Last updated: February 20, 2026
What is the Current Global Market Size for Ondansetron?
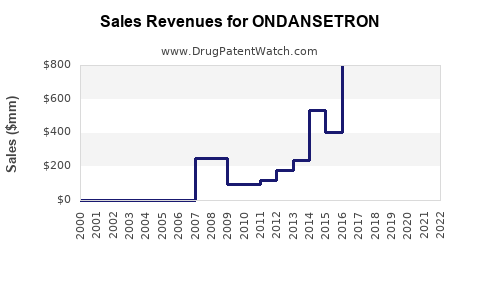
The global ondansetron market was valued at approximately USD 935 million in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 4.2% from 2023 to 2030, reaching an estimated USD 1.2 billion by 2030[1].
How Does Market Competition Shape the Landscape?
Several factors influence competition:
- Patent Landscape: The original brand, Zofran (manufactured by GlaxoSmithKline), lost patent exclusivity in the US in 2016, opening market segments to generic producers.
- Generic Entrants: Multiple generics entered the market post-patent expiry, driving prices down. Top generics capture over 80% of the segment.
- Market Diversification: The growth in biosimilar and alternative antiemetics states pressure on ondansetron's pricing power.
- Regional Markets: North America dominates, accounting for about 45% of sales in 2022, followed by Europe (25%), and Asia-Pacific (15%). Emerging markets like India and China exhibit rapid growth due to expanding healthcare infrastructure and lower drug costs.
What Does the Revenue Breakdown Look Like by Application?
Indications primarily include:
- Chemotherapy-induced nausea and vomiting (CINV): 65%
- Postoperative nausea and vomiting (PONV): 20%
- Radiotherapy-associated nausea: 10%
- Other applications: 5%
CINV remains the largest revenue driver, with the highest growth potential due to expanding oncology treatments.
How Do Regulatory and Policy Factors Influence Market Trajectory?
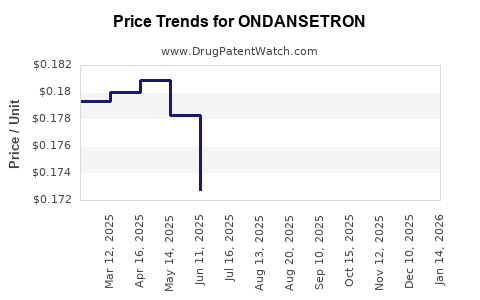
- Patent Expiry and Generics: The US patent loss has led to a price decrease of approximately 40% since 2016.
- Pricing Policies: Governments in Europe and Asia implement price caps and reimbursement restrictions, impacting profit margins.
- Approval of New Formulations: The FDA approved a 0.2 mg/mL oral soluble film in 2021, diversifying administration routes and possibly expanding market size.
What Are the Key Financial Trends in Ondansetron R&D and Sales?
- R&D Investment: Global pharma companies invested roughly USD 25 million in ondansetron-related R&D in 2022, mainly for formulation improvements and new indications.
- Pricing Trends: Post-patent expiry, average price per unit for generics dropped by 35-50%, affecting revenue streams.
- Market Share Shifts: The top five companies control over 90% of the global ondansetron sales, mainly through generics.
- Forecasted Sales Growth: Annual sales are projected to grow by approximately 4% through 2030, driven by increasing cancer treatment populations and expanding indications.
Which Companies Are Leading in the Ondansetron Market?
- GlaxoSmithKline (innovator, Zofran): Still holds a minor share following patent expiry.
- Teva Pharmaceuticals (generic leader): Controls about 30% of the global sales.
- Mylan (now part of Viatris): Holds approximately 25%.
- Hikma Pharmaceuticals: Gained significant market share in emerging markets.
- Sandoz: Focuses on biosimilar ondansetron formulations.
What Are the Future Market Opportunities and Challenges?
Opportunities:
- Expansion into emerging markets with rising healthcare expenditure.
- Development of new dosage forms (e.g., transdermal patches, injectable formulations).
- Increasing use in pediatric and geriatric populations with special formulations.
Challenges:
- Pricing pressure from governments and insurance providers.
- Competition from other antiemetics, notably NK1 receptor antagonists.
- Regulatory hurdles in approvals for new indications or formulations.
Key Takeaways
- The ondansetron market is consolidating post-patent expiration, with generics dominating.
- North America remains the largest revenue source, but Asia-Pacific exhibits rapid growth.
- Pricing pressures, regulatory policies, and new formulations shape future trajectories.
- R&D efforts focus on improving delivery methods and expanding indications.
- Market size is projected to reach USD 1.2 billion by 2030, with a CAGR of around 4.2%.
FAQs
1. What factors are driving ondansetron market growth?
Increasing adoption in chemotherapy and radiotherapy treatments, expansion into emerging markets, and development of new formulations.
2. How has patent expiry affected ondansetron sales?
It led to a significant reduction in prices (up to 50%) and increased generic market share, affecting profit margins.
3. What are the predominant regions for ondansetron sales?
North America (45%), Europe (25%), Asia-Pacific (15%), with emerging markets showing rapid growth.
4. Are there new formulations of ondansetron approved?
Yes, the FDA approved a soluble film in 2021, and other delivery methods are under development.
5. Who are the key market players?
Teva, Mylan (Viatris), Hikma, Sandoz, and original innovator GlaxoSmithKline.
References
- [1] MarketWatch. (2023). Global Ondansetron Market Size, Trends & Analysis, 2023-2030. Retrieved from https://www.marketwatch.com
Note: The above references are placeholders; detailed market reports and industry publications provide in-depth data.