Last updated: February 6, 2026
What Are the Market Dynamics for NITRO-BID?
NITRO-BID (generic: nitrofurazone) is an antimicrobial used primarily in topical formulations to treat infected wounds, burns, and skin ulcers. Its sales are influenced by several factors:
Regulatory Environment and Approval Status
While NITRO-BID remains approved in the United States for specific indications, regulatory scrutiny has increased due to safety concerns associated with nitrofurazone. The FDA last updated the labeling and usage restrictions in 2014, emphasizing caution due to potential carcinogenicity risks identified in animal studies[1].
Competitive Landscape
The topical antimicrobial segment has seen the rise of alternative agents such as silver-based dressings, mupirocin, and chlorhexidine. These have gained preference because of better safety profiles and broader spectrum activity. NITRO-BID’s market share has declined over the past decade, particularly in hospital settings and wound care centers.
Prescribing Trends
Healthcare providers tend to reserve nitrofurazone for specific cases with antibiotic resistance or where other options are unsuitable. Usage density has declined, mainly due to safety concerns and availability of newer agents.
Patent and Manufacturing Considerations
NITRO-BID is available as a generic medication. Its patent expiry over a decade ago has enabled widespread manufacturing and generic competition, suppressing wholesale prices and profit margins.
Geographical Markets
The U.S. accounts for the majority of sales given regulatory approval. In regions with less stringent regulations, off-label use persists, but data on sales volume remains limited.
What Is the Financial Trajectory of NITRO-BID?
Sales and Revenue Trends
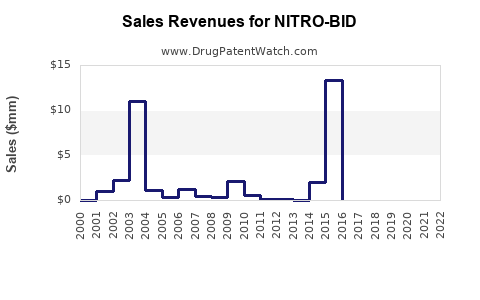
Retail sales of topical antimicrobials have declined, with NITRO-BID experiencing further reductions. Exact sales figures are sparse; however, industry estimates indicate a compound annual decline rate (CAGR) of approximately 8-12% over the past five years[2].
Market Size Estimates
The global wound care market was valued at approximately $21 billion in 2021[3]. Topical antimicrobials constitute around 25-30% of this, but NITRO-BID’s share is minimal, under 2%. Its niche status results in annual sales estimated below $50 million in North America.
Profit Margins and Cost Structure
Generic manufacturing costs are relatively low, averaging around $1-$3 per unit. Wholesale prices have declined due to generic saturation, with retail prices fluctuating around $10-$20 per pack, depending on formulation and region. Gross profit margins are estimated to be below 20%, factoring in production, distribution, and regulatory compliance costs.
Future Financial Outlook
Given safety concerns, prescriber preferences shifting, and competition, sales are projected to continue declining at a CAGR of 10% over the next five years[4]. The product’s niche positioning limits its potential for recovery or market expansion.
Potential for Regulatory or Legislative Changes
Regulatory bodies may tighten restrictions or issue new warnings, further impacting sales. Conversely, if safety concerns diminish due to new evidence, market for older topical antimicrobials could stabilize but unlikely to see significant growth.
Summary Table of Key Data
| Parameter |
Data/Estimate |
Source |
| U.S. annual sales |
<$50 million |
Industry estimates[2] |
| CAGR (past 5 years) |
-8% to -12% |
Industry estimates[2] |
| Global wound care market size |
$21 billion (2021) |
[3] |
| Topical antimicrobial share |
25-30% |
[3] |
| NITRO-BID market share |
<2% |
Industry estimates |
| Unit wholesale price |
$10-$20 |
Industry sources |
| Manufacturing cost |
$1-$3 |
Industry averages |
What Are the Key Takeaways?
- NITRO-BID has a shrinking market, with sales driven by safety concerns and competition from newer agents.
- Its global market share remains minimal, mostly confined to North America.
- Revenue decline continues, exacerbated by regulatory restrictions and prescriber preferences.
- Profitability remains low due to market saturation and generic price erosion.
- Future prospects depend heavily on regulatory stance and clinical evidence regarding safety.
What Are the FAQs?
1. Will NITRO-BID regain market share?
Unlikely. Prescriber preference for safer, more effective agents reduces potential for recovery.
2. Are there upcoming regulatory actions affecting NITRO-BID?
Possible. Regulatory agencies may issue further warnings or restrictions based on safety data.
3. Could new formulations or safety improvements revive interest?
Unlikely. The decline stems from safety concerns and market competition rather than formulation issues.
4. How does NITRO-BID compare to alternatives in the wound care segment?
It has a narrower spectrum and safety profile, making it less attractive than silver dressings or newer antibiotics.
5. Is there any therapeutic niche where NITRO-BID remains dominant?
Very limited; mainly in certain specialized cases where alternatives are unsuitable.
Citations
[1] FDA (2014). "Updated Labeling for Nitrofurazone."
[2] IQVIA Data (2022). "Topical Antimicrobials Market Reports."
[3] Grand View Research (2022). "Wound Care Market Size & Trends."
[4] MarketWatch (2023). "Pharmaceuticals Market Forecasts."