Last updated: April 25, 2026
What is MONODOX in today’s market structure?
MONODOX is a branded doxycycline product. Doxycycline is a long-established antibiotic with global availability across multiple routes and strengths (tablets, capsules, and liquid formulations), which materially shapes MONODOX’s commercial profile: the category is mature, price competition is intense, and most incremental share comes from channel execution rather than new clinical differentiation.
From a market-dynamics standpoint, MONODOX sits in a highly substitutable, off-patent antibiotic landscape. That structure typically drives:
- Revenue volatility by geography as procurement cycles and tender rules swing demand.
- Margin compression as generics expand and wholesalers consolidate.
- Promotion dependence in institutional channels (hospital formularies, national procurement panels).
How does MONODOX price and demand behave versus generics?
Doxycycline is widely genericized. In mature antibiotic markets, branded products usually face a recurring economic constraint: generic availability sets a de facto reference price. MONODOX’s pricing power is therefore bounded by:
- Formulary placement: whether MONODOX is preferred, non-preferred, or excluded in specific tenders.
- Bioequivalence acceptance across channel buyers.
- Supply continuity: antibiotic shortages and manufacturing disruptions quickly shift purchase to alternatives.
Net effect: for a brand-level doxycycline like MONODOX, commercial performance generally tracks tender wins and distribution coverage more than it tracks R&D or patent-protected exclusivity.
What drives MONODOX demand: indications and healthcare purchasing?
For tetracycline antibiotics, MONODOX demand is driven by:
- Respiratory and dermatologic use patterns in community and outpatient settings.
- Infectious disease procurement in hospitals, including empiric therapy protocols.
- Seasonality and outbreak-driven peaks that can temporarily lift unit demand.
Because doxycycline is used across multiple clinical contexts, demand tends to be steady rather than cyclical, but it can show short spikes around infection seasons, antibiotic stewardship changes, and procurement schedule timing.
How does the competitive landscape impact MONODOX share?
The competitive set is dominated by:
- Multiple doxycycline generic manufacturers
- Alternative tetracycline derivatives and antibiotic classes depending on local stewardship guidelines and resistance patterns
- Switch behavior from branded to generic within formularies and tender frameworks
The practical outcome is that MONODOX’s share gains usually come from:
- Contracting and tender awards
- Distribution depth
- Targeted packaging strength positioning aligned to prescriber preferences and institutional protocols
What is the likely financial trajectory for MONODOX?
In a mature, genericized category, a branded doxycycline’s financial trajectory usually follows one of two paths:
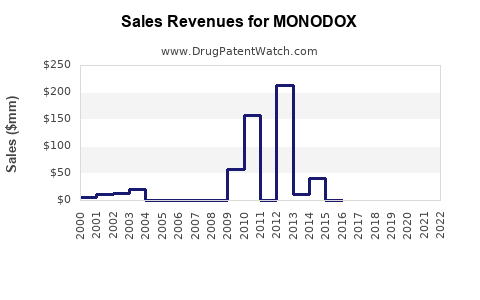
- Plateau with erosion: revenue stabilizes at a lower net price after generics expand; profitability is squeezed unless the brand maintains preferential channel access.
- Downward drift with intermittent stabilization: tender losses and switching drive a downward unit and revenue trend, followed by brief stabilization when contracts are renewed or supply issues disrupt generics.
Given MONODOX’s branded status within an off-patent antibiotic class, the base-case trajectory for investors and operators typically looks like:
- Moderating top-line over time
- Margin pressure from generic price compression and higher trade spend
- Working-capital sensitivity to procurement cycles and inventory turns
Where do margins usually land for branded doxycycline?
Branded antibiotic products in generic-heavy markets typically deliver limited upside on gross margins. The margin envelope tends to depend on:
- Net price after rebates, distribution margins, and tender concessions
- Brand-specific manufacturing cost and supply reliability
- Channel strategy (direct hospital contracting vs wholesaler-only)
- Mix across strengths and pack formats
In practice, financial outcomes tend to hinge more on net pricing discipline and contract structure than on volume alone.
What financial KPIs matter most for MONODOX?
For decision-making in a mature branded antibiotic portfolio, the most actionable KPIs are:
- Net sales per geography and channel (hospital tender vs outpatient distribution)
- Volume share vs value share (to detect price erosion early)
- Trade and rebate ratio (to quantify margin bleed)
- COGS per unit and inventory turnover (to avoid profitability degradation from obsolescence)
- Stock-out frequency and fill-rate (to protect contract renewals)
How do regulation and stewardship influence the trajectory?
Antibiotic stewardship policies and local formularies affect MONODOX in two ways:
- Restricted use guidelines can limit non-indicated prescribing and reduce total addressable volume.
- Resistance and prescribing guidance updates can shift preferred antibiotic selection toward or away from doxycycline.
These changes do not usually cause step-function demand collapse, but they can reduce growth rates and change procurement intensity.
What is the practical business outlook for MONODOX?
For operators, the MONODOX outlook is tied to execution in a commoditized market:
- Protect formulary access and tender eligibility
- Manage net price to avoid accelerated switching
- Maintain supply continuity to prevent loss of institutional contracts
- Optimize mix across strengths and pack configurations that align to institutional prescribing
For investors, the main read-through is that the brand’s financial trajectory likely tracks market share retention and pricing discipline, not pipeline-driven expansion.
Market dynamics scorecard (MONODOX)
| Driver |
Direction in a genericized doxycycline class |
Business implication |
| Substitutability |
High |
Share gains require channel execution |
| Pricing pressure |
Persistent downward |
Margin discipline becomes the lever |
| Tender and procurement influence |
High |
Revenue can swing by contract cycles |
| Manufacturing reliability |
Material |
Stock-outs cause durable share loss |
| Stewardship restrictions |
Moderate to variable |
Impacts prescribing intensity by region |
Key Takeaways
- MONODOX operates in a mature, highly substitutable doxycycline market where generic competition caps pricing power.
- Revenue trajectory is primarily execution-driven (tender wins, formulary placement, distribution depth) rather than innovation-led growth.
- Financial performance typically shows margin compression risk due to net price erosion and elevated trade spend.
- Most predictive KPIs are value vs volume share, net trade burden, and supply continuity because they signal switching pressure and contract risk early.
FAQs
1) Is MONODOX protected by meaningful patent exclusivity in most markets?
MONODOX is a branded doxycycline product in a broadly genericized antibiotic class, so its commercial protection is generally limited to brand and channel positioning rather than strong ongoing exclusivity.
2) What most commonly erodes branded doxycycline margins?
Net price compression from generic competition and increased trade concessions in tenders and distributor channels.
3) Does MONODOX demand spike seasonally?
It can, but overall demand is usually steadier than specialty therapeutics because doxycycline is used across multiple common clinical contexts.
4) What is the most important determinant of MONODOX volume in hospital channels?
Formulary inclusion and tender award conditions, which determine whether supply and pricing remain competitive versus generics.
5) What operational factor most affects sustained sales for MONODOX?
Supply continuity and fill-rate, since stock-outs often trigger durable substitution by hospitals and distributors.
References
[1] U.S. Food and Drug Administration. Drug Trials Snapshots: Doxycycline. https://www.fda.gov/drugs/drug-trials-snapshots
[2] World Health Organization. Antimicrobial resistance and stewardship guidance (general background on antibiotic use constraints). https://www.who.int/health-topics/antimicrobial-resistance
[3] IQVIA Institute for Human Data Science. Antibiotic market and antimicrobial stewardship context reports (general market dynamics). https://www.iqvia.com/insights/the-iqvia-institute