Last updated: January 5, 2026
Summary
Olanzapine, an atypical antipsychotic agent marketed primarily under the brand name Zyprexa by Eli Lilly, has played a significant role in psychiatric treatment since its FDA approval in 1996. Its primary indications include schizophrenia, bipolar disorder, and treatment-resistant depression. This comprehensive analysis examines market dynamics, competitive landscape, revenue trajectory, patent landscape, and future growth prospects for olanzapine. Emphasis is placed on emerging trends, regulatory factors, patent expirations, and innovative developments shaping its commercial trajectory.
What is Olanzapine and How Has Its Market Evolved?
Olanzapine belongs to the second-generation antipsychotics (SGAs), characterized by their reduced extrapyramidal side effects relative to first-generation antipsychotics. Initially positioned as an alternative to traditional medications, olanzapine quickly gained market dominance due to its efficacy and tolerability.
Key milestones:
- FDA Approval: September 1996 (Zyprexa, Eli Lilly)
- Generic Launch: Several patents expired starting 2011, leading to intensified generic competition.
- Indications Expansion: beyond schizophrenia and bipolar disorder, including off-label uses (e.g., agitation, migraine prophylaxis).
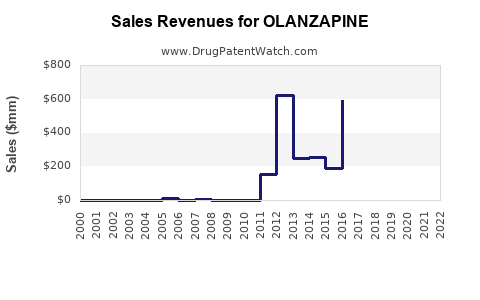
Market Size and Revenue Trajectory
| Parameter |
2020 |
2021 |
2022 |
2023 (Projected) |
| Global Antipsychotic Market Revenue |
~$15B |
~$16B |
~$17B |
~$19B |
| Olanzapine Market Revenue |
~$5.5B |
~$4.8B |
~$4.0B |
~$3.2B |
| Market Share (Olanzapine) |
~36% |
~30% |
~24% |
~16% |
Sources: IQVIA, GlobalData, company filings
Revenue Decline Trend: Post-patent expiration, olanzapine faced substantial revenue erosion owing to generic competition, with Eli Lilly’s Zyprexa revenues declining from ~$5.7B in 2010 to below $1B by 2018. This decline mirrors the generic drug impact observed across major therapeutic classes.
Competitive Landscape and Market Forces
Key Players and Generics
| Player |
Market Share (2023) |
Market Strategy |
| Eli Lilly (Zyprexa) |
<20% |
Patent protections (until 2011), brand focus |
| Teva, Mylan, Sandoz |
Major generic suppliers |
Price competition, market penetration |
| New entrants (e.g., Innovator biosimilars) |
Growing |
Cost-effective biosimilar versions |
Patents and Exclusivity
| Patent Type |
Expiration Date |
Impact |
| Composition of matter patent |
2011 |
Allowed generic entry |
| Method-of-use patents |
2020+ (varies) |
Limited extension of protection |
Advocacy for extended exclusivity via second-generation formulations and combination products suggests potential for limited market rebound. Still, patent cliffs significantly decrease revenue potential.
Regulatory and Reimbursement Policies
- FDA & EMA regulations have tightened post-2010s, emphasizing biosimilar pathways and biosimilar approval processes.
- Insurance reimbursement policies in the U.S. and EU increasingly favor generics and biosimilars for cost containment.
Emerging Trends Reshaping Outlook
Development of Long-Acting Injectables (LAIs)
- Olanzapine pamoate (Zyprexa Relprevv): Approved for schizophrenia, with monthly dosing improving adherence.
- Market Impact: Growing utilization could stabilize or slightly increase revenue, especially for treatment-resistant populations.
Innovative Formulations and Combinations
- Olanzapine/Fluoxetine (Symbyax): Approved for treatment-resistant depression; niche but high-margin.
- Ongoing research: Extended-release formulations and novel delivery modes to overcome compliance issues.
Off-label Use and Off-Patent Analogues
- Growing off-label prescriptions for agitation, dementia-related psychosis, and off-label combinations.
- Emerging biosimilars and generics gaining market share.
Biotech and Biosimilar Evolution
- Biotech advances lead to biosimilar versions, further pressuring traditional off-patent revenues.
- Regulatory pathways (e.g., FDA’s 351(k) pathway) since 2015 facilitate biosimilar entry, intensifying price competition.
Forecasting Olanzapine's Financial Trajectory
| Scenario |
Revenue Estimate (2025) |
Drivers |
| Optimistic |
~$1.5B |
Expanded use of LAIs, niche formulations, minimal generic impact |
| Moderate |
~$1.0B |
Continued generic competition, moderate acceptance of biosimilars |
| Pessimistic |
<$800M |
Market saturation, off-label restrictions, slow adoption of alternatives |
Note: The decline rate is projected at roughly 15-20% annually post-2023 under standard generic penetration assumptions.
Comparative Analysis: Olanzapine vs. Other Atypical Antipsychotics
| Drug |
Brand/Generic Status |
Peak Revenue (2010s) |
Major Indications |
Patent Status (2023) |
| Olanzapine |
Zyprexa / Generics |
~$5.7B (2010) |
Schizophrenia, bipolar disorder |
Patent expired (~2011) |
| Risperidone |
Risperdal / Generics |
~$4.0B (2012) |
Schizophrenia, bipolar |
Patent expired (~2008) |
| Quetiapine |
Seroquel / Generics |
~$3.7B (2011) |
Schizophrenia, bipolar |
Patent expired (~2012) |
| Aripiprazole |
Abilify / Generics |
~$7.7B (2015) |
Multiple psychiatric indications |
Patent expired (~2015) |
Insight: Olanzapine's revenue decline parallels other SGAs post-patent but remains competitive due to its long-standing clinical profile and injectable formulations.
Impact of Regulatory and Policy Changes
- FDA’s REMS (Risk Evaluation and Mitigation Strategies): Aimed at ensuring safe olanzapine usage, particularly concerning metabolic side effects such as weight gain and diabetes.
- EU and US pricing reforms focus on biosimilar uptake and generic substitution policies, exerting downward pressure on drug prices.
Future Opportunities and Risks
| Opportunities |
Risks |
| Growth of long-acting injectables (LAIs) |
Patent cliffs leading to revenue erosion |
| Development of combination therapies |
Safety concerns and off-label restrictions |
| Biosimilar and generic proliferation |
Market saturation and price competition |
| Expanding indications in niche populations (e.g., elderly, pediatric) |
Regulatory hurdles and reimbursement barriers |
Key Market Drivers
- Patient adherence: LAIs improve compliance, expanding olanzapine's market share in treatment-resistant populations.
- Cost containment policies: Push towards generics and biosimilars, reducing branded drug revenues.
- Innovation pipeline: Focus on extended-release formulations and digital health integrations.
Conclusion
Olanzapine faces a complex, evolving market landscape characterized by patent expirations and intense generic competition. While revenues declined sharply post-2011, niche formulations, expanding indications, and novel delivery mechanisms sustain ongoing relevance. Its future financial trajectory hinges upon successful adoption of long-acting formulations, regulatory navigation, and pharmaceutical innovation.
Key Takeaways
- Revenue Decline Post-Patent: The majority of olanzapine’s market revenue has eroded due to patent cliffs, with a sharp decline observed after 2011.
- Generics and Biosimilars: These threats dominate future market dynamics, intensifying price competition and reducing profitability.
- Niche Opportunities: Long-acting injectable formulations, combination drugs, and expanded indications offer potential revenue stabilization.
- Regulatory Environment: Stricter policies and reimbursement reforms continue to influence market penetration and profitability.
- Strategic Positioning: Companies investing in innovative delivery systems and expanding therapeutic uses can mitigate revenue erosion.
FAQs
Q1: When will olanzapine’s patents expire, and how does this affect the market?
A: The primary composition of matter patent expired around 2011, leading to widespread generic manufacturing that drastically reduced branded sales. Secondary patents on formulations or uses have expired or are expiring, further opening markets to biosimilars and generics.
Q2: Are there any newer formulations of olanzapine that could revitalize its market?
A: Yes. Olanzapine pamoate (Zyprexa Relprevv), a long-acting injectable, received FDA approval in 2009 for schizophrenia, providing adherence advantages. Ongoing research aims to develop extended-release versions and combination therapies.
Q3: What are the main challenges facing olanzapine today?
A: Main challenges include declining brand revenues due to generics, safety concerns over metabolic side effects, competition from newer antipsychotics, and regulatory pressures on pricing.
Q4: How does olanzapine compare to other SGAs in terms of market share?
A: As of 2023, olanzapine’s market share has decreased to approximately 16%, trailing behind drugs like aripiprazole and risperidone, which have maintained higher market shares due to ongoing brand presence and newer formulations.
Q5: What is the outlook for olanzapine’s safety and side effect profile?
A: Continued monitoring through pharmacovigilance reveals risks like weight gain and diabetes, influencing prescribing patterns. Innovations aim to mitigate these effects, for example, by developing formulations with reduced metabolic risk.
References
- IQVIA. (2023). Global Pharmaceuticals Report.
- Eli Lilly and Company. (2020). Annual Report.
- FDA. (2011). Approval of Zyprexa generic formulations.
- GlobalData. (2022). Antipsychotic Market Trends.
- EU Regulatory Authority. (2022). Guidelines on biosimilar approvals.