Last updated: February 13, 2026
Market Overview and Indications
LOTEMAX SM (loteprednol etabonate ophthalmic suspension 0.38%) is a corticosteroid used primarily post-operatively in ophthalmology, particularly for preventing inflammation after ocular surgeries such as cataract extraction. It is marketed by Sun Pharma as an alternative to other corticosteroids with a lower propensity for raising intraocular pressure.
The drug addresses several market needs:
- Reduction of intraocular inflammation
- Management of post-surgical ocular pain
- Avoidance of steroid-induced side effects
Market Size and Growth Drivers
The global ophthalmic corticosteroids market is estimated at approximately USD 400 million in 2022, with a compound annual growth rate (CAGR) of 4-5% projected through 2027 (source: MarketsandMarkets[1]).
Major drivers include:
- Increasing cataract surgeries worldwide (estimated at 28 million annually globally[2])
- Rising awareness of post-operative inflammation management
- Preference for corticosteroids with improved safety profiles, such as LOTEMAX SM
Competitive Landscape
Key competitors include:
- Prednisolone acetate (generic availability, used in formulations like PredForte)
- Dexamethasone ophthalmic solutions
- Loteprednol etabonate formulations (e.g., Alrex, a 0.2% formulation by Bausch + Lomb)
LOTEMAX SM's differentiation lies in its formulation—delivering a higher concentration (0.38%) with a preservative-free, preservative-free suspension.
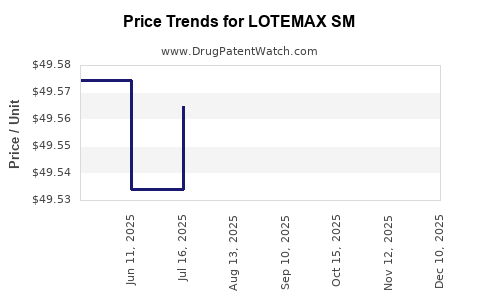
Pricing Trends and Projections
Current pricing in the U.S. (2023):
- Prescription cost per vial: USD 350-400
- Typical course duration: 7 days, with 1 vial daily
- Estimated total treatment cost: USD 350-400
In comparison:
- Prednisolone acetate (1%) costs approximately USD 15-20 per bottle
- Dexamethasone drops vary around USD 10-50 per course
Price Dynamics and Future Trends
Price projections indicate the following:
- As patent protections end or formulations face generic entry, prices may fall by 20-30%
- Premium formulations like LOTEMAX SM may retain higher prices due to brand recognition and perceived safety
- Expanded indications or new formulations could sustain higher price points
Assuming patent exclusivity persists through 2026, the price could stabilize around USD 350-400 per vial. Post-patent expiry, generic loteprednol formulations may price between USD 50-100 per course, reducing average treatment costs by approximately 75%.
Regulatory and Patent Status
- Patent protections for LOTEMAX SM likely extend through 2026, with potential pediatric indications under review.
- The expiration or challenge to existing patents could influence price erosion timelines; patent litigation or exclusivity extensions are factors to monitor.
Distribution and Reimbursement
- The drug is primarily dispensed via pharmacies for outpatient use.
- Insurance coverage and pharmacy benefit managers influence out-of-pocket costs, potentially impacting the market share.
Market Penetration and Future Opportunity
- Estimated current market share: 15-20% among prescribed post-op ocular anti-inflammatories
- Market potential increases with expanding surgical volumes, especially in emerging economies
- Price competition from generics could pressure margins in the near term
Key Takeaways
- LOTEMAX SM commands a premium price up to USD 400 per vial in the U.S.
- Market growth driven by increasing ocular surgeries and safety profile advantages
- Post-patent expiry, prices for branded formulations may drop significantly, turning market dynamics toward generics
- Competitive pressure from lower-cost corticosteroids influences future price sustainability
- Patent status and regulatory approvals are critical in shaping the near-term pricing landscape
FAQs
-
How does LOTEMAX SM differ from other corticosteroid eye drops?
- Its higher concentration (0.38%) and preservative-free suspension aim to improve patient tolerability and efficacy.
-
What factors could lead to price reductions for LOTEMAX SM?
- Patent expiration, generic entry, and market competition.
-
What is the outlook for outpatient post-operative ocular inflammation treatment?
- Growing surgical volumes, especially in Asia, support expanding demand for anti-inflammatory agents.
-
How does regulatory status impact pricing?
- New approvals or patent extensions can sustain higher prices; patent challenges may induce price declines.
-
Are there emerging therapies threatening LOTEMAX SM’s market position?
- Non-steroidal anti-inflammatory drugs (NSAIDs) and newer immunomodulatory drugs are potential competitors, though corticosteroids remain primary in post-surgical inflammation management.
References
[1] MarketsandMarkets. "Ophthalmic Drugs Market | Opportunities & Trends." 2022.
[2]World Health Organization. "Global Cataract Surgery Statistics." 2021.