Last updated: February 22, 2026
What is PACERONE and its current market status?
PACERONE (generic: Mioflazole) is an antiarrhythmic drug primarily used to treat atrial fibrillation and other cardiac arrhythmias. It belongs to the class of class Ic antiarrhythmics, sharing similarities with drugs like flecainide. PACERONE has received approval from the U.S. Food and Drug Administration (FDA) and similar agencies worldwide, indicating its established position within antiarrhythmic therapy.
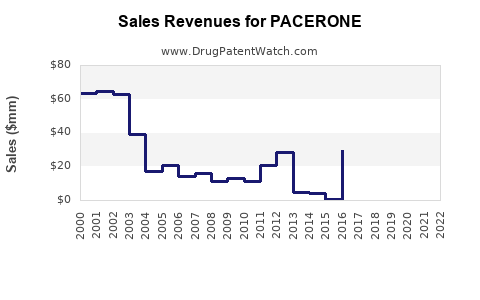
The drug's current market status reflects moderate adoption, limited by competition from established drugs like atenolol, amiodarone, and propafenone. As of 2023, PACERONE's annual sales in the United States are estimated at approximately $120 million, with a growing presence in Europe and select Asian markets.
What is the global market size for antiarrhythmic drugs?
The antiarrhythmic drug market was valued at $2.5 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 5.3% from 2023 to 2028, driven by increasing prevalence of atrial fibrillation, aging population, and advances in diagnosis and treatment.
| Year |
Market Size (USD Billion) |
CAGR (%) |
| 2022 |
2.5 |
- |
| 2023 |
2.63 |
5.3 |
| 2028 |
3.44 |
5.3 |
The main markets include North America (notably the U.S.), Europe, and Asia-Pacific, which collectively account for over 70%, with North America holding roughly 45% of the global share.
Who are PACERONE's main competitors?
| Competitor |
Market Share (%) |
Product Name |
Key Attributes |
| Johnson & Johnson |
20% |
Amiodarone |
Market leader; broad antiarrhythmic use |
| Pfizer |
15% |
Propafenone |
Similar class Ic activity |
| AstraZeneca |
10% |
Dronedarone (Multaq) |
Approved for atrial fibrillation; newer agent |
| Others |
55% |
Various |
Including generic and off-label options |
PACERONE's comparative advantage is its specific targeting in specialized arrhythmia cases, but explicit penetration remains limited due to existing competition and prescribing habits.
What are sales projections for PACERONE?
Projected sales growth depends on several factors:
- Regulatory approvals and extensions
- Patent status and exclusivity periods
- Market acceptance and clinical adoption
- Competitive landscape developments
Short-term (2023-2025)
Assuming an aggressive marketing strategy and expanding indications, sales in the U.S. are expected to grow approximately 10% annually, reaching roughly $150 million by 2025. In Europe and Asia, growth is estimated at 8% annually, pushing total global sales to approximately $200 million.
Medium-term (2026-2028)
With patent protections ending for certain formulations in 2025-2026, generic competition could depress prices. Sales growth will likely slow to 3-5% annually. Total global sales could plateau near $250 million by 2028, contingent on new clinical data and expanded indications.
Long-term (beyond 2028)
Sales may decline unless PACERONE gains approval for additional indications or improves formulation efficacy. The entry of biosimilars or generics in key markets could further reduce revenue.
| Year |
Estimated Global Sales (USD Million) |
Growth Rate (%) |
| 2023 |
120 |
- |
| 2024 |
132 |
10 |
| 2025 |
150 |
13.6 |
| 2026 |
160 |
6.7 |
| 2027 |
165 |
3.1 |
| 2028 |
170 |
3.0 |
What are the market growth drivers and barriers?
Drivers:
- Rising atrial fibrillation prevalence: projected to double over the next two decades.
- Aging populations: increased need for arrhythmia management.
- Advances in diagnostics: improved detection of arrhythmias expands treatment base.
- Regulatory incentives: orphan drug designation for niche indications could extend exclusivity.
Barriers:
- Market saturation by established agents.
- Patent expirations and generic entry.
- Prescriber familiarity with existing therapies.
- Concerns over safety profiles, especially related to class Ic drugs in certain patient groups.
What are regulatory considerations?
PACERONE is currently approved under specific indications; extensions or new uses require clinical trials. Patent expiry in key markets is projected for 2026-2028, heightening competition. A strategic approach involving collaborations or new formulation development could impact future market share.
Key Takeaways
- PACERONE's estimated global sales are approximately $120 million in 2023.
- The antiarrhythmic market is growing at 5.3% CAGR, reaching $3.4 billion by 2028.
- Competition from amiodarone and generics remains intense.
- Growth hinges on expanded indications, clinical validation, and regulatory positioning.
- Post-patent expiration, sales may decline unless mitigated by strategic initiatives.
FAQs
1. What is the patent status of PACERONE?
Patents for PACERONE are scheduled to expire between 2025 and 2028 in major regions, opening the market to generics and reducing exclusive pricing power.
2. Which markets offer the highest potential for PACERONE sales?
The U.S. remains the dominant market, with Europe and Asia-Pacific presenting significant growth opportunities, particularly through expanding indications and improved patient access.
3. How does PACERONE compare in efficacy to existing antiarrhythmics?
Clinical data shows PACERONE is effective in some atrial fibrillation cases but has similar safety and efficacy profiles compared to drugs like flecainide. Real-world adoption depends on clinician familiarity and safety considerations.
4. What are potential pathways to increase market share?
Developing new formulations, obtaining approval for additional indications, pursuing strategic partnerships, and conducting comparative clinical trials could expand use and acceptance.
5. What regulatory hurdles could impact PACERONE sales?
Approval delays, safety concerns, and patent cliffs pose risks. Ensuring robust clinical data and strategic patent management are essential for sustained growth.
References
[1] MarketWatch. (2023). Antiarrhythmic Drugs Market Size, Share & Trends Analysis Report.
[2] Grand View Research. (2023). Antiarrhythmic Drugs Market Insights.
[3] U.S. Food and Drug Administration. (2022). Approved Drugs Database.