ISENTRESS Drug Patent Profile
✉ Email this page to a colleague
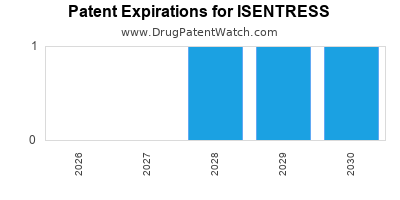
Which patents cover Isentress, and when can generic versions of Isentress launch?
Isentress is a drug marketed by Msd Sub Merck and is included in three NDAs. There are five patents protecting this drug.
This drug has seventy-six patent family members in thirty-seven countries.
The generic ingredient in ISENTRESS is raltegravir potassium. There are five drug master file entries for this compound. Four suppliers are listed for this compound. Additional details are available on the raltegravir potassium profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Isentress
A generic version of ISENTRESS was approved as raltegravir potassium by LUPIN LTD on May 6th, 2025.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for ISENTRESS?
- What are the global sales for ISENTRESS?
- What is Average Wholesale Price for ISENTRESS?
Summary for ISENTRESS
| International Patents: | 76 |
| US Patents: | 3 |
| Applicants: | 1 |
| NDAs: | 3 |
| Finished Product Suppliers / Packagers: | 4 |
| Raw Ingredient (Bulk) Api Vendors: | 76 |
| Clinical Trials: | 88 |
| Patent Applications: | 350 |
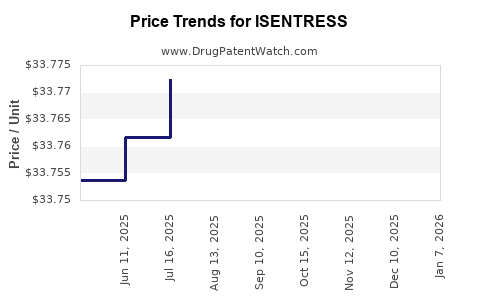
| Drug Prices: | Drug price information for ISENTRESS |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for ISENTRESS |
| What excipients (inactive ingredients) are in ISENTRESS? | ISENTRESS excipients list |
| DailyMed Link: | ISENTRESS at DailyMed |
Recent Clinical Trials for ISENTRESS
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Canadian Institutes of Health Research (CIHR) | Phase 2 |
| Unity Health Toronto | Phase 2 |
| St. Michael's Hospital, Toronto | Phase 2 |
Pharmacology for ISENTRESS
| Drug Class | Human Immunodeficiency Virus Integrase Strand Transfer Inhibitor |
| Mechanism of Action | HIV Integrase Inhibitors |
US Patents and Regulatory Information for ISENTRESS
ISENTRESS is protected by three US patents.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Msd Sub Merck | ISENTRESS | raltegravir potassium | POWDER;ORAL | 205786-001 | Dec 20, 2013 | RX | Yes | Yes | 7,754,731*PED | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Msd Sub Merck | ISENTRESS | raltegravir potassium | TABLET;ORAL | 022145-001 | Oct 12, 2007 | RX | Yes | Yes | 7,754,731*PED | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Msd Sub Merck | ISENTRESS | raltegravir potassium | TABLET, CHEWABLE;ORAL | 203045-002 | Dec 21, 2011 | RX | Yes | Yes | 7,754,731*PED | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Msd Sub Merck | ISENTRESS HD | raltegravir potassium | TABLET;ORAL | 022145-002 | May 26, 2017 | AB | RX | Yes | Yes | 8,771,733 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | |
| Msd Sub Merck | ISENTRESS | raltegravir potassium | TABLET, CHEWABLE;ORAL | 203045-001 | Dec 21, 2011 | RX | Yes | No | 7,754,731*PED | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Msd Sub Merck | ISENTRESS | raltegravir potassium | TABLET, CHEWABLE;ORAL | 203045-002 | Dec 21, 2011 | RX | Yes | Yes | 8,771,733 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for ISENTRESS
When does loss-of-exclusivity occur for ISENTRESS?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Australia
Patent: 10313571
Patent: Solid pharmaceutical compositions containing an integrase inhibitor
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 2012009857
Patent: composições farmacêuticas sólidas contendo um inibidor de integrase
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 77937
Patent: COMPOSITIONS PHARMACEUTIQUES SOLIDES CONTENANT UN INHIBITEUR D'INTEGRASE (SOLID PHARMACEUTICAL COMPOSITIONS CONTAINING AN INTEGRASE INHIBITOR)
Estimated Expiration: ⤷ Start Trial
China
Patent: 2655752
Patent: Solid pharmaceutical compositions containing an integrase inhibitor
Estimated Expiration: ⤷ Start Trial
Patent: 6074411
Patent: 包含整合酶抑制剂的固体药物组合物 (SOLID PHARMACEUTICAL COMPOSITIONS CONTAINING AN INTEGRASE INHIBITOR)
Estimated Expiration: ⤷ Start Trial
Colombia
Patent: 31485
Patent: COMPOSICIONES FARMACÉUTICAS SÓLIDAS QUE CONTIENEN UN INHIBIDOR DE LA INTEGRASA
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0211826
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 24914
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 93312
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 93312
Patent: COMPOSITIONS PHARMACEUTIQUES SOLIDES CONTENANT UN INHIBITEUR D'INTÉGRASE (SOLID PHARMACEUTICAL COMPOSITIONS CONTAINING AN INTEGRASE INHIBITOR)
Estimated Expiration: ⤷ Start Trial
Patent: 70702
Patent: COMPOSITIONS PHARMACEUTIQUES SOLIDES CONTENANT UN INHIBITEUR D'INTÉGRASE (SOLID PHARMACEUTICAL COMPOSITIONS CONTAINING AN INTEGRASE INHIBITOR)
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 57248
Estimated Expiration: ⤷ Start Trial
India
Patent: 77DEN2012
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 9369
Patent: תכשירים רוקחיים מוצקים המכילים מעכב אינטגראז (Solid pharmaceutical compositions containing an integrase inhibitor)
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 22639
Estimated Expiration: ⤷ Start Trial
Patent: 13508395
Estimated Expiration: ⤷ Start Trial
Patent: 16034962
Patent: インテグラーゼ阻害剤を含有する固形医薬組成物 (SOLID PHARMACEUTICAL COMPOSITIONS CONTAINING INTEGRASE INHIBITOR)
Estimated Expiration: ⤷ Start Trial
Lithuania
Patent: 93312
Estimated Expiration: ⤷ Start Trial
Malaysia
Patent: 2494
Patent: SOLID PHARMACEUTICAL COMPOSITIONS CONTAINING AN INTEGRASE INHIBITOR
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 5227
Patent: COMPOSICIONES FARMACEUTICAS SOLIDAS QUE CONTIENEN UN INHIBIDOR DE LA INTEGRASA. (SOLID PHARMACEUTICAL COMPOSITIONS CONTAINING AN INTEGRASE INHIBITOR.)
Estimated Expiration: ⤷ Start Trial
Patent: 12004903
Patent: COMPOSICIONES FARMACEUTICAS SOLIDAS QUE CONTIENEN UN HINIBIDOR DE LA INTEGRASA. (SOLID PHARMACEUTICAL COMPOSITIONS CONTAINING AN INTEGRASE INHIBITOR.)
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 0331
Patent: Solid pharmaceutical compositions containing an integrase inhibitor
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 93312
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 93312
Estimated Expiration: ⤷ Start Trial
Russian Federation
Patent: 02865
Patent: ТВЕРДЫЕ ФАРМАЦЕВТИЧЕСКИЕ КОМПОЗИЦИИ, СОДЕРЖАЩИЕ ИНГИБИТОР ИНТЕГРАЗЫ (SOLID PHARMACEUTICAL COMPOSITIONS CONTAINING INTEGRASE INHIBITOR)
Estimated Expiration: ⤷ Start Trial
Patent: 12121857
Patent: ТВЕРДЫЕ ФАРМАЦЕВТИЧЕСКИЕ КОМПОЗИЦИИ, СОДЕРЖАЩИЕ ИНГИБИТОР ИНТЕГРАЗЫ
Estimated Expiration: ⤷ Start Trial
Serbia
Patent: 600
Patent: ČVRSTE FARMACEUTSKE KOMPOZICIJE KOJE SADRŽE INHIBITOR INTEGRAZE (SOLID PHARMACEUTICAL COMPOSITIONS CONTAINING AN INTEGRASE INHIBITOR)
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 93312
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 1203012
Patent: SOLID PHARMACEUTICAL COMPOSITIONS CONTAINING AN INTERGRASE INHIBITOR
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 1835893
Estimated Expiration: ⤷ Start Trial
Patent: 120102063
Patent: SOLID PHARMACEUTICAL COMPOSITIONS CONTAINING AN INTEGRASE INHIBITOR
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 98348
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering ISENTRESS around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Slovenia | 2493312 | ⤷ Start Trial | |
| Lithuania | PA2008007 | ⤷ Start Trial | |
| Russian Federation | 2602865 | ТВЕРДЫЕ ФАРМАЦЕВТИЧЕСКИЕ КОМПОЗИЦИИ, СОДЕРЖАЩИЕ ИНГИБИТОР ИНТЕГРАЗЫ (SOLID PHARMACEUTICAL COMPOSITIONS CONTAINING INTEGRASE INHIBITOR) | ⤷ Start Trial |
| Canada | 2588398 | SELS DE POTASSIUM D'UN INHIBITEUR D'INTEGRASE HIV (POTASSIUM SALT OF AN HIV INTEGRASE INHIBITOR) | ⤷ Start Trial |
| Slovenia | 1819700 | ⤷ Start Trial | |
| China | 102229605 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for ISENTRESS
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1441735 | PA2008 007, C1441735 | Lithuania | ⤷ Start Trial | PRODUCT NAME: RALTEGRAVIRUM; REGISTRATION NO/DATE: EU/1/07/436/001-002 20071220 |
| 1441735 | C20080001 | Estonia | ⤷ Start Trial | PRODUCT NAME: ISENTRESS; AUTHORISATION NO.: EMA/106827/2018; AUTHORISATION DATE: 20180222 |
| 1441735 | 0890018-5 | Sweden | ⤷ Start Trial | PRODUCT NAME: RALTEGRAVIR |
| 1441735 | 67 3-2008 | Slovakia | ⤷ Start Trial | PRODUCT NAME: RALTEGRAVIR; REGISTRATION NO/DATE: EU/1/07/436/001 - EU/1/07/436/002 20080102 |
| 1441735 | C200800017 | Spain | ⤷ Start Trial | PRODUCT NAME: RALTEGRAVIR; NATIONAL AUTHORISATION NUMBER: UE/1/07/436/001-002; DATE OF AUTHORISATION: 20071220; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): UE/1/07/436/001-002; DATE OF FIRST AUTHORISATION IN EEA: 20071220 |
| 1441735 | C300340 | Netherlands | ⤷ Start Trial | PRODUCT NAME: RALTEGRAVIR OF FARMACEUTISCH AANVAARDBAAR ZOUT DAARVAN, IN HET BIJZONDER HET KALIUMZOUT; REGISTRATION NO/DATE: EU/1/07/436/001-002 20071220 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
ISENTRESS: Market Dynamics and Financial Trajectory
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.