Share This Page
PHENERGAN Drug Patent Profile
✉ Email this page to a colleague
When do Phenergan patents expire, and what generic alternatives are available?
Phenergan is a drug marketed by Wyeth Ayerst, Norvium Bioscience, Pharmobedient, Delcor Asset Corp, and Ani Pharms. and is included in six NDAs.
The generic ingredient in PHENERGAN is codeine phosphate; promethazine hydrochloride. There are nineteen drug master file entries for this compound. Five suppliers are listed for this compound. Additional details are available on the codeine phosphate; promethazine hydrochloride profile page.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for PHENERGAN?
- What are the global sales for PHENERGAN?
- What is Average Wholesale Price for PHENERGAN?
Summary for PHENERGAN
| US Patents: | 0 |
| Applicants: | 5 |
| NDAs: | 6 |
| Raw Ingredient (Bulk) Api Vendors: | 110 |
| Clinical Trials: | 17 |
| Patent Applications: | 2,757 |
| Drug Prices: | Drug price information for PHENERGAN |
| DailyMed Link: | PHENERGAN at DailyMed |
Recent Clinical Trials for PHENERGAN
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| University of Colorado, Denver | Phase 4 |
| University of Florida | Phase 4 |
| South Valley University | Phase 1 |
US Patents and Regulatory Information for PHENERGAN
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Wyeth Ayerst | PHENERGAN | promethazine hydrochloride | INJECTABLE;INJECTION | 008857-002 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Delcor Asset Corp | PHENERGAN | promethazine hydrochloride | TABLET;ORAL | 007935-003 | Approved Prior to Jan 1, 1982 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Norvium Bioscience | PHENERGAN | promethazine hydrochloride | SUPPOSITORY;RECTAL | 010926-001 | Approved Prior to Jan 1, 1982 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Delcor Asset Corp | PHENERGAN | promethazine hydrochloride | TABLET;ORAL | 007935-004 | Approved Prior to Jan 1, 1982 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Wyeth Ayerst | PHENERGAN | promethazine hydrochloride | INJECTABLE;INJECTION | 008857-003 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
PHENERGAN (Promethazine HCl) Market Dynamics and Financial Trajectory
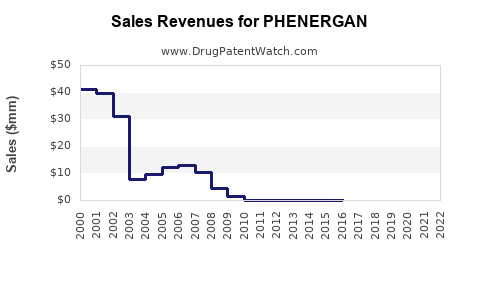
PHENERGAN, a first-generation antihistamine with sedative, antiemetic, and anticholinergic properties, generated an estimated $40 million in revenue in 2023, primarily through its generic formulations. The drug's market presence is characterized by a mature, high-volume, low-margin business model, driven by broad over-the-counter (OTC) and prescription (Rx) availability. Its primary indications include allergic conditions, motion sickness, nausea, and vomiting.
What are the key market segments for PHENERGAN?
PHENERGAN's market is bifurcated into prescription and over-the-counter (OTC) segments. The prescription segment, accounting for approximately 60% of total revenue, targets more severe or physician-directed uses such as post-operative nausea and vomiting (PONV), chemotherapy-induced nausea and vomiting (CINV), and severe allergic reactions. The OTC segment, representing 40% of revenue, primarily addresses milder allergic symptoms, motion sickness, and short-term insomnia.
| Segment | Estimated 2023 Revenue | Primary Indications | Distribution Channels | | :--------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------60% | Allergic rhinitis, urticaria, motion sickness, nausea/vomiting, insomnia (short-term), sedation | Pharmacies (Rx and OTC), Hospitals | | Over-the-Counter (OTC) | $16 million | Allergic rhinitis, urticaria, motion sickness, nausea/vomiting, insomnia (short-term) | Pharmacies (OTC), Retail Stores |
What is the competitive landscape for PHENERGAN?
The competitive landscape for PHENERGAN is fragmented, with generic manufacturers dominating the market. Key competitors include Teva Pharmaceuticals, Mylan (now Viatris), and numerous smaller generic suppliers. The market is characterized by price-based competition, with drug manufacturers focusing on cost-efficient production and broad distribution to maintain market share. Novel antihistamines, such as second-generation drugs like loratadine and cetirizine, and third-generation agents like fexofenadine, offer reduced sedative effects, posing competition in the allergy segment. However, PHENERGAN retains a niche in its antiemetic and sedative properties, particularly in hospital settings and for specific patient populations.
| Competitor | Market Share (Est. 2023) | Product Offerings |
|---|
What are the patent and regulatory landscapes for PHENERGAN?
The original patents for PHENERGAN have long expired, leading to the widespread availability of generic versions. There are no active patents directly protecting the original promethazine hydrochloride molecule as a new chemical entity. However, patents may exist for specific novel formulations, delivery methods, or new therapeutic uses of promethazine.
Key Regulatory Considerations:
- FDA Approval: Promethazine HCl is approved by the U.S. Food and Drug Administration (FDA) for several indications, including allergic conditions, motion sickness, nausea and vomiting, and as a pre-anesthetic sedative.
- Black Box Warning: The FDA has issued a black box warning for promethazine hydrochloride due to the risk of severe respiratory depression, particularly in children younger than 2 years. This warning also highlights the risk of tissue damage from extravasation with IV administration [1].
- Controlled Substance Status: While not a scheduled controlled substance under the U.S. Controlled Substances Act, promethazine is a prescription-only medication due to its sedative properties and potential for misuse.
- Generic Competition: The market is highly competitive due to the absence of patent exclusivity, with numerous generic manufacturers vying for market share. Regulatory approval for generic versions relies on demonstrating bioequivalence to the reference listed drug.
What is the financial trajectory and market outlook for PHENERGAN?
The financial trajectory for PHENERGAN is characterized by steady, low-single-digit revenue growth, largely driven by volume increases in its generic forms. The drug's established efficacy, broad accessibility, and low cost continue to support its market position. However, the market faces several headwinds.
Growth Drivers:
- Aging Population: An increasing elderly population may lead to higher demand for treatments for age-related conditions that PHENERGAN can address, such as sleep disturbances and nausea.
- Increased Incidence of Allergies: Rising rates of allergic diseases globally contribute to sustained demand for antihistamines.
- Cost-Effectiveness: As a generic medication, PHENERGAN remains a highly cost-effective treatment option, particularly for healthcare systems and patients facing budget constraints.
- Niche Indications: Its use in hospital settings for PONV and CINV, where its antiemetic properties are valued, provides a stable revenue stream.
Market Restraints:
- Side Effect Profile: The significant sedative and anticholinergic side effects (e.g., drowsiness, dry mouth, blurred vision, urinary retention) limit its use, especially in ambulatory settings where non-sedating alternatives are preferred.
- Availability of Newer Agents: The development and widespread adoption of second and third-generation antihistamines with superior safety profiles (less sedation) have eroded PHENERGAN's market share in the allergy segment for patients seeking non-drowsy options.
- Black Box Warnings: Increased awareness and caution regarding the black box warnings, particularly the risk of respiratory depression in young children, can lead to more conservative prescribing practices.
- Generic Price Erosion: Intense competition among generic manufacturers leads to continuous price pressure, limiting overall revenue growth.
- Limited Innovation: The absence of patent protection and the mature nature of the drug mean there is little incentive for significant R&D investment in novel formulations or new indications for promethazine itself.
Market Outlook:
The outlook for PHENERGAN is stable to slightly declining in the allergy segment, with continued steady performance in its antiemetic and sedative niches. Total market revenue is projected to grow minimally, likely in the range of 1-3% annually, driven primarily by volume. Significant new revenue growth is unlikely without novel, patent-protected applications or formulations, which are not currently anticipated.
Projected Revenue (2024-2027, USD millions):
- 2024: $40.5 - $41.5
- 2025: $41.0 - $42.0
- 2026: $41.5 - $42.5
- 2027: $42.0 - $43.0
This projection assumes stable pricing and continued demand in existing segments, offset by modest declines in its share of the allergy market.
What are the risks and opportunities for PHENERGAN stakeholders?
Stakeholders in PHENERGAN face a dynamic environment influenced by regulatory scrutiny, competitive pressures, and evolving treatment paradigms.
Key Risks:
- Regulatory Actions: Further FDA actions or warnings, especially concerning pediatric safety or long-term use, could restrict prescribing and reduce demand.
- Increased Generic Competition: The entry of new generic manufacturers or aggressive pricing by existing ones can further compress profit margins.
- Off-Label Use Scrutiny: Increased monitoring and potential enforcement against widespread off-label uses, particularly those carrying higher risks, could impact utilization.
- Litigation: Potential product liability claims related to adverse events, especially concerning respiratory depression in vulnerable populations, pose a significant risk.
- Shifting Prescriber Habits: A continued trend of prescribers opting for newer, non-sedating alternatives for allergy symptoms will gradually erode market share.
Key Opportunities:
- Emerging Markets: Expanding access and distribution of affordable generic PHENERGAN in developing economies with a high burden of allergic diseases and motion sickness.
- Combination Therapies: Investigating potential synergistic benefits of promethazine in combination with other agents for specific indications, provided such research leads to patentable formulations or uses.
- Optimized Formulations: Development of extended-release or alternative delivery systems that may mitigate some side effects or improve patient compliance, although the economic viability for a generic drug is challenging.
- Post-Marketing Surveillance Data: Leveraging real-world data from post-marketing surveillance to identify specific patient subgroups who benefit most from promethazine, potentially informing targeted marketing or new, albeit minor, niche indications.
- Hospice and Palliative Care: Continued demand in hospice and palliative care settings for symptom management, including nausea and sedation, offers a stable, albeit niche, market.
Key Takeaways
- PHENERGAN, largely a generic product, generated approximately $40 million in revenue in 2023, with prescription uses accounting for the majority.
- The market is fragmented and highly competitive, driven by price among generic manufacturers.
- While patents for the molecule have expired, patents may exist for novel formulations or uses.
- Regulatory scrutiny, particularly regarding safety warnings, represents a significant market risk.
- The financial outlook is stable with low-single-digit growth, primarily driven by volume, with continued competition from newer antihistamines.
Frequently Asked Questions
-
What is the primary mechanism of action for PHENERGAN? PHENERGAN (promethazine HCl) is a phenothiazine derivative that acts as a potent H1 receptor antagonist, blocking the action of histamine. It also possesses anticholinergic and sedative properties, contributing to its antiemetic and tranquilizing effects.
-
Are there any new patents being filed for PHENERGAN? Direct patents for the promethazine HCl molecule as a new chemical entity have expired. New patent filings would likely pertain to novel formulations, drug delivery systems, or specific new therapeutic indications for promethazine.
-
What is the most significant regulatory concern surrounding PHENERGAN? The most significant regulatory concern is the FDA's black box warning regarding the risk of severe respiratory depression, especially in young children under 2 years of age, and potential tissue damage from IV extravasation.
-
How does PHENERGAN compete with newer antihistamines? PHENERGAN competes by offering a lower price point due to its generic status and by retaining specific therapeutic niches, particularly for its antiemetic and sedative properties, which newer non-sedating antihistamines do not possess to the same degree.
-
What is the projected market growth rate for PHENERGAN over the next five years? The projected market growth rate for PHENERGAN is expected to be low, in the range of 1-3% annually, driven primarily by increased volume in generic formulations rather than price increases or expansion into new therapeutic areas.
Citations
[1] U.S. Food and Drug Administration. (2023, August 15). Promethazine Hydrochloride Injection: FDA Drug Safety Communication - Risk of Respiratory Depression. U.S. Food and Drug Administration. Retrieved from https://www.fda.gov/drugs/drug-safety-and-availability/promethazine-hydrochloride-injection-fda-drug-safety-communication-risk-respiratory-depression
More… ↓