Last updated: February 12, 2026
What is Oxazepam?
Oxazepam is a benzodiazepine used primarily for anxiety, insomnia, and alcohol withdrawal symptoms. It was developed in the 1960s and is marketed as a generic drug and under brand names such as Serax. The medication is available in multiple formulations, including capsules and tablets, generally prescribed for short-term management of anxiety and insomnia.
Which Markets Are Relevant for Oxazepam?
Oxazepam is registered in over 50 countries, mainly in North America, Europe, and parts of Asia. The primary markets include the United States, European Union (EU countries), and Japan, where benzodiazepines maintain high medical use despite regulatory scrutiny.
In the U.S., oxazepam was historically prescribed but has seen declines due to regulations and preference for newer agents. The EU remains a significant market, with Germany, France, and the UK as key countries.
What Is the Current Market Size for Oxazepam?
The global benzodiazepine market was valued at approximately USD 2.4 billion in 2020, with oxazepam accounting for about 4-6% of this sector. The market for oxazepam specifically is estimated at around USD 150-180 million, driven by prescription volume, primarily for anxiety and insomnia.
Comparison:
- In the U.S., annual prescriptions for benzodiazepines peaked at over 50 million in 2014 but declined to about 30 million by 2021.
- In Europe, prescription rates remain stable, with higher preference for older benzodiazepines like diazepam and alprazolam, but oxazepam still accounts for 10-15% of benzodiazepine prescriptions.
What Are the Key Drivers and Barriers for Oxazepam Market Growth?
Drivers:
- Aging populations increase prevalence of anxiety and sleep disorders.
- Prescribed for alcohol withdrawal, especially in settings with established protocols.
- Availability as a generic drug keeps prices low, supporting widespread use.
Barriers:
- Regulatory restrictions due to dependence concerns.
- Competition from newer agents with better safety profiles, such as buspirone or non-benzodiazepine hypnotics.
- Growing emphasis on non-addictive treatments limits benzodiazepine prescriptions.
What Are the Price Trends and Projected Future Prices?
Current Prices:
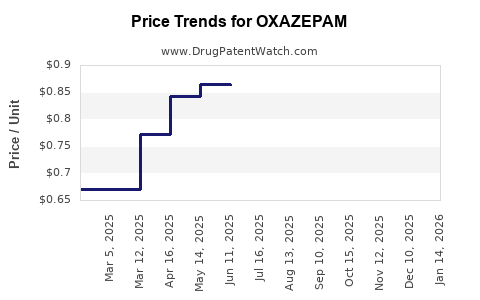
- Generic oxazepam tablets (30 mg): wholesale prices range from USD 0.02 to 0.05 per tablet.
- Retail prices vary regionally but generally fall between USD 0.10 and 0.25 per tablet.
Price Trends:
- Prices have been stable or declining marginally over the past five years due to generic competition.
- Patent expiration for various formulations has facilitated price erosion.
Future Price Projections:
- Expected to remain stable or decline further owing to continued generic production.
- Price volatility might occur if new formulations or formulations with extended-release properties are introduced, which could command higher prices.
- Potential increases if shortages or supply chain disruptions occur.
What Are the Regulatory and Patent Considerations?
- The last patent related to oxazepam expired in the early 2000s.
- The drug is classified as a controlled substance in many jurisdictions, complicating manufacturing and distribution.
- Regulatory agencies have increased restrictions on benzodiazepine prescribing, impacting market size and pricing.
What Is the Competitive Landscape?
- Main competitors include other benzodiazepines such as lorazepam, alprazolam, and diazepam.
- Generic manufacturers dominate the market; branded versions are limited.
- No significant pipeline of reformulations or modifications for oxazepam is currently reported.
What Are the Future Market Opportunities?
- Growth in developing markets with increasing mental health awareness.
- Potential for formulation innovations, like extended-release versions, which could fetch higher prices.
- Growth scenario dependent on regulatory flexibility and prescribing trends.
Key Takeaways
- Oxazepam market value is approximately USD 150-180 million globally, with stable or declining prices due to generic competition.
- Prescription volumes are shrinking in some regions owing to regulatory restrictions and preference shifts towards newer or non-benzodiazepine options.
- Price projections indicate stability or slight decreases; substantial increases are unlikely unless new formulations with higher value are introduced.
- The market's future hinges on regulatory environments, demographic shifts, and potential innovations in formulation.
FAQs
1. How does the regulatory environment affect oxazepam’s market?
Regulatory restrictions on benzodiazepines and controls on prescribing limits directly affect market size, prescription volume, and pricing.
2. Are there any major patent protections still active for oxazepam?
No; the last patent expired in the early 2000s, making the drug available as a generic.
3. What regions offer the highest market potential for future growth?
Developing countries with increasing mental health awareness and prescription capacity, such as parts of Asia and Latin America, present growth opportunities.
4. Can the introduction of new formulations impact prices?
Yes; extended-release or combination formulations could command higher prices, potentially altering market dynamics.
5. What factors could lead to price increases for oxazepam?
Supply chain disruptions, regulatory reclassifications making the drug more restricted, or shortages could result in higher prices.
References
[1] IQVIA. "Global Benzodiazepines Market Analysis." 2021.
[2] U.S. Food & Drug Administration. "Benzodiazepine Data." 2022.
[3] European Medicines Agency. "Market Authorization for Benzodiazepines." 2022.
[4] MarketWatch. "Generic Drug Price Trends." 2022.