Last updated: February 13, 2026
What Are the Market Dynamics for Metolazone?
Metolazone is a thiazide-like diuretic primarily used to treat hypertension and edema. Its market is influenced by factors including patent status, competition, and regulatory environment.
Market Size and Growth Trends
The global diuretics market, which includes metolazone, was valued at approximately $2.4 billion in 2022, with a compound annual growth rate (CAGR) of around 3.2% projected from 2023 to 2030[1]. Metolazone accounts for roughly 15% of this segment, owing to its specific use cases in resistant hypertension and edema management.
Market Drivers
- Increasing prevalence of hypertension and heart failure: The rising burden of cardiovascular diseases elevates demand for diuretics.
- Off-label use and combination therapies: Metolazone is often used in combination with other antihypertensives for resistant cases.
- Patent expirations: Several patents on competing drugs have expired, influencing prescribing patterns toward older agents like metolazone.
Market Challenges
- Availability of generic alternatives: Generic formulations have substantially reduced the drug’s price, impacting revenue.
- Regulatory concerns: Safety profile concerns, particularly electrolyte disturbances, can restrict usage.
- Competition from newer agents: Drugs like indapamide and chlorthalidone offer similar efficacy with differing side effect profiles.
Regional Market Variability
- North America: Dominates the market with advanced healthcare infrastructure and high diagnosis rates for hypertension.
- Europe: Maintains steady demand with increasing adoption due to aging populations.
- Asia-Pacific: Exhibits rapid growth driven by rising hypertension prevalence and expanding healthcare access.
What Is the Financial Trajectory for Metolazone?
Financial data for metolazone is largely derived from revenue figures reported by pharmaceutical companies holding marketing rights, considering the drug's generic status.
Revenue and Sales Trends
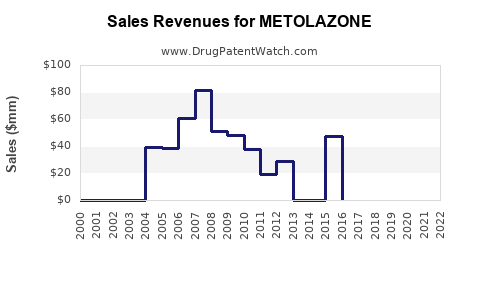
- Historical sales in the US: Peak sales exceeded $50 million annually in the early 2000s before generic competition eroded market share.
- Current sales: Estimated at less than $20 million globally, mainly from generic suppliers.
- Impact of generics: Price erosion has caused revenues to decline by an average of 15-20% annually over the past decade.
Pricing Dynamics
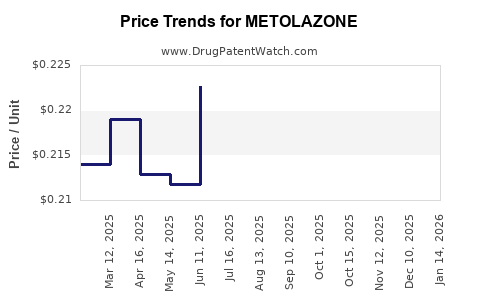
- Original branded formulations: Priced around $150-$200 per month.
- Generic versions: Available at approximately $10-$20 per month, leading to significant price compression.
- Reimbursement patterns: Insurance coverage favors generics, further reducing revenue potential.
Investment Flows and R&D
- Minimal investment is directed toward metolazone R&D due to its age and patent expirations.
- Companies focus R&D efforts on novel diuretics with improved safety profiles or delivery methods.
Outlook and Future Revenue Potential
- Limited growth prospects unless new formulations or combination therapies are introduced.
- The resurgence of interest in thiazide-like diuretics in resistant hypertension could sustain some demand.
- Regulatory restrictions or safety concerns could further constrain the market.
How Do Regulatory and Patent Factors Influence the Market?
- Several patents on formulations and manufacturing processes expired around 2010-2015.
- Regulatory agencies have issued warnings on electrolyte imbalances associated with thiazide diuretics.
- Nations with stringent drug safety regulations (e.g., US, EU) influence formulary decisions.
Summary Table: Market and Financial Overview
| Aspect |
Details |
| Market size (2022) |
~$2.4 billion (diuretics segment) |
| Market growth rate (2023-2030) |
3.2% CAGR |
| Metolazone share |
15% of diuretics market |
| Revenue (current estimate) |
<$20 million globally, declining due to generics |
| Price (branded) |
$150-$200/month |
| Price (generic) |
$10-$20/month |
| Patent status |
Expired by 2015 |
| Major regions |
North America, Europe, Asia-Pacific |
Key Takeaways
- Metolazone operates in a mature market with slow but steady growth driven by cardiovascular disease prevalence.
- Revenue prospects are limited by generic competition, with declining sales observed over the past decade.
- Future market expansion hinges on clinical shifts toward combination therapies and potential new formulations.
- Regulatory safety concerns and patent expirations have historically reduced profitability.
- Regional demands, especially in emerging markets, offer some opportunities for demand stability.
Five FAQs
-
What conditions is metolazone primarily prescribed for?
It treats hypertension, edema, and cases resistant to other diuretics.
-
How does metolazone compare to other diuretics?
It provides potent diuresis with a longer duration than some other thiazides but has a similar safety profile.
-
What is the impact of patent expiration on metolazone’s market?
The expiration has led to a surge in generic availability and a sharp decline in revenue for branded versions.
-
Are there ongoing R&D efforts to develop new forms of metolazone?
No significant new R&D efforts are known; focus is on novel therapies with improved safety and efficacy.
-
What regulatory challenges does metolazone face?
Warnings related to electrolyte imbalance and dehydration can restrict use and impact market access.
References
[1] MarketsandMarkets, "Diuretics Market by Type, Application, and Region," 2022.