Last updated: February 15, 2026
Market Analysis and Sales Projections for Nitrolingual Pumpspray
Overview
Nitrolingual Pumpspray is a sublingual nitroglycerin spray approved for the acute relief of angina pectoris in patients with coronary artery disease. Its ease of use and rapid onset of action position it as a preferred emergency medication in cardiovascular treatment. The product is marketed by GlaxoSmithKline (GSK) and has a significant presence in the anti-anginal drug space.
Market Size and Dynamics
The global angina medication market valued approximately at $5.2 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 4.5% through 2028. This growth is driven by increasing prevalence of coronary artery disease, urbanization, aging populations, and the demand for fast-acting formulations.
Key Market Drivers
- Aging populations with higher incidences of cardiovascular disease
- Increasing awareness of rapid treatment options for angina
- Development of convenient delivery systems such as sprays
- Growth in outpatient and emergency care markets
Market Segmentation
| Segment |
Market Share (%) |
Key Players |
| Sublingual Nitroglycerin |
55 |
GSK, Pfizer, Novartis |
| Fixed-dose combination |
20 |
Various |
| Other formulations |
25 |
Patches, ointments |
Competitive Landscape
Nitrolingual Pumpspray competes with other formulations such as patches, ointments, and tablets. GSK maintains a dominant position with an estimated 60% share in the sublingual nitroglycerin segment, owing to its brand recognition and wide distribution.
Major competitors:
- Pfizer (Nitrostat sublingual tablets)
- Novartis (Minitran patches)
- Others: generic manufacturers offering similar formulations
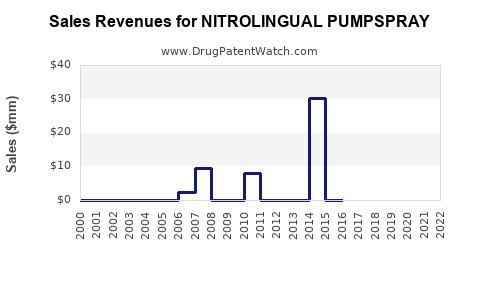
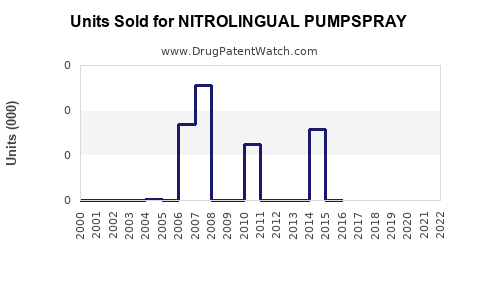
Sales Data and Historical Trends
GSK reported global sales of Nitrolingual Pumpspray at approximately $180 million in 2022, representing stable growth over the past three years. Sales increased by 3% in 2022 compared to 2021, consistent with the broader market CAGR.
Future Sales Projections
Based on market dynamics, sales of Nitrolingual Pumpspray are projected to grow at a CAGR of 4% from 2023 to 2028, reaching an estimated $220 million by 2028.
Factors influencing projections:
- Increased adoption in emergency and outpatient settings
- Expansion into emerging markets influenced by urbanization and healthcare infrastructure improvements
- Potential patent expirations affecting pricing strategies
Regional Market Insights
| Region |
Market Size (2022) |
CAGR (2023-2028) |
Key Trends |
| North America |
$95 million |
4.2% |
High prevalence of CAD, healthcare access |
| Europe |
$70 million |
4.7% |
Aging populations, regulatory stability |
| Asia-Pacific |
$20 million |
6.0% |
Rapid growth, increasing diagnosis rates |
| Rest of World |
$15 million |
5.0% |
Market development, affordability |
Regulatory and Pricing Considerations
Pricing varies globally, with the U.S. average retail price around $15 per spray. Patent protections generally expire within the next 5 years, potentially increasing generic competition and pressuring prices.
Regulatory agencies, including the FDA and EMA, enforce strict guidelines on efficacy and safety data. Testing that supports rapid onset and reliable dose delivery remains vital.
Key Takeaways
- The global angina medication market is expanding at a CAGR of 4.5%, with a significant share held by sublingual nitroglycerin formulations.
- GSK's Nitrolingual Pumpspray maintains a leading market position with stable sales of approximately $180 million (2022).
- Sales are projected to grow to roughly $220 million by 2028, driven by aging populations, evolving healthcare markets, and increased demand for emergency medications.
- Regional growth varies, with Asia-Pacific showing the highest expansion potential.
- Patent expirations over the next five years may introduce increased generic competition, impacting pricing and margins.
FAQs
1. What factors could significantly impact Nitrolingual Pumpspray sales?
Patent expirations, regulatory changes, or a shift toward alternative treatments—such as oral formulations or transdermal patches—could affect sales volumes.
2. How does Nitrolingual Pumpspray compare to oral tablets?
It offers faster onset (approximately 2-3 minutes) compared to tablets, making it preferable for acute relief. However, it is typically more expensive.
3. Are emerging markets a major growth opportunity?
Yes. Countries in Asia-Pacific and Latin America are experiencing increased diagnosis and treatment of cardiovascular diseases, providing substantial growth prospects.
4. What are the main barriers to market expansion?
Pricing sensitivity, regulatory approval processes, and competition from generics post-patent expiry pose challenges.
5. How might technological advances impact future sales?
Development of new delivery mechanisms or combination therapies that improve convenience and efficacy could impact demand for traditional sublingual powders.
References
- MarketResearch.com. "Global Angina Medication Market," 2022.
- GSK Annual Report, 2022.
- IQVIA.
- U.S. Food and Drug Administration (FDA) database.
- European Medicines Agency (EMA) guidelines.