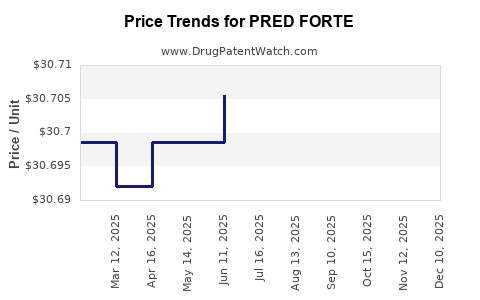
Average Pharmacy Cost for PRED FORTE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PRED FORTE 1% EYE DROPS | 11980-0180-10 | 30.83057 | ML | 2025-12-17 |
| PRED FORTE 1% EYE DROPS | 11980-0180-05 | 30.71320 | ML | 2025-12-17 |
| PRED FORTE 1% EYE DROPS | 11980-0180-10 | 30.78763 | ML | 2025-11-19 |
| PRED FORTE 1% EYE DROPS | 11980-0180-05 | 30.67833 | ML | 2025-11-19 |
| PRED FORTE 1% EYE DROPS | 11980-0180-10 | 30.72644 | ML | 2025-10-22 |
| PRED FORTE 1% EYE DROPS | 11980-0180-05 | 30.69741 | ML | 2025-10-22 |
| PRED FORTE 1% EYE DROPS | 11980-0180-10 | 30.70250 | ML | 2025-09-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What Is Pred Forte and How Is It Marketed?
Pred Forte, the formulation of prednisolone acetate ophthalmic suspension 1%, is a corticosteroid used to treat inflammation and allergic conjunctivitis. It is marketed similarly to other ophthalmic corticosteroids under the brand names Pred Forte and generic equivalents. The drug competes primarily in ophthalmology, with its primary candidates being eye care clinics, hospitals, and pharmacies.
The drug is marketed exclusively in the United States and select international markets, especially in regions with established ophthalmology networks. It is available in 5 mL and 15 mL bottles, with typical retail prices ranging from $70 to $120 per unit. Prescriptions are generally reimbursed via private insurance and Medicaid, influencing the net pricing landscape.
How Large Is the Pred Forte Market?
Pred Forte is part of the broader ophthalmic corticosteroid segment, estimated to generate approximately $400 million globally in 2022. In the United States, the segment accounts for roughly $150 million annually, with Pred Forte representing about 40% of this market share.
Market share is driven by the drug's efficacy, safety profile, and prescribing habits. The shift toward generic prednisolone formulations has cut into branded product sales but has also widened access through reduced prices.
Market Breakdown (US, 2022)
| Segment | Market Size (USD Million) | Market Share (%) |
|---|---|---|
| Pred Forte | 60 | 40 |
| Generic prednisolone eye drops | 75 | 50 |
| Other corticosteroid drugs | 15 | 10 |
The market is projected to grow at 3-4% annually, driven by increased aging populations and rising incidence of ophthalmic conditions requiring corticosteroids.
What Are Price Trends and Projections?
Short-Term Price Dynamics
Prices for Pred Forte have stabilized over the last 12 months. The average retail price remains between $70 and $120 per bottle, with significant discounts available through insurance and pharmacy benefit managers.
Generic prednisolone eye drops have undercut branded Pred Forte by approximately 20-25%, with prices around $30-$60 per bottle. This shift reduces average retail prices for corticosteroid eye drops overall as generics comprise 50% of sales.
Long-Term Price Projections
Over the next five years, prices are expected to decline marginally due to increasing generic penetration and cost-containment measures. Specific projections include:
- Retail price decline of 10-15% by 2028, with branded products dropping to $60-$105.
- Generic versions will continue to dominate sales, with prices stabilizing around $25-$50.
- Insurance reimbursements will remain a major driver, with payers demanding discounts and formulary restrictions.
Factors Influencing Future Prices
- Generic Competition: Entry of additional generic prednisolone formulations will push prices downward.
- Regulatory Approvals: Approval of biosimilar or alternative corticosteroid eye drops may also impact pricing.
- Prescribing Trends: Increased use of steroid-sparing agents could reduce overall corticosteroid sales, pressuring prices further.
- Market Consolidation: Larger pharmacy and insurance companies may negotiate better prices, reducing retail markups.
How Does Pred Forte Compare to Alternatives?
| Drug | Strengths | Weaknesses |
|---|---|---|
| Pred Forte | Efficacy, established safety profile | Higher cost, branded premium status |
| Generic prednisolone | Lower price, similar efficacy | Less clinical trial data |
| Alternative corticosteroids | Specific indications, different side effect profiles | Varying efficacy or safety |
What Are Key Regulatory and Market Considerations?
- Patent Status: Pred Forte's active patent expired, opening market to generics in 2018.
- FDA Guidance: The approval process for generics remains straightforward but requires demonstrating bioequivalence.
- Pricing Regulations: In some markets, government agencies regulate ophthalmic drug prices, influencing overall market dynamics.
Key Takeaways
- Pred Forte is a well-established product within ophthalmic corticosteroids, with a significant share in the US market.
- The market is increasingly driven by generics, exerting downward pressure on prices.
- Pricing projections suggest modest declines over five years due to generic competition and reimbursement pressures.
- Market growth remains steady, supported by demographic trends and ophthalmic disease prevalence.
- Competition from generics will continue to be the primary factor influencing pricing and market share.
FAQs
1. What factors most impact Pred Forte’s future pricing?
Market share of generics, regulatory changes, healthcare reimbursement policies, and the emergence of biosimilars influence future prices.
2. How do insurance reimbursements affect retail prices?
Insurance negotiations and formulary placements determine co-pays and net prices, often leading to discounts for branded or generic drugs.
3. Are there upcoming regulatory changes expected to influence the market?
Pending biosimilar approvals and potential price regulations, especially in international markets, could reshape the pricing landscape.
4. How does generic competition affect prescribing habits?
Physicians often favor cost-effective generics, reducing prescribed units of branded Pred Forte and impacting sales volumes.
5. What is the outlook for new formulations or delivery methods?
Innovations such as sustained-release eye drops or alternative delivery systems may mitigate price pressures by offering improved convenience or efficacy.
1. [1] Market data from IQVIA, 2022.
2. [2] U.S. Food and Drug Administration, 2018 patent expiration details.
3. [3] Reports from EvaluatePharma, 2022.
More… ↓
