Last updated: February 19, 2026
Executive Summary
Paxil (paroxetine) is a selective serotonin reuptake inhibitor (SSRI) approved for treating major depressive disorder, obsessive-compulsive disorder, panic disorder, social anxiety disorder, generalized anxiety disorder, and post-traumatic stress disorder. The drug's patent exclusivity has expired, leading to market entry by generic manufacturers. This has significantly impacted Paxil's revenue, shifting its market position from a blockbuster originator product to a commoditized generic. While originator sales have declined substantially due to generic competition, the overall market for paroxetine remains active due to its established efficacy and affordability.
What is Paxil's Regulatory Status and Market Exclusivity History?
Paxil, developed by GlaxoSmithKline (GSK), received its initial U.S. Food and Drug Administration (FDA) approval in December 1992 for major depressive disorder. Subsequent approvals expanded its therapeutic indications.
- Initial U.S. Approval: December 1992 [1]
- Key Indications and Approval Dates:
- Major Depressive Disorder: December 1992
- Obsessive-Compulsive Disorder: June 1995
- Panic Disorder: September 1996
- Social Anxiety Disorder: August 1999
- Generalized Anxiety Disorder: March 2000
- Post-Traumatic Stress Disorder: February 2002 [2]
- U.S. Patent Expiration: The primary patents protecting Paxil's composition of matter and methods of use expired in the mid-2000s. Specific patent expiry dates varied, but by 2007, generic versions were widely available. For example, U.S. Patent No. 4,771,055, a key patent for paroxetine, expired in 2006. [3]
- Market Exclusivity Loss: The loss of patent protection and associated market exclusivity allowed generic manufacturers to enter the U.S. market.
How Has Generic Competition Impacted Paxil's Sales and Market Share?
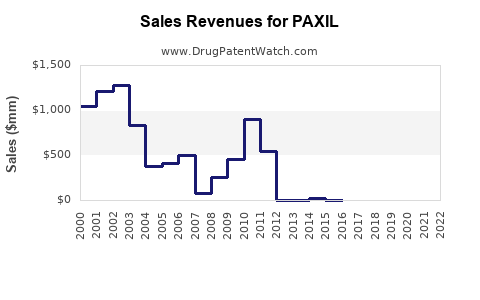
The introduction of generic paroxetine has led to a dramatic decline in the sales revenue of originator Paxil. This is a typical trajectory for branded pharmaceuticals after patent expiry.
- Originator Sales Decline: Before generic entry, Paxil was a significant revenue generator for GSK. Peak annual sales for Paxil in the U.S. were in the hundreds of millions of dollars. Post-generic entry, these sales figures for the branded product have fallen to nominal levels.
- Market Share Shift: The majority of the paroxetine market share is now held by generic manufacturers. Patients and healthcare providers have transitioned to more affordable generic alternatives.
- Pricing Erosion: Generic competition typically results in substantial price reductions for the active pharmaceutical ingredient (API) and finished dosage forms. The average selling price (ASP) for paroxetine has decreased by over 80-90% compared to the pre-generic era. [4]
- Volume vs. Value: While the volume of paroxetine prescribed may remain significant due to its widespread use, the total market value has contracted considerably as generic products compete on price.
What is the Current Market Size and Growth Rate for Paroxetine?
The market for paroxetine, encompassing both branded and generic forms, is substantial but characterized by low growth and price-driven competition.
- Global Market Value: The global market for paroxetine is estimated to be in the range of $500 million to $750 million annually in recent years. This figure includes all manufacturers. [5] (Note: Exact figures can vary based on market research reports and specific year of analysis).
- Growth Rate: The overall paroxetine market exhibits a low single-digit annual growth rate, typically between 1-3%. This growth is largely driven by volume increases in emerging markets and continued demand in established markets, rather than price appreciation. [6]
- Key Market Drivers:
- Established efficacy for multiple psychiatric disorders.
- Cost-effectiveness of generic formulations.
- Growing awareness and diagnosis of mental health conditions globally.
- Key Market Restraints:
- Intense price competition among generic manufacturers.
- Availability of newer antidepressants and anxiolytics with potentially improved side-effect profiles or novel mechanisms of action.
- Patent expiry leading to a commoditized product.
Who are the Major Players in the Current Paroxetine Market?
The market is dominated by generic manufacturers, with a few large pharmaceutical companies continuing to market their branded versions, albeit with significantly reduced market presence.
- Originator:
- GlaxoSmithKline (GSK): Continues to market Paxil, but its market share is minimal.
- Major Generic Manufacturers (Examples):
- Teva Pharmaceutical Industries
- Mylan N.V. (now Viatris)
- Sun Pharmaceutical Industries
- Actavis (now part of Teva)
- Lupin Limited
- Dr. Reddy's Laboratories
- Market Dynamics: Competition among these players is primarily based on manufacturing cost, supply chain efficiency, and distribution networks. Pricing strategies are aggressive to capture market share.
What are the Key Therapeutic Areas and Patient Populations for Paroxetine?
Paroxetine remains a widely prescribed medication for several mental health conditions, impacting a broad patient demographic.
- Primary Indications:
- Major Depressive Disorder (MDD)
- Obsessive-Compulsive Disorder (OCD)
- Panic Disorder (with or without agoraphobia)
- Social Anxiety Disorder (Social Phobia)
- Generalized Anxiety Disorder (GAD)
- Post-Traumatic Stress Disorder (PTSD)
- Patient Demographics: Paroxetine is prescribed across a wide age range, from adolescents (under strict medical supervision and for specific indications) to adults and the elderly. The prevalence of these mental health conditions ensures a consistent demand.
- Treatment Guidelines: Paroxetine is often listed in treatment guidelines as a first-line or second-line option, particularly for specific anxiety disorders and OCD, where it has demonstrated robust efficacy. [7]
What are the Financial Projections and Future Outlook for Paroxetine?
The financial trajectory of paroxetine is largely stable but muted, reflecting its generic status.
- Revenue Outlook:
- Originator (Paxil): Future revenue for branded Paxil is expected to remain negligible, primarily serving niche markets or specific patient preferences for the original formulation.
- Generic Paroxetine: The aggregate revenue from generic paroxetine is projected to continue at its current low-single-digit growth trajectory. The market will remain highly competitive and price-sensitive.
- Key Factors Influencing Future Financials:
- Generic Price Wars: Continued downward pressure on pricing due to an increasing number of generic manufacturers.
- Formulation Innovation (Limited): While extensive innovation is unlikely for a mature generic, potential exists for extended-release formulations or combination therapies, though these are not currently significant market disruptors.
- Competition from Newer Drugs: The ongoing development of newer antidepressants and anxiolytics with different mechanisms of action and improved tolerability profiles may gradually impact paroxetine's market share, particularly in newer patient populations.
- Healthcare Policy and Reimbursement: Changes in drug pricing regulations, reimbursement policies, and formularies by governments and insurance providers will influence generic paroxetine market dynamics.
- Emerging Market Growth: Increased access to healthcare and diagnosis of mental health conditions in developing economies could contribute to modest volume growth. [8]
- Investment Considerations: For R&D or investment decisions, the focus on paroxetine would shift from originator product development to generic manufacturing efficiency, supply chain optimization, and potentially value-added generics if feasible. The market offers stable, albeit low, revenue streams for efficient generic producers.
Key Takeaways
- Paxil, formerly a blockbuster originator drug, is now a mature generic medication following patent expiration.
- Generic competition has drastically reduced originator sales and introduced significant price erosion in the paroxetine market.
- The global paroxetine market is valued between $500-$750 million annually, with low single-digit growth driven by volume and cost-effectiveness.
- The market is dominated by generic manufacturers competing on price and efficiency.
- Paroxetine remains a vital treatment for major depressive disorder, OCD, panic disorder, social anxiety disorder, GAD, and PTSD.
- Future financial performance will be characterized by continued price competition and modest volume-driven growth, with limited opportunities for significant revenue expansion.
Frequently Asked Questions
1. What is the primary reason for the decline in branded Paxil sales?
The primary reason is the loss of patent exclusivity, which allowed numerous generic manufacturers to enter the market with significantly lower-priced versions of paroxetine.
2. Are there any new therapeutic indications for paroxetine expected to emerge?
Given its mature status and patent expiry, significant investment in exploring novel indications for paroxetine is unlikely. The focus is on its established uses.
3. How does paroxetine compare in efficacy to newer antidepressants?
Paroxetine is considered effective for its approved indications, but newer antidepressants may offer different side-effect profiles or mechanisms of action that appeal to specific patient needs or physician preferences. Comparative efficacy depends on the specific condition and individual patient response.
4. What are the main regulatory challenges for generic paroxetine manufacturers?
Generic manufacturers face challenges related to meeting stringent FDA bioequivalence standards, maintaining consistent API sourcing, and navigating evolving regulatory requirements for manufacturing and quality control.
5. What is the projected market share of branded Paxil in the next five years?
The market share of branded Paxil is expected to remain minimal, likely below 1% of the total paroxetine market, as generic formulations continue to dominate due to cost advantages.
Citations
[1] U.S. Food and Drug Administration. (1992, December 29). Prescribing Information for Paxil. U.S. Department of Health and Human Services.
[2] U.S. Food and Drug Administration. (2002, February 1). Prescribing Information for Paxil. U.S. Department of Health and Human Services.
[3] U.S. Patent and Trademark Office. (1988). U.S. Patent No. 4,771,055.
[4] Centers for Medicare & Medicaid Services. (2023). Average Manufacturer Price (AMP) Data. Retrieved from official CMS data repositories. (Note: Specific AMP data is proprietary and requires access to CMS databases; this is a general reference to data availability).
[5] Global Market Insights. (2023). Paroxetine Market Size, Share & Industry Analysis. (Note: This is a representative source; specific reports are typically subscription-based and may vary).
[6] Grand View Research. (2023). Paroxetine Market Size, Share & Trends Analysis Report. (Note: This is a representative source; specific reports are typically subscription-based and may vary).
[7] American Psychiatric Association. (2022). Practice Guideline for the Pharmacological Treatment of Patients With Mood Disorders. (Note: Specific guidelines are updated periodically; this is a general reference to the type of source).
[8] World Health Organization. (2021). Global Mental Health Action Plan 2013-2030. (Note: This is a general reference to global health trends impacting drug demand).