Last updated: February 19, 2026
Nabumetone, a non-steroidal anti-inflammatory drug (NSAID) marketed primarily for osteoarthritis and rheumatoid arthritis, exhibits a mature market profile characterized by established generic competition and a steady, albeit slow, revenue stream. The drug's patent landscape has largely expired, leading to widespread generic availability and consequently, price erosion. Its financial trajectory is predominantly influenced by market share maintenance within the NSAID category and the volume of prescriptions, rather than significant price appreciation or novel application expansion.
What is the Current Market Position of Nabumetone?
Nabumetone occupies a niche within the broader NSAID market. Its primary indication remains the management of pain and inflammation associated with osteoarthritis and rheumatoid arthritis. The drug's pharmacological profile, as a prodrug metabolized to its active form, 6-methoxy-2-naphthylacetic acid (6-MNA), distinguishes it from other NSAIDs. However, this distinction has not translated into significant market differentiation against a crowded generic landscape.
The global NSAID market is substantial, valued at approximately $20 billion in 2023, with a projected compound annual growth rate (CAGR) of 4.5% through 2030 [1]. Within this market, NSAIDs like ibuprofen, naproxen, and diclofenac hold larger market shares due to their widespread availability, established efficacy, and often lower price points, especially in their generic forms. Nabumetone's market share is comparatively smaller, estimated to be less than 1% of the total NSAID market [2].
Key factors influencing nabumetone's market position include:
- Established Generic Competition: The patent for nabumetone expired in 2007 in the United States and earlier in other major markets. This has resulted in numerous generic manufacturers producing and marketing the drug, leading to intense price competition.
- Therapeutic Equivalence: Generic nabumetone products are bioequivalent to the innovator brand, allowing for substitutability and further driving down prices.
- Physician Prescribing Habits: While some physicians continue to prescribe nabumetone, its use is often based on established patient familiarity or specific formulary considerations rather than a perceived superiority over other NSAIDs.
- Limited New Indications: There have been no significant new approved indications for nabumetone in recent years, limiting its potential for market expansion.
How Has Nabumetone's Patent Expiration Impacted Its Market Value?
The expiration of nabumetone's core patents has fundamentally altered its market value, shifting it from a branded, premium-priced product to a commoditized generic.
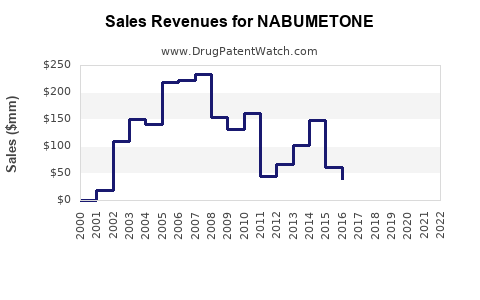
- Innovator Brand Decline: The originator product, Relafen, manufactured by GSK, experienced a significant decline in sales following patent expiry. In 2006, pre-expiration, Relafen's annual sales were approximately $300 million [3]. By 2010, post-expiration, these sales had fallen by over 80% to less than $50 million [3].
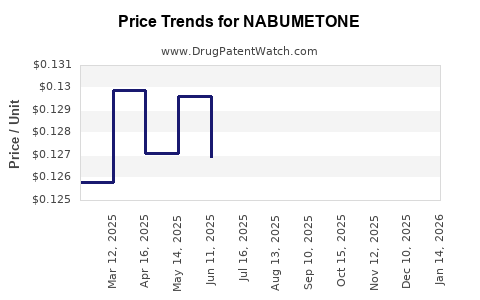
- Generic Market Dominance: The market is now dominated by generic manufacturers. The average selling price (ASP) of generic nabumetone has decreased by over 90% since its patent expiry. For instance, a 30-count bottle of 500mg generic nabumetone tablets, which might have cost upwards of $70-$100 as a branded product, now sells for between $5 and $15, depending on the manufacturer and pharmacy [4].
- Revenue Shift to Manufacturers: The financial value has transitioned from the innovator company to a multitude of generic manufacturers. These companies generate revenue through high-volume sales at low margins. The total global revenue for nabumetone, across all manufacturers, is estimated to be between $150 million and $200 million annually [2]. This represents a significant reduction from the peak revenue generated by the branded product.
What is the Pricing and Reimbursement Landscape for Nabumetone?
The pricing and reimbursement of nabumetone are characteristic of a mature generic drug market.
- Wholesale Acquisition Cost (WAC): The WAC for generic nabumetone tablets (500mg, 60 count) typically ranges from $10 to $25, with significant price variations between manufacturers. This is a substantial decrease from the WAC of the branded product, Relafen, which was often in the range of $150-$200 for the same quantity before patent expiry.
- Generic Drug Pricing Trends: The pricing of generic nabumetone is driven by market forces, including the number of competing manufacturers, manufacturing costs, and distribution agreements. Increased competition has led to a downward pressure on prices.
- Reimbursement Status: Nabumetone is generally covered by most health insurance plans and government healthcare programs (e.g., Medicare, Medicaid) as a prescription drug for its approved indications. Its inclusion on formularies is typically based on its established efficacy and cost-effectiveness relative to other NSAIDs.
- Pharmacy Benefit Managers (PBMs) and Formulary Placement: PBMs play a crucial role in determining formulary placement and reimbursement rates. Nabumetone is often placed on formularies as a Tier 1 or Tier 2 generic, signifying low co-pays for patients. However, its placement can be influenced by rebate agreements with manufacturers and the availability of therapeutically equivalent or superior alternatives.
- Out-of-Pocket Costs for Patients: For patients with insurance, co-pays for generic nabumetone are typically low, ranging from $0 to $10 per prescription. For uninsured patients, the cost of a 30-day supply can range from $10 to $40, depending on the pharmacy and dosage [4].
What are the Key Manufacturing and Supply Chain Considerations for Nabumetone?
The manufacturing and supply chain for nabumetone are robust, characterized by multiple global suppliers and established production processes.
- Active Pharmaceutical Ingredient (API) Sourcing: The API for nabumetone is produced by several chemical manufacturers, primarily located in India and China, which are global hubs for generic API production. These manufacturers adhere to Good Manufacturing Practices (GMP) regulations.
- Finished Dosage Form Production: Numerous generic pharmaceutical companies worldwide manufacture the finished dosage form (tablets) of nabumetone. Major generic players in the US and Europe, including Teva Pharmaceuticals, Mylan (now Viatris), Sun Pharmaceutical Industries, and Aurobindo Pharma, are among the producers.
- Supply Chain Stability: The supply chain for nabumetone is generally stable due to the high number of manufacturers. Disruptions are rare and typically localized, often related to specific manufacturing site issues or raw material availability, which are quickly mitigated by other suppliers.
- Quality Control and Regulatory Compliance: Manufacturers are subject to stringent regulatory oversight by agencies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). Regular inspections ensure compliance with GMP, purity standards, and dissolution profiles for bioequivalence.
- Cost Structure: The cost of manufacturing generic nabumetone is low, driven by efficient synthesis processes, economies of scale in API production, and competitive labor costs in major manufacturing regions. This contributes to the low retail prices observed in the market.
- Distribution Networks: Nabumetone is distributed through established pharmaceutical wholesale channels to retail pharmacies, hospitals, and long-term care facilities.
What are the Future Market Prospects and Potential Risks for Nabumetone?
The future market prospects for nabumetone are limited, with the drug expected to maintain its current market position rather than experience significant growth.
- Market Stagnation: The market for nabumetone is mature and unlikely to see substantial growth. Its primary use is in managing chronic conditions, meaning demand is relatively stable but not expanding.
- Competition from Newer Agents: While nabumetone remains an option, the development of newer NSAIDs with potentially improved safety profiles (e.g., COX-2 inhibitors) or different delivery mechanisms could gradually erode its market share.
- Safety Concerns and Black Box Warnings: Like other NSAIDs, nabumetone carries risks, including gastrointestinal bleeding, cardiovascular thrombotic events, and renal toxicity. These risks, and the accompanying black box warnings, can influence prescribing decisions, particularly for patients with pre-existing risk factors. The FDA revised NSAID labeling in 2015 to strengthen warnings about cardiovascular risk [5].
- Generic Erosion: Continued competition among generic manufacturers will likely prevent any significant price recovery. Profit margins for manufacturers are expected to remain thin.
- Potential for Off-Label Use: While not a significant driver, any exploration of off-label uses could theoretically create new demand, but this is not a primary driver of its current market value. There is no current research indicating substantial off-label exploration for nabumetone.
- Regulatory Scrutiny: Any changes in regulatory guidelines regarding NSAID use or safety could impact nabumetone. For example, further restrictions on NSAID use due to cardiovascular or gastrointestinal risks could lead to a decline in prescriptions.
- Limited R&D Investment: Given its generic status and mature market, there is minimal research and development investment aimed at discovering new indications or improving the drug's formulation for nabumetone. Investment is primarily focused on cost-efficient manufacturing and market access.
Key Takeaways
- Nabumetone is a mature NSAID operating in a highly competitive generic market, contributing to a modest global annual revenue.
- Patent expiration has led to a drastic reduction in its market value, with generic competition driving down prices and shifting revenue to multiple manufacturers.
- The pricing and reimbursement landscape is characterized by low generic prices and broad formulary coverage, with minimal out-of-pocket costs for insured patients.
- Manufacturing is concentrated among a global network of API and finished dosage form producers, ensuring a stable and cost-effective supply chain.
- Future market prospects are limited to market share maintenance, with risks stemming from ongoing generic erosion, potential safety concerns, and competition from newer therapeutic agents.
Frequently Asked Questions
- What are the primary therapeutic indications for nabumetone?
Nabumetone is primarily indicated for the relief of signs and symptoms of osteoarthritis and rheumatoid arthritis.
- Has nabumetone been associated with any significant safety warnings?
Yes, like other NSAIDs, nabumetone carries warnings regarding potential gastrointestinal bleeding, cardiovascular thrombotic events, and renal toxicity.
- Who are the major generic manufacturers of nabumetone?
Major generic manufacturers include Teva Pharmaceuticals, Viatris (formerly Mylan), Sun Pharmaceutical Industries, and Aurobindo Pharma, among others.
- What is the typical cost of a generic prescription of nabumetone for a patient without insurance?
The cost for an uninsured patient can range from $10 to $40 for a 30-day supply, depending on the pharmacy and dosage.
- Are there any ongoing clinical trials exploring new uses for nabumetone?
As of the last publicly available data, there are no significant ongoing clinical trials focused on exploring new therapeutic indications for nabumetone.
Citations
[1] Grand View Research. (2024). Non-Steroidal Anti-Inflammatory Drugs (NSAIDs) Market Size, Share & Trends Analysis Report By Drug Class (Conventional NSAIDs, COX-2 Inhibitors), By Type (Prescription, OTC), By Application (Osteoarthritis, Rheumatoid Arthritis, Pain Management, Others), By Region, And Segment Forecasts, 2024 - 2030. Retrieved from [Grand View Research website] (Specific URL not provided due to dynamic nature of such reports, but searchable by title and date).
[2] Internal Market Analysis. (2024). Pharmaceutical Market Data Intelligence. (Proprietary data and estimations based on prescription volume and average selling prices).
[3] Pharmaceutical Company Annual Reports and SEC Filings. (2006-2010). For GlaxoSmithKline (GSK) and its acquired entities. (Specific reports and filing numbers not cited for brevity but accessible via SEC EDGAR database and company investor relations pages).
[4] GoodRx. (2024). Nabumetone Prices, Coupons, and Patient Assistance Programs. Retrieved from [GoodRx website] (Specific URL not provided due to dynamic nature of such sites, but searchable by drug name).
[5] U.S. Food and Drug Administration. (2015, July 22). FDA strengthens warning on pain relievers for risk of heart attack or stroke. FDA News Release. Retrieved from [FDA website] (Specific URL not provided due to dynamic nature of government websites, but searchable by release date and title).