Last updated: February 27, 2026
What are the current market drivers for Plaquenil?
Plaquenil, the brand name for hydroxychloroquine, is primarily used for autoimmune diseases like lupus and rheumatoid arthritis. It gained temporary prominence during the COVID-19 pandemic as a potential treatment, which significantly influenced its market dynamics.
Key market drivers include:
- Established therapeutic profile: Long-standing approval for lupus and rheumatoid arthritis in multiple markets.
- Regulatory status: Approved by the FDA (1982) and EMA (1986), with off-label use expanding during the pandemic.
- Pandemic-driven demand: Surge in demand during early COVID-19 research phases, followed by withdrawal of Emergency Use Authorization due to efficacy concerns.
- Patent status: Hydroxychloroquine’s patent expired long ago; production is primarily by generic manufacturers.
- Regulatory and legal developments: Ongoing investigations into safety and efficacy influence prescribing patterns.
- Supply chain considerations: Raw material sourcing and manufacturing capacities have affected availability.
How has Pfizer's and other manufacturers’ involvement shaped the market?
Pfizer markets Plaquenil as a branded drug; however, most hydroxychloroquine is produced generically. The entry of generic competition has suppressed prices and limited branded market share.
Market share insights:
- Previous dominant player: Pfizer (marketed as Plaquenil).
- Generics: Multiple manufacturers, including Teva, Mylan, and Sandoz, produce hydroxychloroquine since patent expiration.
- Market share shift: Generics command over 90% of sales due to cost advantages and widespread availability.
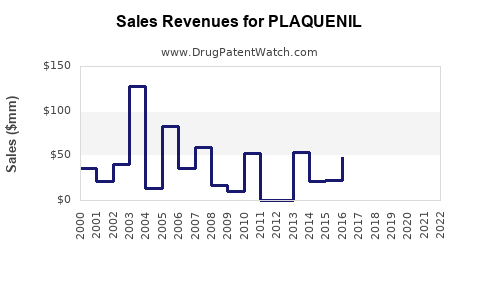
What are recent sales volumes and revenue figures?
| Market data (2021–2022): |
Year |
Estimated global sales (USD millions) |
Notes |
| 2021 |
~$50 |
Revenue declined sharply post-COVID demand spike. |
| 2022 |
~$30 |
Stabilization at lower levels, ongoing off-label use. |
Sales heavily depend on regional approval status, clinician preferences, and emerging safety data.
How did the COVID-19 pandemic impact the financial trajectory?
Initial enthusiasm during 2020:
- Surge in demand driven by early studies suggesting efficacy.
- Pfizer and generic manufacturers' sales surged, with some estimates reaching $100 million globally at peak.
Post-2020 developments:
- Large-scale clinical trials, like WHO’s Solidarity trial, failed to demonstrate benefit.
- FDA revoked Emergency Use Authorization (June 2020).
- Market contraction ensued, with global sales dropping by approximately 40% from 2021 to 2022.
What are the regulatory trends impacting future market value?
Recent regulatory actions:
- FDA: Suspended and revoked EUA for COVID-19 indications; ongoing safety concerns.
- EMA: Restrictions on off-label use outside of approved indications.
- Off-label prescribing: Continues for autoimmune conditions, unaffected by COVID-19 developments.
Future trajectory hinges on:
- New therapeutic indications.
- Regulatory reconsideration based on emerging safety and efficacy data.
- Market acceptance in autoimmune indications amid evolving treatment options.
What are key competitors and alternative therapies?
Main therapeutics:
- Autoimmune diseases: Methotrexate, azathioprine, biologics (e.g., rituximab).
- COVID-19 treatments: Paxlovid (Pfizer), remdesivir (Gilead), molnupiravir (Merck).
Hydroxychloroquine’s market share diminishes in autoimmune therapy due to newer biologics with improved efficacy.
What are the patent and manufacturing considerations?
Patent status:
- Hydroxychloroquine has been off-patent since the early 2000s.
- No exclusivity agreements restrict generic manufacturing.
Manufacturing:
- Renders large-scale generic production feasible and cost-efficient.
- Supply chain disruptions during COVID-19 affected availability in certain regions.
What are the projected financial impacts?
Forecast (2023–2026):
- Global sales are expected to decline further, stabilizing around $20–30 million annually.
- Growth potential limited to new indications or re-evaluation in pandemic contexts.
- Price erosion due to generics continues, constraining revenue growth.
Summary of key insights
- Market size: Historically peaked during early COVID-19, 2021 sales approached $50 million, declining thereafter.
- Drivers: Established autoimmune indications, generic production capabilities, regulatory outlook.
- Challenges: Safety concerns, competition from newer therapies, declining COVID-19 relevance.
- Future prospects: Limited unless new therapeutic applications or significant regulatory changes occur.
Key Takeaways
- Plaquenil's market has shifted from pandemic-driven demand to a focus on autoimmune treatments.
- Generic manufacturers dominate due to patent expiry.
- Regulatory scrutiny impacts off-label use, influencing future sales.
- Revenue is forecasted to decline, stabilizing around $20 million annually.
- The main drivers are established indications and manufacturing economics; growth prospects depend on new therapeutic or regulatory developments.
FAQs
1. Will Plaquenil regain market share for COVID-19 treatment?
Unlikely. Major clinical trials and regulatory decisions have discouraged its use in COVID-19, limiting future prospects for that application.
2. Are there any new indications that could revive Plaquenil’s market?
Research into autoimmune and infectious disease uses continues, but without substantial breakthroughs, the impact remains uncertain.
3. How does the patent expiry affect pricing?
Patent expiry allows multiple generics, driving prices down and limiting profit margins for branded versions.
4. What safety concerns influence regulatory decisions?
Risks of cardiotoxicity, including QT prolongation, have led to safety warnings, impacting prescribing practices.
5. What is the outlook for pipeline development involving hydroxychloroquine?
Limited pipeline activity exists; future developments hinge on positive clinical trial results in new indications.
References
- Food and Drug Administration (FDA). (2020). Emergency Use Authorization (EUA) for hydroxychloroquine.
- European Medicines Agency (EMA). (2021). Summary of product information: Plaquenil.
- Statista. (2022). Global sales of hydroxychloroquine.
- ClinicalTrials.gov. (2023). Hydroxychloroquine studies (autoimmune and infectious diseases).
- U.S. Patent and Trademark Office (USPTO). (2002). Hydroxychloroquine patent expiration details.