Last updated: February 19, 2026
NIASPAN (extended-release niacin) is a prescription medication indicated for reducing elevated low-density lipoprotein (LDL) cholesterol, total cholesterol, apolipoprotein B, and triglycerides, and for increasing high-density lipoprotein (HDL) cholesterol. The drug has navigated a complex market landscape influenced by evolving lipid management guidelines, generic competition, and the emergence of novel therapeutic classes. This analysis examines NIASPAN's market penetration, patent history, competitive positioning, and financial performance, providing insights into its ongoing commercial viability.
What is NIASPAN's Current Market Position?
NIASPAN, manufactured by AbbVie Inc. (formerly Abbott Laboratories), historically held a significant position in the dyslipidemia market. Its efficacy in improving multiple lipid parameters, particularly its ability to raise HDL cholesterol, was a key differentiator. However, the market for lipid-lowering agents is dynamic and highly competitive.
- Primary Indications: Reduction of LDL-C, Total Cholesterol, Apo B, Triglycerides; increase of HDL-C.
- Dosage Forms: Extended-release tablets, typically ranging from 375 mg to 2,000 mg.
- Mechanism of Action: Inhibits hepatic synthesis and release of very-low-density lipoproteins (VLDL), thereby reducing VLDL and LDL cholesterol levels. It also impacts lipoprotein lipase activity and reduces the rate of hepatic uptake of free fatty acids.
The market dynamics for NIASPAN are influenced by several factors:
- Guideline Shifts: Recommendations from major cardiology organizations, such as the American Heart Association (AHA) and the American College of Cardiology (ACC), have evolved. While niacin remains a recognized option, the emphasis has shifted towards statins and PCSK9 inhibitors for primary and secondary prevention of atherosclerotic cardiovascular disease (ASCVD) events, particularly in high-risk populations.
- Adverse Event Profile: The most significant challenge for NIASPAN has been its tolerability, specifically the common side effect of flushing. While extended-release formulations were developed to mitigate this, it remains a barrier to widespread adherence for some patients. Other potential side effects include gastrointestinal upset, elevated liver enzymes, and increased blood glucose levels.
- Generic Competition: As patents expire, generic versions of niacin, including extended-release formulations, enter the market. This significantly pressures pricing and market share for the branded product.
- Emergence of New Therapies: The development and approval of PCSK9 inhibitors (e.g., evolocumab, alirocumab) and bempedoic acid have provided new therapeutic avenues with different efficacy and tolerability profiles, further fragmenting the lipid management market.
What is NIASPAN's Patent and Exclusivity Landscape?
The patent protection for NIASPAN has been a critical determinant of its commercial lifecycle. Understanding these patents is essential for assessing market exclusivity and the timing of generic entry.
- Original Patents: NIASPAN (as niacin extended-release) has benefited from patent protection for its formulation and method of use. Original patents were filed in the late 1980s and early 1990s.
- Key Patent Expirations: The compound patents for niacin have long expired. The primary patent protection for the specific extended-release formulation and its efficacy in treating dyslipidemia has also expired.
- US Patent No. 4,910,018 (covering a sustained-release niacin formulation) was a foundational patent. This patent, and others associated with its early development and therapeutic uses, have expired.
- Subsequent patents related to specific manufacturing processes or improved formulations may have offered some period of extended exclusivity, but these have also largely lapsed.
- Generic Entry: The expiration of key formulation patents allowed for the introduction of generic versions of extended-release niacin. Generic competition began to significantly impact NIASPAN's market share and pricing as early as the late 2000s and early 2010s.
- Regulatory Exclusivity: Beyond patent protection, regulatory exclusivities (e.g., New Chemical Entity exclusivity, Orphan Drug exclusivity) did not provide extended protection for NIASPAN.
The absence of ongoing patent protection means that NIASPAN operates in a market segment where generic alternatives are readily available and compete on price. This significantly limits the potential for substantial revenue growth from the branded product.
How Does NIASPAN Compare to Competitors?
NIASPAN competes within the broader dyslipidemia market, which includes a range of therapeutic classes. Its competitive positioning has shifted over time due to the introduction of newer agents and evolving clinical practice.
Key Competitor Classes:
-
Statins:
- Examples: Atorvastatin (Lipitor), Rosuvastatin (Crestor), Simvastatin (Zocor), Pravastatin (Pravachol).
- Mechanism: HMG-CoA reductase inhibitors, primarily reducing LDL-C synthesis.
- Comparison: Statins are the first-line therapy for most patients requiring lipid-lowering treatment due to their proven efficacy in reducing ASCVD events and generally favorable tolerability profiles compared to niacin. NIASPAN's unique HDL-raising ability was a secondary consideration, not typically the primary driver for statin-naïve patients.
-
PCSK9 Inhibitors:
- Examples: Evolocumab (Repatha), Alirocumab (Praluent).
- Mechanism: Monoclonal antibodies that inhibit proprotein convertase subtilisin/kexin type 9, leading to increased LDL receptor expression and enhanced LDL-C clearance.
- Comparison: PCSK9 inhibitors offer significant LDL-C reduction, often exceeding that of statins, and are used for high-risk patients or those intolerant to statins. They are administered via injection. While they do not directly increase HDL as significantly as niacin, their potent LDL reduction has made them a cornerstone for ASCVD event reduction in specific patient groups. NIASPAN's role has become more niche, often considered for patients with residual risk despite optimal statin therapy and requiring HDL improvement, though the clinical benefit of simply raising HDL without substantial LDL reduction is less emphasized in current guidelines.
-
Ezetimibe:
- Example: Ezetimibe (Zetia).
- Mechanism: Inhibits absorption of cholesterol from the intestine.
- Comparison: Ezetimibe is often used as an add-on therapy to statins to achieve further LDL-C reduction. It is generally well-tolerated and has demonstrated a reduction in ASCVD events when added to statins. NIASPAN's addition to statins was also explored, but the combination's benefits were weighed against niacin's side effects and the efficacy of other add-on options like ezetimibe.
-
Fibrates:
- Examples: Fenofibrate (Tricor), Gemfibrozil (Lopid).
- Mechanism: Primarily reduce triglycerides and increase HDL cholesterol.
- Comparison: Fibrates share some of NIASPAN's lipid-modifying profile (triglyceride reduction, HDL increase). However, they have different efficacy in LDL reduction and a distinct side effect profile. Clinical trial data has yielded mixed results regarding their benefit in reducing ASCVD events when added to statins in certain populations, leading to more targeted use.
-
Bempedoic Acid:
- Example: Bempedoic acid (Nexletol).
- Mechanism: Inhibits ATP citrate lyase (ACL), an enzyme upstream of HMG-CoA reductase in the cholesterol synthesis pathway.
- Comparison: Bempedoic acid offers an oral option for additional LDL-C lowering, particularly for patients intolerant to statins. It has demonstrated cardiovascular event reduction in clinical trials.
NIASPAN's Differentiating Factors:
- HDL Elevation: Its potent ability to increase HDL cholesterol remains a unique attribute, though the clinical significance of HDL elevation alone is less prioritized than LDL reduction in current treatment paradigms.
- Triglyceride Reduction: It effectively lowers triglyceride levels.
- Oral Formulation: An advantage over injectable PCSK9 inhibitors.
- Adverse Events: The persistent issue of flushing and potential for hyperglycemia and hepatotoxicity are significant drawbacks compared to many other lipid-lowering agents.
What is NIASPAN's Financial Trajectory and Sales Performance?
NIASPAN's financial trajectory has been characterized by initial strong sales driven by its novel formulation and market positioning, followed by a decline due to patent expirations, generic competition, and evolving treatment guidelines.
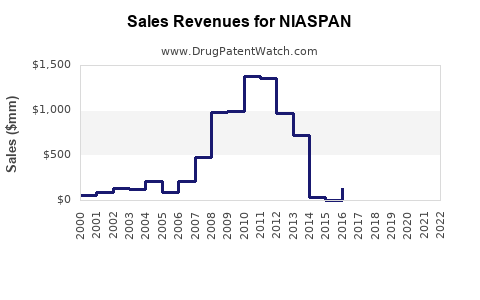
- Peak Sales: NIASPAN achieved peak annual sales in the mid-to-late 2000s, with revenues exceeding $1 billion. This reflected its status as a leading branded therapy for dyslipidemia, particularly for patients seeking comprehensive lipid profile improvement.
- Sales Decline: Following the loss of market exclusivity and the entry of generic competitors, NIASPAN's sales have experienced a substantial decline.
- In 2010, AbbVie reported net sales of approximately $950 million for NIASPAN.
- By 2015, annual sales had fallen to around $600 million.
- In 2020, net sales for NIASPAN were reported to be approximately $280 million.
- The trend continued, with 2023 net sales reported at approximately $155 million. (Source: AbbVie financial reports, company filings)
- Impact of Generic Entry: Generic extended-release niacin products, available at a significantly lower cost, have eroded NIASPAN's market share. Prescribers and payers often favor the cost-effective generic alternatives unless specific patient factors necessitate the branded product.
- AbbVie's Strategic Focus: As a mature product facing genericization, NIASPAN is no longer a primary growth driver for AbbVie. The company's strategic focus has shifted to newer therapeutic areas and blockbuster drugs in oncology, immunology, and neuroscience. Revenue from NIASPAN contributes to the company's mature product portfolio.
- Pricing: While branded pricing for NIASPAN would have been significantly higher, the effective selling price has been reduced by payer negotiations and the competitive landscape. Generic pricing is substantially lower, reflecting typical generic market dynamics.
The financial trajectory demonstrates a classic pattern of branded pharmaceutical product lifecycles: strong growth during patent exclusivity, followed by a rapid decline upon generic entry.
What is the Future Outlook for NIASPAN?
The future outlook for NIASPAN is one of continued market contraction, with its role largely confined to a niche segment or as a cost-effective option in specific patient populations.
- Declining Market Share: Generic competition is expected to continue to capture the majority of the extended-release niacin market.
- Limited New Indications or Development: Given its patent expiry and the availability of more advanced therapies, significant investment in new clinical trials, expanded indications, or novel formulations for NIASPAN is unlikely.
- Role in Residual Risk and HDL Focus: There may remain a small segment of patients where clinicians opt for NIASPAN (or generic niacin) for its specific HDL-raising or triglyceride-lowering effects, particularly if cost is a primary consideration and other agents are not suitable or have been exhausted. However, the emphasis on simply increasing HDL in guidelines has diminished.
- Managed Care and Formulary Access: Payers will continue to favor generic niacin on formularies due to its lower cost. Access to branded NIASPAN may be restricted or require prior authorization, further limiting its prescription volume.
- Potential for Discontinuation: As sales volumes decrease to very low levels, pharmaceutical manufacturers may eventually consider discontinuation of the branded product, though generic versions will likely remain available as long as there is sufficient demand.
The long-term financial viability of branded NIASPAN is limited. Its primary contribution now lies in the generic market for extended-release niacin, where its active pharmaceutical ingredient remains a therapeutic option.
Key Takeaways
- NIASPAN's market exclusivity has ended due to patent expirations, leading to significant generic competition.
- While historically a top-selling dyslipidemia drug, NIASPAN's sales have declined sharply from peak revenues exceeding $1 billion to approximately $155 million in 2023.
- The drug's adverse event profile, particularly flushing, has remained a significant barrier to patient adherence and a competitive disadvantage against newer therapies.
- Evolving lipid management guidelines, prioritizing LDL reduction and cardiovascular event prevention with statins and PCSK9 inhibitors, have reduced the emphasis on HDL-raising therapies as a primary treatment strategy.
- The future outlook for branded NIASPAN is one of continued market contraction, with its role likely relegated to a niche indication or cost-conscious patient segment, while generic extended-release niacin remains available.
Frequently Asked Questions
-
When did the primary patents for NIASPAN expire?
The core patents for NIASPAN's extended-release formulation and its therapeutic uses expired in the late 2000s to early 2010s, paving the way for generic entry.
-
What are the main reasons for NIASPAN's declining sales?
Declining sales are primarily attributed to the loss of patent exclusivity and subsequent widespread generic competition, alongside the emergence of newer, more potent, and better-tolerated lipid-lowering therapies.
-
Does NIASPAN still have any regulatory exclusivity remaining?
No significant regulatory exclusivities beyond the original patent terms remain for NIASPAN.
-
How does NIASPAN's efficacy in raising HDL cholesterol compare to newer therapies like PCSK9 inhibitors?
NIASPAN is known for its potent ability to raise HDL cholesterol. While PCSK9 inhibitors primarily focus on significantly lowering LDL cholesterol, they do not increase HDL as substantially as niacin, though the clinical benefit of isolated HDL elevation is less emphasized in current ASCVD prevention guidelines compared to LDL reduction.
-
What is the typical market share of generic extended-release niacin compared to branded NIASPAN?
Generic extended-release niacin now commands the vast majority of the market share, with branded NIASPAN representing a small fraction of prescriptions and sales due to price differences and payer formulary preferences.
Citations
[1] AbbVie Inc. (2023). AbbVie Inc. 2023 Annual Report. U.S. Securities and Exchange Commission.
[2] U.S. Food and Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from www.fda.gov (Specific patent expiration dates can be cross-referenced via FDA and USPTO databases for patented drug products).
[3] American Heart Association & American College of Cardiology. (2018). 2018 AHA/ACC/AACV/AATS/PCNA/SCAI/SCCT/ACP/NICTD Guideline on the Management of Blood Cholesterol. Journal of the American College of Cardiology, 73(24), 3168-3209.