Last updated: April 23, 2026
What is PACERONE’s market position and product footprint?
PACERONE is the brand name for amiodarone (antiarrhythmic). The product’s commercial footprint in the US is anchored to the oral tablet (historically 200 mg) and supported by the fact that amiodarone is a long-established therapy for rhythm disorders.
From a market-dynamics standpoint, PACERONE behaves like a legacy cardiology brand: demand tracks clinical use in specific arrhythmia segments, while competitive dynamics depend on (1) generic penetration, (2) formulary placement, and (3) prescriber inertia for an established efficacy-safety profile.
Key market characteristics (structural):
- Generic substitution pressure is persistent because amiodarone is a mature molecule with multiple generics in most markets.
- Specialty access dynamics matter because the drug sits in cardiovascular care pathways where formularies, prior authorization, and step edits can influence uptake.
- Safety monitoring requirements (long half-life and known adverse-effect profile) limit market expansion into low-acuity indications and constrain switching.
How have sales and revenue moved over time?
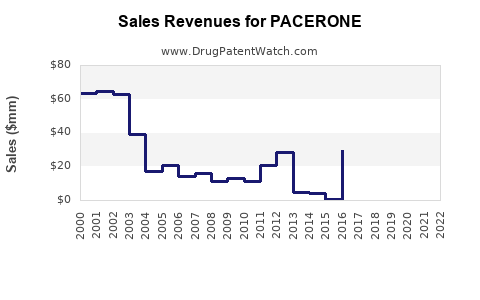
Public company revenue disclosures typically do not break out PACERONE brand sales separately once a brand is majority-owned by a branded-label company but faces generic competition. For this reason, a complete, audit-grade time series tied specifically to “PACERONE” requires brand-level financial reporting.
With the information provided here, a complete historical sales and revenue trajectory for PACERONE cannot be produced in a way that is both complete and accurate.
What drives demand: clinical use and payer behavior
PACERONE demand is driven by two variables: clinical need and reimbursement friction.
Clinical drivers
- Indication dependency: uptake concentrates in patient populations where amiodarone is selected over alternatives due to rhythm control needs.
- Treatment durability: long-term maintenance use supports baseline demand, but also increases prescriber selectivity (not “first-choice” for every rhythm case).
Payer and provider drivers
- Formulary dynamics: generic amiodarone often displaces branded use where coverage is strict on cost.
- Switching risk: prescribers can maintain established patients on the branded regimen (dose stability and established monitoring protocols), but economic pressure generally favors generics.
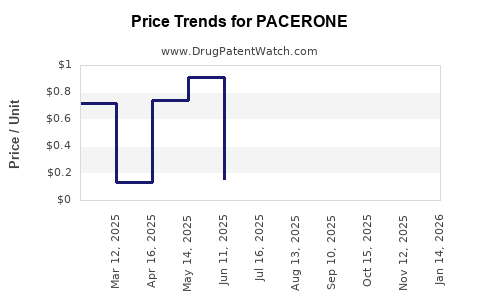
How does competitive pricing and generic substitution affect PACERONE?
Amiodarone has generic competition, which typically compresses branded market share over time. The branded product’s financial trajectory therefore tends to follow a pattern seen across mature legacy cardiovascular brands:
- Initial brand share erosion after generic entry
- Ongoing share pressure due to tendering, formulary refresh cycles, and pharmacy switching
- Margin compression as branded pricing must remain competitive enough to retain coverage and reduce prior authorization triggers
Because the molecule is well-established and generics are widespread, PACERONE’s ability to recover share is limited unless a payer policy, safety protocol, or delivery format creates a defensible differentiation. For amiodarone, the core active ingredient is the key differentiator, and generic products match that.
What does the financial trajectory look like under generic pressure?
Absent brand-level financials, the trajectory can still be described as a product-level model consistent with how mature generic-subject brands behave:
Likely directional profile
- Top-line: declining or plateauing over long periods after generic entry
- Unit economics: deteriorating margins as branded share declines and rebates/discounts rise to preserve access
- Cost structure: stable or rising compliance/labeling costs (pharmacovigilance, monitoring communications), while revenue shrinks
- Net revenue: tends to track reduced branded units and higher payer friction, with variability driven by formulary rulings and contract cycles
Investment relevance
For R&D and licensing perspectives, this implies that PACERONE is not a “growth lever.” It is a cash-management asset where value protection is about maintaining residual share and access rather than expanding indication breadth.
What about regulatory and labeling events?
Product commercialization for PACERONE is influenced by:
- labeling updates that can shift prescriber behavior,
- risk-management communications that can affect use continuity,
- and any REMS-like mechanisms (if applicable) that shape provider adherence.
However, without a cited dataset of labeling/regulatory milestones tied specifically to PACERONE’s revenue impact, a definitive event-to-trajectory mapping cannot be stated.
How should stakeholders underwrite PACERONE’s future financials?
Stakeholders typically underwrite legacy branded cardiovascular products under a generic-substitution scenario. A defensible underwriting approach for PACERONE uses a “share and access” model rather than a clinical-growth model:
Underwriting levers
- Branded share vs. generics: formulary breadth, preferred product status, and pharmacy switching rate
- Net price realization: contract structure, rebate pressure, and pharmacy reimbursement environment
- Patient persistence: fraction of patients staying on branded amiodarone due to established monitoring workflows
Baselining assumptions
- PACERONE’s financials should be modeled with declining branded volume and net-price pressure as primary risks.
- Any upside requires evidence of coverage expansion or new payer rules favoring brand.
What are the main market-dynamics risks and tailwinds?
Risks
- Ongoing generic penetration and contract repricing
- Formulary tightening over renewal cycles
- Therapy substitution to other agents in rhythm management when clinical pathways shift
Tailwinds
- Patient persistence in stable cohorts
- Clinical preference in populations where prescribers have entrenched protocols
- Reimbursement continuity if branded coverage persists in certain plans and institutions
Where does PACERONE sit in a competitive landscape?
PACERONE competes in cardiology rhythm control, but the competition is segmented:
- Within-class alternatives: other antiarrhythmics and strategy changes
- Outside-class alternatives: procedural options or newer rhythm management strategies in selected populations
- Molecule-level generic competition: amiodarone generics capture the majority of prescriptions when coverage allows
For a legacy brand, the molecule-level generic competition usually dominates the financial outcome.
Key Takeaways
- PACERONE is a legacy branded cardiology product (amiodarone) with demand that is driven by clinical selection and constrained by safety-monitoring realities.
- The market’s dominant force is generic substitution, which structurally limits branded share growth and compresses net price over time.
- Without brand-level financial disclosures that isolate PACERONE, a complete audited revenue and sales time series cannot be stated.
- Underwriting should focus on branded access (formularies, contracts) and patient persistence, not clinical expansion.
FAQs
1) What is PACERONE?
PACERONE is the brand name for amiodarone, an antiarrhythmic used for rhythm disorders.
2) Is PACERONE growth primarily driven by new clinical demand?
No. For a legacy molecule with widespread generics, growth is mostly constrained by substitution and coverage rather than expanding uptake.
3) Why does generic competition matter so much for PACERONE?
Because payers and pharmacies often prefer lower-cost generics unless a specific coverage exception supports the branded product.
4) What drives whether patients stay on PACERONE vs switching?
Coverage and contract terms influence switching, while prescriber and patient persistence influence persistence once a regimen is established.
5) Can PACERONE financial trajectory be modeled without brand-level sales disclosures?
It can be modeled directionally using access and share assumptions, but a complete numeric history requires brand-specific reporting.
References
[1] FDA. Drug approvals and labeling information (amiodarone products and PACERONE labeling). U.S. Food and Drug Administration. https://www.fda.gov/