LINZESS Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Linzess, and what generic alternatives are available?
Linzess is a drug marketed by Abbvie and is included in one NDA. There are seven patents protecting this drug and two Paragraph IV challenges.
This drug has two hundred and twenty-two patent family members in forty-five countries.
The generic ingredient in LINZESS is linaclotide. There are ten drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the linaclotide profile page.
DrugPatentWatch® Generic Entry Outlook for Linzess
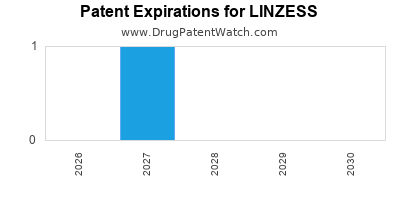
Linzess was eligible for patent challenges on August 30, 2016.
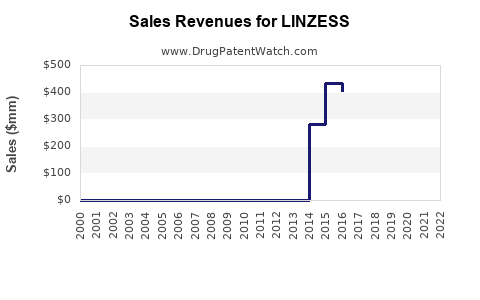
Annual sales in 2022 were $1.6bn, indicating a strong incentive for generic entry.
There have been eleven patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
There is one tentative approval for the generic drug (linaclotide), which indicates the potential for near-term generic launch.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for LINZESS?
- What are the global sales for LINZESS?
- What is Average Wholesale Price for LINZESS?
Summary for LINZESS
| International Patents: | 222 |
| US Patents: | 7 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 1 |
| Clinical Trials: | 19 |
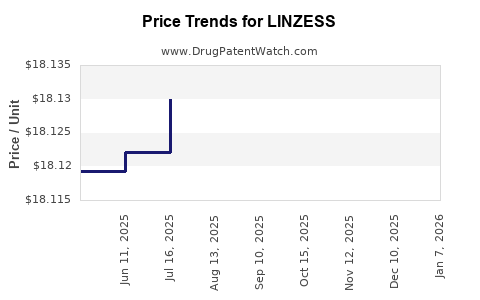
| Drug Prices: | Drug price information for LINZESS |
| Drug Sales Revenues: | Drug sales revenues for LINZESS |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for LINZESS |
| What excipients (inactive ingredients) are in LINZESS? | LINZESS excipients list |
| DailyMed Link: | LINZESS at DailyMed |
Recent Clinical Trials for LINZESS
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| AbbVie | Phase 2 |
| National Cancer Institute (NCI) | Early Phase 1 |
| RenJi Hospital | Phase 4 |
Pharmacology for LINZESS
| Drug Class | Guanylate Cyclase-C Agonist |
| Mechanism of Action | Guanylate Cyclase Activators |
Paragraph IV (Patent) Challenges for LINZESS
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| LINZESS | Capsules | linaclotide | 72 mcg | 202811 | 1 | 2017-11-07 |
| LINZESS | Capsules | linaclotide | 145 mcg and 290 mcg | 202811 | 4 | 2016-08-30 |
US Patents and Regulatory Information for LINZESS
LINZESS is protected by seven US patents and three FDA Regulatory Exclusivities.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Abbvie | LINZESS | linaclotide | CAPSULE;ORAL | 202811-003 | Jan 25, 2017 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Abbvie | LINZESS | linaclotide | CAPSULE;ORAL | 202811-002 | Aug 30, 2012 | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Abbvie | LINZESS | linaclotide | CAPSULE;ORAL | 202811-001 | Aug 30, 2012 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Abbvie | LINZESS | linaclotide | CAPSULE;ORAL | 202811-003 | Jan 25, 2017 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Abbvie | LINZESS | linaclotide | CAPSULE;ORAL | 202811-001 | Aug 30, 2012 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for LINZESS
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Abbvie | LINZESS | linaclotide | CAPSULE;ORAL | 202811-001 | Aug 30, 2012 | ⤷ Start Trial | ⤷ Start Trial |
| Abbvie | LINZESS | linaclotide | CAPSULE;ORAL | 202811-003 | Jan 25, 2017 | ⤷ Start Trial | ⤷ Start Trial |
| Abbvie | LINZESS | linaclotide | CAPSULE;ORAL | 202811-001 | Aug 30, 2012 | ⤷ Start Trial | ⤷ Start Trial |
| Abbvie | LINZESS | linaclotide | CAPSULE;ORAL | 202811-003 | Jan 25, 2017 | ⤷ Start Trial | ⤷ Start Trial |
| Abbvie | LINZESS | linaclotide | CAPSULE;ORAL | 202811-001 | Aug 30, 2012 | ⤷ Start Trial | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
EU/EMA Drug Approvals for LINZESS
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| AbbVie Deutschland GmbH & Co. KG | Constella | linaclotide | EMEA/H/C/002490Constella is indicated for the symptomatic treatment of moderate to severe irritable-bowel syndrome with constipation (IBS-C) in adults. | Authorised | no | no | no | 2012-11-26 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for LINZESS
When does loss-of-exclusivity occur for LINZESS?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Canada
Patent: 46230
Patent: TRAITEMENTS POUR DES TROUBLES GASTRO-INTESTINAUX (TREATMENTS FOR GASTROINTESTINAL DISORDERS)
Estimated Expiration: ⤷ Start Trial
China
Patent: 4053449
Patent: Treatments for gastrointestinal disorders
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 76055
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 76055
Patent: TRAITEMENTS POUR DES TROUBLES GASTRO-INTESTINAUX (TREATMENTS FOR GASTROINTESTINAL DISORDERS)
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 32237
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 12592
Estimated Expiration: ⤷ Start Trial
Patent: 14524444
Estimated Expiration: ⤷ Start Trial
Patent: 18002740
Patent: 消化器疾患の治療 (TREATMENT OF GASTROINTESTINAL DISORDERS)
Estimated Expiration: ⤷ Start Trial
Patent: 19085415
Patent: 消化器疾患の治療 (TREATMENT OF GASTROINTESTINAL DISORDERS)
Estimated Expiration: ⤷ Start Trial
Patent: 20128424
Patent: 消化器疾患の治療 (TREATMENTS FOR GASTROINTESTINAL DISORDERS)
Estimated Expiration: ⤷ Start Trial
Patent: 22010265
Patent: 消化器疾患の治療
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 7354
Patent: TRATAMIENTOS PARA TRASTORNOS GASTROINTESTINALES. (TREATMENTS FOR GASTROINTESTINAL DISORDERS.)
Estimated Expiration: ⤷ Start Trial
Patent: 14001798
Patent: TRATAMIENTOS PARA TRASTORNOS GASTROINTESTINALES. (TREATMENTS FOR GASTROINTESTINAL DISORDERS.)
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 76055
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 14864
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering LINZESS around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Japan | 2018002740 | 消化器疾患の治療 (TREATMENT OF GASTROINTESTINAL DISORDERS) | ⤷ Start Trial |
| Japan | 6034327 | ⤷ Start Trial | |
| South Korea | 20050106404 | METHODS AND COMPOSITIONS FOR THE TREATMENT OF GASTROINTESTINAL DISORDERS | ⤷ Start Trial |
| Norway | 20053864 | ⤷ Start Trial | |
| New Zealand | 549519 | Peptides that activate the guanylate cyclase C (GC-C) receptor for treating gastrointestinal disorders | ⤷ Start Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for LINZESS
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2246360 | 122013000045 | Germany | ⤷ Start Trial | PRODUCT NAME: LINACLOTID UND JEDE THERAPEUTISCH AEQUIVALENTE FORM DAVON, WELCHE DURCH DAS GRUNDPATENT GESCHUETZT IST, EINSCHLIESSLICH DESSEN PHARMAZEUTISCH VERTRAEGLICHE SALZE; REGISTRATION NO/DATE: EU/1/12/801/001-004 20121126 |
| 2246360 | 300594 | Netherlands | ⤷ Start Trial | PRODUCT NAME: LINACLOTIDE; REGISTRATION NO/DATE: EU/1/12/801/001-004 20121126 |
| 1594517 | 1390024-6 | Sweden | ⤷ Start Trial | PRODUCT NAME: LINAKLOTID; FIRST MARKETING AUTHORIZATION NUMBER SE: EG EU/1/12/801, 2012-11-28 |
| 1594517 | C01594517/01 | Switzerland | ⤷ Start Trial | PRODUCT NAME: LINACLOTID; REGISTRATION NO/DATE: SWISSMEDIC-ZULASSUNG 62684 20.06.2013 |
| 1594517 | 24/2013 | Austria | ⤷ Start Trial | PRODUCT NAME: LINACLOTID UND PHARMAZEUTISCH ANNEHMBAREN SALZE HIERVON; REGISTRATION NO/DATE: EU/1/12/801/001-004 (MITTEILUNG) 20121128 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for LINZESS (LINACLOTIDE)
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.