Last updated: February 19, 2026
LOTEMAX SM (loteprednol etabonate ophthalmic suspension) is a corticosteroid used for the treatment of ocular inflammation and pain following ocular surgery. Its market performance is driven by factors including the prevalence of ocular conditions, surgical volumes, competitive landscape, and intellectual property status.
What is the current market position of LOTEMAX SM?
LOTEMAX SM is a prescription ophthalmic corticosteroid developed by Bausch + Lomb. It is formulated as a 0.5% ophthalmic suspension and is indicated for the treatment of post-operative inflammation and pain in patients who have undergone ocular surgery. The drug's mechanism of action involves suppressing inflammation by inhibiting the synthesis of prostaglandins, key mediators of inflammatory processes [1].
The market for ophthalmic corticosteroids is mature, with several established products. LOTEMAX SM competes with other topical corticosteroids such as prednisolone acetate, fluorometholone, and dexamethasone, as well as non-steroidal anti-inflammatory drugs (NSAIDs) for post-operative pain management. The primary differentiator for LOTEMAX SM is its novel etho-amino corticosteroid ester prodrug technology, which aims to reduce ocular side effects commonly associated with traditional corticosteroids, such as increased intraocular pressure (IOP) and delayed wound healing [2].
As of the latest available data, LOTEMAX SM holds a significant share within the ophthalmic corticosteroid segment. Its market penetration is influenced by physician prescribing habits, formulary access, and patient adherence. The drug's convenience in terms of dosing frequency compared to some older formulations also contributes to its adoption.
What are the key therapeutic indications and patient populations for LOTEMAX SM?
The primary indication for LOTEMAX SM is the treatment of inflammation and pain following ocular surgery. This includes procedures such as cataract surgery, LASIK, and other refractive surgeries. Post-operative inflammation is a common complication that can affect visual recovery and patient comfort.
The patient population includes individuals undergoing these surgical procedures. These can range from adults seeking vision correction to older adults undergoing cataract surgery. The drug's safety profile, particularly its reduced potential for IOP elevation, makes it an attractive option for a broad patient demographic, including those with pre-existing ocular hypertension or glaucoma, provided they are carefully monitored.
Specific patient scenarios where LOTEMAX SM is considered include:
- Cataract Surgery: Patients experiencing post-operative inflammation and pain, aiming for rapid visual rehabilitation and comfort.
- Refractive Surgery (e.g., LASIK): Management of inflammation and pain to ensure optimal healing and visual outcomes.
- Other Ophthalmic Surgical Procedures: Addressing inflammatory responses in various anterior segment surgeries.
The efficacy and safety data supporting these indications are derived from clinical trials demonstrating its ability to reduce clinical signs of inflammation and alleviate pain in the post-operative period.
What is the intellectual property landscape and patent expiration timeline for LOTEMAX SM?
The intellectual property surrounding LOTEMAX SM is critical to its market exclusivity and financial trajectory. The active pharmaceutical ingredient, loteprednol etabonate, is protected by various patents. Bausch + Lomb has secured patents covering the composition of matter, methods of use, and formulations of loteprednol etabonate [3].
Key patents and their expiration dates are crucial for projecting future market dynamics. While specific patent numbers and their precise expiration dates are often complex and subject to extension due to regulatory delays (e.g., Hatch-Waxman Act), general timelines can be analyzed.
The original patents for loteprednol etabonate began to expire in the early to mid-2010s. However, Bausch + Lomb has strategically filed secondary patents and sought patent term extensions to protect its market position for extended periods. These secondary patents often cover specific formulations, such as the SM (Submicron) particle size formulation, which enhances penetration and efficacy, or new delivery systems.
| Patent Type |
Key Expiration Period (Approximate) |
Impact on Exclusivity |
| Composition of Matter |
Mid-2010s |
Original exclusivity eroded; allowed for generics. |
| Formulation Patents |
Late 2020s - Early 2030s |
Extended market protection for branded LOTEMAX SM. |
| Method of Use Patents |
Varies |
May offer protection for specific therapeutic applications |
The emergence of generic versions of loteprednol etabonate is a significant factor following the expiration of core patents. Generic competition typically leads to substantial price erosion and a decline in market share for the branded product. However, the successful prosecution of formulation patents can create a significant barrier to entry for generic manufacturers, especially if the generic formulation cannot replicate the specific characteristics and performance of the branded product. For LOTEMAX SM, the SM formulation's enhanced properties may offer a degree of continued differentiation even in the presence of generics of the base molecule.
Companies are also exploring new indications or combination therapies involving loteprednol etabonate, which could lead to new patent filings and further extend market exclusivity.
What is the competitive landscape and market share of LOTEMAX SM?
The ophthalmic corticosteroid market is highly competitive. LOTEMAX SM competes with a range of products, including:
- Other Loteprednol Etabonate Products: Different formulations of loteprednol etabonate, such as LOTEMAX GEL and LOTEMAX OINTMENT, which may have their own patent protections and market segments.
- Prednisolone Acetate: A widely used and often lower-cost corticosteroid.
- Dexamethasone: A potent corticosteroid with a longer history of use.
- Fluorometholone: Another corticosteroid option.
- NSAIDs: Drugs like ketorolac tromethamine and nepafenac are used for pain management and can sometimes reduce the need for corticosteroids.
- Combination Products: Products that combine a corticosteroid with an antibiotic.
Bausch + Lomb's strategic positioning of LOTEMAX SM, emphasizing its favorable safety profile regarding IOP elevation and its convenient dosing, aims to capture a significant share of the post-operative inflammation market. The company also benefits from established relationships with ophthalmologists and strong distribution networks.
Market share data for LOTEMAX SM is typically proprietary and reported by market research firms. However, industry analysis indicates that LOTEMAX SM has achieved substantial market penetration. Its share is likely to be strongest in the post-operative setting where its benefits are most pronounced and widely recognized by surgeons.
The introduction of generic loteprednol etabonate products has begun to impact LOTEMAX SM's market share. However, the extent of this impact depends on the ability of generic manufacturers to produce bioequivalent formulations that achieve similar therapeutic outcomes and on the pricing strategies of both branded and generic products.
What are the recent financial performance trends and revenue drivers for LOTEMAX SM?
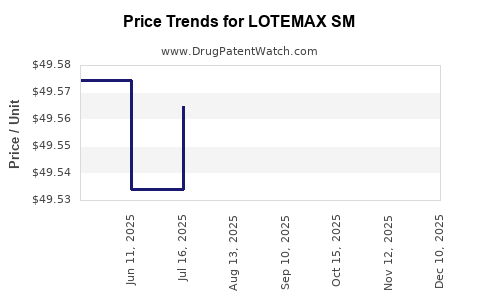
The financial performance of LOTEMAX SM is directly linked to its sales volume and pricing. As a branded pharmaceutical product, its revenue drivers include prescription volume, average selling price (ASP), and market access through insurance formularies.
Revenue Drivers:
- Prescription Volume: Driven by the number of ocular surgeries performed and physician adoption of LOTEMAX SM for post-operative management.
- Average Selling Price (ASP): Influenced by Bausch + Lomb's pricing strategy, competitor pricing, and payer negotiations. Branded products typically command higher prices than generics.
- Market Access: Inclusion on insurance formularies and favorable reimbursement rates are critical for patient access and prescription volume.
- Sales and Marketing Efforts: Bausch + Lomb's investment in detailing, medical education, and promotional activities impacts physician prescribing.
- Launch of New Formulations or Indications: While LOTEMAX SM is established, further development could boost revenue.
Financial Performance Trends:
- Pre-Generic Entry: In the period prior to significant generic competition for loteprednol etabonate, LOTEMAX SM likely experienced steady revenue growth, driven by increasing surgical volumes and market penetration.
- Impact of Generic Entry: The introduction of generic loteprednol etabonate products leads to an expected decline in LOTEMAX SM's revenue due to price erosion and market share shifts to lower-cost alternatives. The speed and magnitude of this decline depend on the number of approved generic competitors and their market penetration.
- Sustained Revenue from Branded Differentiation: Bausch + Lomb aims to mitigate generic impact through its focus on the proprietary SM formulation, emphasizing its differentiated performance characteristics, and potentially through lifecycle management strategies (e.g., combination products).
- Revenue Contribution: LOTEMAX SM is a significant product for Bausch + Lomb's ophthalmic portfolio. Its exact contribution to the company's overall revenue would be detailed in their financial reports.
Analyzing Bausch + Lomb's annual and quarterly financial reports would provide specific revenue figures for LOTEMAX SM or its parent product line, as well as insights into sales trends and geographic performance. However, specific product-level revenue data for LOTEMAX SM may be aggregated within broader financial reporting categories by the company.
What is the projected future market trajectory and potential risks for LOTEMAX SM?
The future market trajectory of LOTEMAX SM will be shaped by several key factors, including the ongoing impact of generic competition, innovation in the ophthalmic space, and evolving clinical practice patterns.
Projected Trajectory:
- Continued Erosion from Generics: The primary challenge to LOTEMAX SM's future revenue will be the increasing availability and adoption of generic loteprednol etabonate. This will lead to sustained price pressure and a reduction in branded market share.
- Market Share Defense through Differentiation: Bausch + Lomb will likely focus on reinforcing the value proposition of the SM formulation, highlighting its clinical benefits and physician preference among those who prioritize its specific attributes. This may allow LOTEMAX SM to retain a premium segment of the market.
- Potential for Lifecycle Management: Bausch + Lomb may explore opportunities for lifecycle management, such as developing new combination products or exploring novel delivery systems for loteprednol etabonate, which could create new revenue streams and extend market exclusivity for certain applications.
- Shifting Surgical Volumes: The overall volume of ocular surgeries, particularly cataract procedures, is projected to grow due to an aging population. This baseline growth provides a potential tailwind for all post-operative treatments, including LOTEMAX SM.
- Competition from Non-Steroidal Agents: Advances in NSAID technology and their expanding role in post-operative pain and inflammation management could limit the growth or even reduce the utilization of corticosteroids, impacting LOTEMAX SM's market share.
Potential Risks:
- Aggressive Generic Pricing: The speed and aggressiveness with which generic manufacturers price their products will significantly influence the rate of LOTEMAX SM's revenue decline.
- Off-Label Use of Generics: Physicians may readily switch to generic loteprednol etabonate due to cost savings, even if the branded product has perceived advantages, particularly if clinical trial data for generics demonstrates equivalent efficacy and safety.
- New Entrants and Innovation: The development of novel anti-inflammatory agents with superior efficacy, safety profiles, or novel mechanisms of action could disrupt the current market for corticosteroids.
- Payer Restrictions: Insurance companies may increasingly favor generic alternatives or implement stricter utilization management policies for branded corticosteroids, limiting access and prescription volume.
- Regulatory Scrutiny: Any adverse events or new safety concerns associated with loteprednol etabonate could lead to regulatory action or impact physician prescribing confidence.
- Patent Litigation: Legal challenges related to patent validity or infringement could impact Bausch + Lomb's ability to defend its intellectual property.
The overall market trajectory for LOTEMAX SM is expected to be one of gradual decline in revenue due to genericization, with the pace and extent of this decline moderated by Bausch + Lomb's efforts to leverage the differentiated aspects of its SM formulation and potential lifecycle management strategies.
Key Takeaways
LOTEMAX SM is a significant product in the ophthalmic corticosteroid market, primarily utilized for post-operative inflammation and pain management. Its market position is defined by its formulation's reduced potential for IOP elevation compared to traditional corticosteroids. The intellectual property landscape, particularly formulation patents, has historically extended market exclusivity, but the expiration of core patents has opened the door for generic competition. This genericization is the primary driver of anticipated future revenue erosion. While the overall volume of ocular surgeries provides a baseline market demand, LOTEMAX SM faces competition from other corticosteroids, NSAIDs, and the increasing prevalence of generic loteprednol etabonate. Future performance will depend on Bausch + Lomb's ability to defend market share through product differentiation and lifecycle management against aggressive generic pricing and potential innovations in the therapeutic landscape.
Frequently Asked Questions
-
When did LOTEMAX SM first receive FDA approval?
LOTEMAX SM (loteprednol etabonate ophthalmic suspension 0.5%) received FDA approval in September 2019. This was subsequent to the approval of the original LOTEMAX (loteprednol etabonate ophthalmic suspension 5%) in 1997. [4]
-
What is the difference between LOTEMAX and LOTEMAX SM?
The primary difference lies in the particle size of the active pharmaceutical ingredient, loteprednol etabonate. LOTEMAX SM features a submicron particle size formulation, designed to improve penetration into ocular tissues and enhance efficacy for treating inflammation and pain. [2]
-
Can LOTEMAX SM be used for conditions other than post-operative inflammation?
While LOTEMAX SM is specifically indicated for post-operative inflammation and pain following ocular surgery, loteprednol etabonate in other formulations (e.g., LOTEMAX GEL) has been approved for broader indications such as allergic conjunctivitis and the treatment of anterior uveitis. [1, 5]
-
How does the cost of LOTEMAX SM compare to generic loteprednol etabonate?
Branded LOTEMAX SM typically has a higher Average Selling Price (ASP) compared to generic loteprednol etabonate products. The exact price difference can vary based on payer contracts, pharmacy dispensing fees, and manufacturer discounts.
-
What are the main side effects associated with LOTEMAX SM?
Common side effects include eye pain, blurred vision, eye irritation, and abnormal taste. More serious but less common side effects can include increased intraocular pressure (IOP), glaucoma, cataracts, and delayed wound healing, similar to other topical corticosteroids. [1]
Citations
[1] Bausch + Lomb. (2019). LOTEMAX SM (loteprednol etabonate ophthalmic suspension) 0.5% Prescribing Information. Retrieved from https://www.bausch.com/ (Specific document link may vary and require account login).
[2] P. R. R. G. K. P. N. G. K. S. N. G. P. H. A. D. (2019). Loteprednol Etabonate Submicron Particle Size Ophthalmic Suspension in the Management of Ocular Inflammation and Pain Post Ocular Surgery. Clinical Ophthalmology, 13, 1747–1753. https://doi.org/10.2147/OPTH.S194798
[3] U.S. Patent and Trademark Office. (n.d.). Patent Search. Retrieved from https://patft.uspto.gov/ (Specific patent numbers and expiration dates require targeted search).
[4] Food and Drug Administration. (n.d.). Drug Approvals and Databases. Retrieved from https://www.fda.gov/ (Specific approval dates can be verified through FDA Orange Book or drug approval databases).
[5] Bausch + Lomb. (n.d.). LOTEMAX GEL Prescribing Information. Retrieved from https://www.bausch.com/ (Specific document link may vary and require account login).