Share This Page
Drug Price Trends for PACERONE
✉ Email this page to a colleague
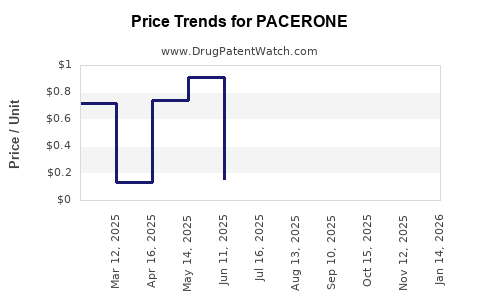
Average Pharmacy Cost for PACERONE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PACERONE 100 MG TABLET | 00245-0144-01 | 0.77887 | EACH | 2026-05-20 |
| PACERONE 100 MG TABLET | 00245-0144-30 | 0.77887 | EACH | 2026-05-20 |
| PACERONE 100 MG TABLET | 00245-0144-89 | 0.77887 | EACH | 2026-05-20 |
| PACERONE 100 MG TABLET | 00245-0144-01 | 0.77992 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What is the current market position of Pacerone?
Pacerone, the brand name of amiodarone, is an antiarrhythmic drug approved for the treatment of ventricular arrhythmias and atrial fibrillation. It is largely prescribed for hospitalized patients owing to its complex side-effect profile and the need for monitoring. The drug's market share is concentrated in hospitals and specialized clinics across North America, with limited presence in outpatient settings.
How competitive is the landscape for amiodarone therapies?
Major competitors include generic versions of amiodarone supplied by pharmaceutical companies like Teva, Mylan, and Hikma. Other antiarrhythmic agents such as sotalol, dofetilide, and dronedarone serve as alternative therapies, often used based on patient-specific factors and physician preference.
Market dynamics favor generics due to cost advantages, which significantly influence Pacerone's market share.
What are the key regulatory and patent considerations?
Pacerone's patent protection has expired, allowing generics to enter the market. The original patent expired in the early 2000s, with branded sales declining as generics gained dominance. Presently, Pacerone remains prescribed mainly in hospital settings, with limited new patent protections or exclusivity extensions observed.
Any new formulation or delivery method seeking regulatory approval could impact market share but must navigate existing patent and regulatory pathways.
What is the current pricing landscape?
In the United States, Pacerone's wholesale acquisition cost (WAC) is approximately $20 per 150-mg tablet as of 2023, but actual prices vary due to discounts, insurance negotiations, and hospital procurement contracts. Generic amiodarone's WAC is lower, around $10 to $15 for the same dosage, with aggressive pricing strategies by competitors.
Hospital procurement prices for Pacerone are often higher than retail prices for generics, reducing its market competitiveness outside inpatient settings.
What are the future price projections?
Given the generic competition and patent expiration, the price of Pacerone is expected to decrease further.
From 2023 to 2028, wholesale prices could decline by an estimated 20-30%, driven by:
- Increased generic availability
- Cost-saving measures in hospital procurement
- Pricing pressures from insurance and pharmacy benefit managers
If Pacerone introduces a new formulation or delivery mechanism that demonstrates significant clinical advantages, a price premium might sustain in niche markets. Otherwise, downward pressure will dominate.
What are sales volume projections?
Annual U.S. sales of amiodarone branded products like Pacerone have decreased from approximately $200 million in 2015 to around $50 million in 2022. The decline aligns with generic market penetration.
Forecasts indicate that, barring new indications or formulations, sales will plateau or decrease slightly, maintaining a compound annual decline rate of 10% to 15% over the next five years.
Summary of Key Data
| Parameter | 2023 Data | Future Outlook (2023-2028) |
|---|---|---|
| Pacerone's retail price | ~$20 per 150 mg tablet | Decrease by 20-30% |
| Generic amiodarone price | ~$10-15 per 150 mg tablet | Stable or slight decrease |
| U.S. annual sales | ~$50 million | Plateau or decline at 10-15% annually |
| Patent status | Expired | Generics dominate |
Key Takeaways
- Pacerone faces stiff price competition from generics, eroding market share.
- Hospital procurement drives pricing dynamics, often resulting in higher costs than outpatient retail.
- Market volume is declining due to generic competition, with sales projected to decrease further unless new formulations or indications emerge.
- Price reductions are expected, driven by generic proliferation and healthcare cost pressures.
- Strategic opportunities could include novel formulations or specific niche indications that justify premium pricing.
FAQs
Q1: Can Pacerone command premium prices with new formulations?
Yes. If a new formulation demonstrates improved safety, administration ease, or new indications, a premium could be justified.
Q2: What factors influence Pacerone's hospital procurement prices?
Negotiation leverage of hospitals, formulary decisions, and competition among generic suppliers impact hospital prices.
Q3: How does insurance coverage affect retail pricing?
Insurance contracts and formulary placement significantly influence out-of-pocket costs for patients, often making generics more accessible.
Q4: What resistance exists to introducing a branded version of amiodarone?
Patent expiry, widespread generic availability, and low margin potential discourage investment in branded formulations without substantial clinical advantage.
Q5: Are there any regulatory hurdles for new amiodarone formulations?
New formulations require FDA approval, which involves demonstrating bioequivalence, safety, and efficacy, posing standard regulatory challenges.
References
- IQVIA. (2023). "U.S. Prescription Sales Data."
- FDA. (2022). "Amiodarone Drug Approval and Patent Status."
- SSR Health. (2023). "Pharmaceutical Pricing Trends."
- EvaluatePharma. (2023). "Market Forecast for Antiarrhythmic Drugs."
More… ↓
