Last updated: February 19, 2026
Glucotrol XL, an extended-release formulation of the oral sulfonylurea glipizide, is a second-generation medication for managing type 2 diabetes mellitus. Its market performance is shaped by the competitive landscape of oral antidiabetic agents, evolving treatment guidelines, and patent expirations. This analysis details the drug's current market standing, patent status, and projected financial trajectory, informed by regulatory filings, market research data, and financial disclosures.
What is the current market positioning of Glucotrol XL?
Glucotrol XL is positioned as a cost-effective oral hypoglycemic agent, primarily targeting patients with type 2 diabetes who require glycemic control beyond diet and exercise. Its established efficacy in lowering fasting and postprandial blood glucose levels has maintained its relevance, particularly in markets where affordability is a significant factor.
The drug competes with a broad spectrum of antidiabetic medications, including other sulfonylureas, biguanides (metformin), thiazolidinediones, DPP-4 inhibitors, SGLT2 inhibitors, and GLP-1 receptor agonists. The therapeutic landscape has shifted towards newer agents offering improved cardiovascular and renal benefits, which can impact Glucotrol XL's market share, especially in developed markets with higher healthcare spending and physician preference for guideline-recommended agents.
- Market Share: While precise, up-to-the-minute market share data is proprietary, industry analyses suggest Glucotrol XL holds a modest but stable share within the sulfonylurea class. Its market share has been impacted by the rise of metformin as a first-line therapy and the adoption of newer drug classes.
- Geographic Penetration: Glucotrol XL has a significant presence in both developed and emerging markets. Its accessibility and lower price point make it a viable option for a larger patient population globally. In developed markets, it is often utilized as a second- or third-line therapy or in specific patient populations prioritizing cost savings.
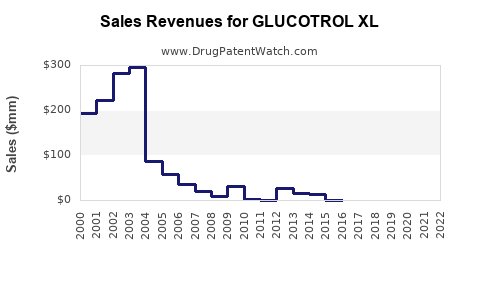
- Sales Performance: Historically, Glucotrol XL has generated substantial revenue. However, sales have experienced a decline due to generic competition and the introduction of novel antidiabetic drugs. The exact figures for recent fiscal periods are contingent on the reporting of the marketing pharmaceutical entity. For instance, Pfizer, the originator, has seen its diabetes portfolio evolve, with some older assets like Glucotrol XL experiencing revenue shifts as newer treatments take prominence.
What is the patent landscape and generic competition status for Glucotrol XL?
The patent protection for the original Glucotrol XL formulation has long expired, paving the way for generic versions. The primary patent for the extended-release formulation, U.S. Patent No. 4,983,417, expired in the early 2000s.
- Exclusivity Periods: Following the expiration of key patents, Glucotrol XL transitioned to a genericized market. The absence of active patent protection means that multiple manufacturers can produce and market generic equivalents.
- Generic Approvals: The U.S. Food and Drug Administration (FDA) and other regulatory bodies have approved numerous generic versions of glipizide extended-release tablets. This has led to significant price erosion and increased competition.
- Legal Challenges: While the primary patents have expired, there can be instances of litigation concerning secondary patents or manufacturing processes. However, such challenges have not demonstrably extended the market exclusivity for the original Glucotrol XL.
- Market Impact of Generics: The availability of generic glipizide extended-release has resulted in substantial cost savings for healthcare systems and patients. This has diminished the revenue potential for the branded product and shifted market dynamics towards a price-sensitive environment. Manufacturers of branded Glucotrol XL have focused on lifecycle management strategies, though these are less impactful now given the drug's mature status.
What are the key drivers and inhibitors of Glucotrol XL's future market performance?
The future market performance of Glucotrol XL is influenced by a confluence of factors, ranging from evolving clinical practice to economic considerations.
Key Drivers
- Cost-Effectiveness: In numerous global markets, particularly those with budget constraints or high out-of-pocket patient costs, Glucotrol XL's affordability remains a significant driver. Its established efficacy at a lower price point makes it a persistent choice for a segment of the diabetic population.
- Established Efficacy and Safety Profile: Glucotrol XL has a long history of clinical use. Prescribers and patients are familiar with its efficacy in lowering HbA1c levels and its general safety profile when used appropriately. This familiarity can lead to continued prescription, especially when newer, more expensive options are not preferred or accessible.
- Guideline Evolution (as a secondary option): While newer drug classes are prioritized, international diabetes treatment guidelines often still include sulfonylureas like glipizide as an option for patients who do not achieve glycemic targets with metformin or when newer agents are contraindicated or unaffordable.
- Emerging Markets: The expansion of healthcare access and the growing prevalence of type 2 diabetes in emerging economies present an ongoing demand for cost-effective treatments like Glucotrol XL.
Key Inhibitors
- Competition from Novel Therapies: The advent of SGLT2 inhibitors and GLP-1 receptor agonists, which offer cardiovascular and renal protective benefits in addition to glycemic control, represents a major challenge. These newer classes are increasingly recommended as first- or second-line therapies for patients with comorbid cardiovascular or renal disease, areas where sulfonylureas lack advantages.
- Weight Gain: A known side effect of sulfonylureas, including Glucotrol XL, is potential weight gain, which is undesirable in the management of type 2 diabetes where weight management is often a primary goal.
- Hypoglycemia Risk: While the extended-release formulation aims to mitigate this, sulfonylureas carry a risk of hypoglycemia, particularly in elderly patients, those with renal impairment, or when combined with other hypoglycemic agents. This risk profile can make clinicians and patients opt for agents with a lower hypoglycemia incidence.
- Limited Cardiovascular/Renal Benefits: Unlike newer drug classes, Glucotrol XL does not offer proven benefits for cardiovascular or renal outcomes, which are critical considerations in modern diabetes management strategies.
- Genericization and Price Erosion: The presence of multiple generic manufacturers has led to significant price reductions, impacting the revenue potential for any remaining branded product and limiting profitability for generic manufacturers as well.
What is the projected financial trajectory for Glucotrol XL?
The financial trajectory for Glucotrol XL is characterized by a mature market status and ongoing, albeit diminishing, revenue generation primarily from generic sales. The branded product's revenue has largely stabilized or declined as generic competition intensified.
- Revenue Streams: The primary revenue now stems from generic glipizide extended-release. Manufacturers of generic Glucotrol XL compete on price and market access.
- Market Size (Global Oral Hypoglycemics): The global market for oral antidiabetic drugs is substantial and growing, driven by the increasing prevalence of diabetes. However, Glucotrol XL's share within this expanding market is expected to remain modest and potentially decline in relative terms as newer, higher-priced drug classes gain traction.
- Pricing Trends: Generic pricing for Glucotrol XL is highly competitive. Profit margins for manufacturers are typically lower compared to branded drugs. The trend is toward continued price pressure in the generic space.
- Volume vs. Value: While the volume of glipizide extended-release prescriptions may remain stable or even increase in certain regions due to cost-effectiveness, the overall value generated by the drug is constrained by pricing.
- Projected Growth: Forecasts generally indicate a stable to slightly declining revenue trend for Glucotrol XL globally over the next five to ten years. Growth opportunities are limited due to its genericized status and the competitive pressures from novel therapies. Any revenue growth would likely be tied to increased diabetes prevalence in emerging markets and the cost-conscious prescribing habits within those regions.
- Investment Outlook: For pharmaceutical companies, Glucotrol XL represents a mature product with limited upside potential. Investment would typically be focused on cost-efficient manufacturing and supply chain management rather than on R&D for new indications or formulations. For investors, the drug's financial contribution is likely to be a steady but minor component of a broader diabetes portfolio, primarily through generic sales.
What are the regulatory considerations and clinical practice trends impacting Glucotrol XL?
Regulatory bodies and evolving clinical guidelines play a significant role in shaping the market for Glucotrol XL.
- Regulatory Approvals: Glucotrol XL, as an established drug, has broad regulatory approvals for type 2 diabetes. The regulatory focus now is on post-market surveillance and ensuring the quality and consistency of generic manufacturing.
- Treatment Guidelines: The American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD) are key influencers of treatment guidelines. Current guidelines emphasize a patient-centered approach, often prioritizing metformin as first-line therapy, followed by agents with proven cardiovascular and renal benefits (SGLT2 inhibitors and GLP-1 receptor agonists) for patients with established cardiovascular disease or high cardiovascular risk. Sulfonylureas are typically placed in later-line treatment algorithms or for specific patient profiles where cost is a primary concern and other agents are not suitable.
- Pharmacovigilance: As with all medications, Glucotrol XL is subject to ongoing pharmacovigilance. Any newly identified serious adverse events could lead to updated prescribing information or, in rare cases, further regulatory action, though this is less likely for a long-established drug.
- Off-Label Use: Off-label uses are not a significant driver for Glucotrol XL, as its primary indication is well-defined.
- Healthcare Policy: Government policies related to drug pricing, reimbursement, and formulary placement significantly impact Glucotrol XL's market access. Policies favoring the lowest-cost generic options can sustain its use, while those promoting newer therapies with documented outcome benefits may diminish its role.
Key Takeaways
Glucotrol XL, a genericized sulfonylurea for type 2 diabetes, maintains a market presence primarily due to its cost-effectiveness and established efficacy. However, its financial trajectory is characterized by price erosion and a diminishing role in treatment guidelines, which increasingly favor novel therapies with superior cardiovascular and renal benefits. Future revenue is largely dependent on generic sales, particularly in emerging markets.
Frequently Asked Questions
1. What is the primary mechanism of action of Glucotrol XL?
Glucotrol XL (glipizide extended-release) is a sulfonylurea that stimulates the pancreas to release more insulin and reduces the amount of glucose produced by the liver. This action helps to lower blood sugar levels in individuals with type 2 diabetes.
2. Does Glucotrol XL offer any cardiovascular or renal benefits?
No, unlike newer classes of antidiabetic medications such as SGLT2 inhibitors and GLP-1 receptor agonists, Glucotrol XL does not provide proven cardiovascular or renal protective benefits. Its primary function is glycemic control.
3. What are the main side effects associated with Glucotrol XL?
The most significant side effect of Glucotrol XL, like other sulfonylureas, is hypoglycemia (low blood sugar). Other potential side effects include weight gain, nausea, diarrhea, and dizziness.
4. How does Glucotrol XL compare to Metformin in current treatment guidelines?
Metformin is generally recommended as the first-line oral therapy for type 2 diabetes by major clinical guidelines due to its efficacy, low risk of hypoglycemia, and potential cardiovascular benefits. Glipizide (including Glucotrol XL) is often considered a second- or third-line option, particularly when metformin is insufficient or not tolerated, or when cost is a primary consideration.
5. What is the outlook for the branded Glucotrol XL product versus its generic versions?
The branded Glucotrol XL product has largely been superseded by its generic counterparts. The market is now dominated by generic glipizide extended-release tablets, with competition focused on price and accessibility. Revenue for the originator brand has significantly declined, while generic manufacturers continue to generate sales based on volume and cost-competitiveness.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Drugs@FDA. Retrieved from https://www.accessdata.fda.gov/scripts/cder/daf/
[2] American Diabetes Association. (2023). Standards of Care in Diabetes—2023. Diabetes Care, 46(Supplement_1).
[3] European Association for the Study of Diabetes. (2019). Guidelines for the management of type 2 diabetes. Retrieved from https://www.easd.org/
[4] Pfizer Inc. (Various Years). Annual Reports and Financial Filings. (Specific reports and dates would vary; accessible via SEC EDGAR database or company investor relations).
[5] Market research reports on the global diabetes drug market (e.g., from companies like EvaluatePharma, IQVIA, GlobalData). Specific reports are proprietary and not publicly cited by title here.