Last updated: February 19, 2026
What is Oxazepam's Current Market Position?
Oxazepam is a benzodiazepine prescribed for the short-term management of anxiety symptoms and alcohol withdrawal. Its primary mechanism of action involves enhancing the effect of the neurotransmitter gamma-aminobutyric acid (GABA) at the GABAA receptor, resulting in a reduction of central nervous system activity.
The global market for benzodiazepines, including oxazepam, is influenced by evolving treatment guidelines, the availability of alternative therapies, and regulatory scrutiny regarding potential for dependence and abuse. While specific market share data for oxazepam alone is often aggregated within broader benzodiazepine or anxiolytic categories, it remains a recognized therapeutic option.
Key market drivers include the prevalence of anxiety disorders and the increasing recognition of mental health issues. However, the market also faces headwinds from the development of newer anxiolytic classes, such as selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs), which often carry a more favorable long-term safety and dependence profile.
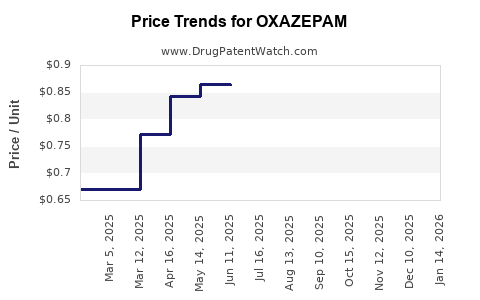
The compound's patent expiry in most major markets has led to widespread generic availability. This has significantly impacted pricing, making it a cost-effective option for healthcare systems and patients. The market landscape is therefore characterized by intense price competition among generic manufacturers.
What are the Key Patent Expiries and Generic Landscape Considerations for Oxazepam?
Oxazepam's foundational patents have long expired, allowing for generic entry. The original patent for oxazepam was filed by Wyeth (now part of Pfizer) and its primary market exclusivity periods concluded decades ago.
- Original Compound Patent: Expired in the late 20th century.
- Formulation/Manufacturing Patents: Various secondary patents related to specific formulations, delivery methods, or manufacturing processes may have expired or are nearing expiry. These typically offer shorter periods of protection (e.g., 10-15 years from filing).
The absence of active patent protection for the oxazepam molecule itself means that the market is dominated by generic manufacturers. This has led to:
- Price Erosion: Generic oxazepam is available at significantly lower price points compared to the original branded product.
- Fragmented Market: Multiple generic companies compete, often on price, leading to thin profit margins for manufacturers.
- Limited R&D Investment: Without patent protection, there is little incentive for significant new R&D investment in novel oxazepam formulations or indications by pharmaceutical companies. Investment is primarily focused on efficient, low-cost generic manufacturing.
The generic landscape for oxazepam typically involves companies with robust manufacturing capabilities and established distribution networks. These companies focus on optimizing production costs to remain competitive. Regulatory approvals for generic versions are based on demonstrating bioequivalence to the reference listed drug.
What is the Projected Financial Trajectory and Revenue Generation for Oxazepam?
The financial trajectory for oxazepam is largely shaped by its generic status and the competitive nature of the benzodiazepine market. Revenue generation is primarily driven by volume sales rather than premium pricing.
Key Financial Aspects:
- Volume-Driven Revenue: The revenue generated by oxazepam is directly correlated with the number of prescriptions dispensed. Its affordability makes it accessible for widespread use, contributing to sustained prescription volumes, particularly in regions with cost-sensitive healthcare systems.
- Price Sensitivity: As a generic drug, oxazepam is highly price-sensitive. Reimbursement rates by public and private payers are critical factors influencing manufacturer revenue. Intense competition among generic suppliers keeps wholesale prices low.
- Market Share Stability: While new anxiolytics emerge, oxazepam is likely to maintain a stable, albeit modest, market share due to its established efficacy for specific indications and its low cost. It is often prescribed for short-term relief or as part of a broader treatment regimen for alcohol withdrawal.
- Profit Margins: Manufacturers of generic oxazepam typically operate on lower profit margins compared to novel drug developers. Success hinges on high-volume production and efficient supply chain management.
- Regulatory Environment: Compliance with Good Manufacturing Practices (GMP) and ongoing regulatory approvals are essential operational costs. Any product recalls or manufacturing issues can have immediate financial repercussions.
Projected Trajectory:
The financial trajectory for oxazepam is expected to be relatively stable, with modest growth or slight decline influenced by:
- Aging Population and Chronic Conditions: Increasing prevalence of anxiety disorders and age-related conditions that may benefit from symptomatic relief could support demand.
- Generic Competition: Continued price pressure from new generic entrants or increased competition among existing players.
- Therapeutic Advancements: The adoption of newer, potentially more effective or safer anxiolytic agents could gradually erode oxazepam's market share.
- Prescribing Patterns: Shifts in physician prescribing habits influenced by clinical guidelines and drug safety concerns regarding benzodiazepine dependence.
Overall, oxazepam is a mature product in its lifecycle. Its financial trajectory is characterized by consistent, but not high-growth, revenue driven by its status as an affordable and accessible generic medication. Companies focusing on oxazepam are likely to be those with strong generic manufacturing capabilities and cost-efficiency.
What is the Competitive Landscape for Oxazepam and Related Anxiolytics?
The competitive landscape for oxazepam is multifaceted, involving direct competition from other benzodiazepines, alternative anxiolytic drug classes, and non-pharmacological interventions.
Direct Competitors (Benzodiazepines):
- Diazepam (Valium): A longer-acting benzodiazepine with similar indications, often used for muscle spasms and seizures.
- Lorazepam (Ativan): A mid-potency, intermediate-acting benzodiazepine, commonly prescribed for anxiety and insomnia.
- Alprazolam (Xanax): A high-potency, short-acting benzodiazepine, primarily used for panic disorder and anxiety.
- Clonazepam (Klonopin): A long-acting benzodiazepine used for seizure disorders and panic disorder.
These drugs compete directly with oxazepam for physician prescriptions and patient use in managing anxiety and related conditions. Competition within this class is often based on pharmacokinetic profiles (onset, duration of action), potency, and cost.
Alternative Anxiolytic Drug Classes:
- Selective Serotonin Reuptake Inhibitors (SSRIs):
- Examples: Fluoxetine (Prozac), Sertraline (Zoloft), Escitalopram (Lexapro).
- These are often considered first-line treatments for chronic anxiety disorders due to a generally lower risk of dependence and abuse compared to benzodiazepines.
- Serotonin-Norepinephrine Reuptake Inhibitors (SNRIs):
- Examples: Venlafaxine (Effexor XR), Duloxetine (Cymbalta).
- Also used for generalized anxiety disorder, social anxiety disorder, and panic disorder.
- Buspirone (Buspar): A non-benzodiazepine anxiolytic that acts as a partial serotonin receptor agonist. It has a slower onset of action but is not associated with the same risks of dependence as benzodiazepines.
- Antihistamines and Beta-Blockers: While not primary anxiolytics, some antihistamines (e.g., hydroxyzine) and beta-blockers (e.g., propranolol) are used for specific situational anxiety symptoms or somatic manifestations of anxiety.
These alternative classes represent a significant competitive threat, as they are frequently favored for long-term management due to improved safety profiles.
Non-Pharmacological Interventions:
- Psychotherapy: Cognitive Behavioral Therapy (CBT), exposure therapy, and other psychotherapeutic approaches are highly effective for anxiety disorders and are often recommended as primary or adjunctive treatments.
- Lifestyle Modifications: Exercise, mindfulness, stress management techniques, and dietary changes can play a supportive role in managing anxiety.
The competitive landscape is dynamic, with ongoing research into novel therapeutic targets and mechanisms of action for anxiety disorders. The trend is towards treatments with improved efficacy, safety, and reduced long-term risks. For oxazepam, its position is largely as a cost-effective, short-term solution, or for specific indications like alcohol withdrawal where its profile remains relevant.
What are the Key Regulatory Considerations and Potential Market Risks for Oxazepam?
The regulatory environment for oxazepam is stringent, reflecting the known risks associated with benzodiazepines. Key considerations and potential market risks include:
Regulatory Considerations:
- Controlled Substance Scheduling: Oxazepam is classified as a controlled substance in many jurisdictions (e.g., Schedule IV in the United States under the Controlled Substances Act). This classification mandates strict regulations on manufacturing, distribution, prescribing, and dispensing.
- Record-Keeping Requirements: Manufacturers and distributors must maintain detailed records of all transactions.
- Prescription Controls: Prescriptions often have limitations on refills and require periodic re-evaluation.
- Security Measures: Storage and handling must comply with security protocols to prevent diversion.
- Black Box Warnings: Prescribing information for oxazepam, like other benzodiazepines, typically includes warnings regarding the risks of:
- Dependence and Withdrawal: Potential for physical and psychological dependence with prolonged use, and severe withdrawal symptoms upon discontinuation.
- Abuse and Misuse: Risk of intentional or unintentional abuse and misuse.
- Respiratory Depression: Potentially fatal respiratory depression, especially when combined with opioids or other central nervous system depressants.
- Neonatal Withdrawal Syndrome: Risks to newborns if used during pregnancy.
- Prescribing Guidelines: Medical bodies and regulatory agencies periodically issue or update guidelines for the safe and appropriate use of benzodiazepines, influencing physician prescribing habits. These guidelines often emphasize short-term use and caution against long-term reliance.
- Manufacturing Standards: Generic manufacturers must adhere to strict Good Manufacturing Practices (GMP) to ensure product quality, purity, and consistency. Inspections by regulatory bodies (e.g., FDA, EMA) are routine.
Potential Market Risks:
- Increased Regulatory Scrutiny and Restrictions: Heightened awareness of benzodiazepine risks could lead to more restrictive prescribing policies, reduced insurance coverage for long-term use, or even outright bans for certain indications or patient populations.
- Litigation: Manufacturers may face litigation related to adverse events, dependence, or alleged marketing of the drug without adequate disclosure of risks.
- Competition from Safer Alternatives: Continued development and adoption of anxiolytic agents with more favorable safety profiles (e.g., SSRIs, SNRIs, novel non-benzodiazepine mechanisms) will continue to erode oxazepam's market share, particularly for chronic anxiety management.
- Drug Diversion and Abuse: Public health concerns and law enforcement efforts aimed at combating the misuse and diversion of controlled substances could indirectly impact the market through increased regulation and reduced accessibility.
- Reimbursement Challenges: Payers may increasingly favor newer, evidence-based treatments for chronic anxiety, potentially limiting reimbursement for oxazepam or requiring stringent justification for its use.
- Supply Chain Disruptions: As a generic product with potentially narrower profit margins, manufacturers may be less resilient to supply chain disruptions (e.g., raw material shortages, manufacturing issues) compared to higher-margin drugs.
The future of oxazepam in the market is thus closely tied to its ability to maintain its niche as a cost-effective, short-term treatment while navigating an evolving regulatory landscape and an increasing array of therapeutic alternatives.
Key Takeaways
Oxazepam, a mature generic benzodiazepine, occupies a cost-effective niche in the anxiolytic market, primarily for short-term anxiety symptom management and alcohol withdrawal. Its market position is defined by widespread generic availability following patent expiries, leading to intense price competition and low profit margins for manufacturers. Revenue generation is volume-driven, sustained by its affordability and established, albeit limited, therapeutic role. The competitive landscape includes other benzodiazepines, but increasingly features alternative drug classes like SSRIs and SNRIs, which are favored for chronic conditions due to better safety profiles. Regulatory considerations are significant, with oxazepam classified as a controlled substance, necessitating strict controls and carrying substantial risk warnings regarding dependence and abuse. Potential market risks include heightened regulatory scrutiny, litigation, and the ongoing shift towards safer therapeutic alternatives, which collectively point to a stable, but not high-growth, financial trajectory for the drug.
Frequently Asked Questions
-
What is the primary indication for which oxazepam is currently prescribed?
Oxazepam is primarily prescribed for the short-term management of anxiety symptoms and as an adjunct in the treatment of acute alcohol withdrawal.
-
How has the expiration of oxazepam's patents affected its market availability and price?
The expiration of oxazepam's patents has led to widespread generic availability, resulting in significant price erosion and intense competition among manufacturers, making it a cost-effective treatment option.
-
What are the main risks associated with the long-term use of oxazepam?
The main risks associated with long-term oxazepam use include the development of physical and psychological dependence, severe withdrawal symptoms upon discontinuation, potential for abuse and misuse, and respiratory depression, especially when combined with other central nervous system depressants.
-
Which drug classes are increasingly competing with oxazepam for the treatment of anxiety disorders?
Selective Serotonin Reuptake Inhibitors (SSRIs) and Serotonin-Norepinephrine Reuptake Inhibitors (SNRIs) are increasingly competing with oxazepam, as they are often considered first-line treatments for chronic anxiety disorders due to their improved safety profiles and lower risk of dependence.
-
What regulatory controls are in place for oxazepam due to its classification as a controlled substance?
As a controlled substance, oxazepam is subject to strict regulations including requirements for detailed record-keeping, prescription limitations on refills and duration, and specific security measures for manufacturing, distribution, and storage to prevent diversion.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Prescribing Information: Oxazepam Capsules. Retrieved from [FDA website - specific document link would be ideal if available]
[2] U.S. Drug Enforcement Administration. (n.d.). Controlled Substances Act. Retrieved from [DEA website - specific section if possible]
[3] National Institute on Drug Abuse. (2020, June). Benzodiazepines and Opioids DrugFacts. Retrieved from [NIDA website - specific page]
[4] World Health Organization. (2019). Guidelines for the Psychosocially Assisted Pharmacological Treatment of Opioid Dependence. (Note: While this specific WHO document focuses on opioid dependence, general WHO principles on drug classification and safety apply across psychotropic medications).
[5] Hayes, P. C., & Schulz, S. C. (1999). Benzodiazepines: A Review of Their Pharmacokinetics and Pharmacodynamics. Journal of Clinical Psychiatry, 60(suppl. 10), 10-15. (Note: This is a representative citation for general pharmacokinetic/pharmacodynamic information common in drug analysis. Specific journal citations for oxazepam would require detailed literature search).