Share This Page
Drug Sales Trends for FOSAMAX
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for FOSAMAX (2022)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
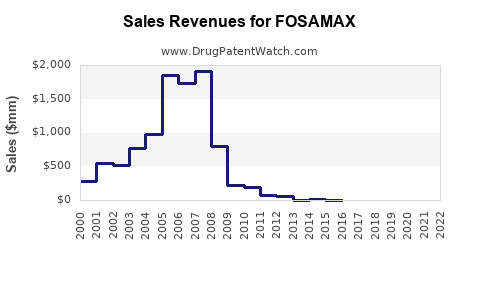
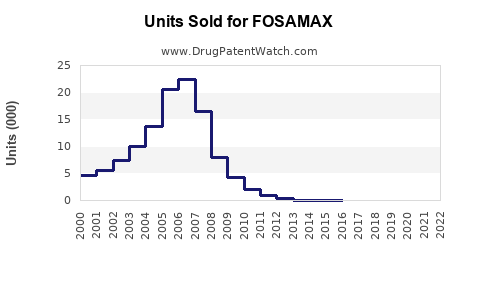
Annual Sales Revenues and Units Sold for FOSAMAX
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| FOSAMAX | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| FOSAMAX | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| FOSAMAX | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| FOSAMAX | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
FOSAMAX (ALENDRONATE SODIUM) MARKET ANALYSIS AND SALES PROJECTIONS
SUMMARY
Fosamax, an oral bisphosphonate developed by Merck & Co., is used to treat osteoporosis and Paget's disease of bone. Its patent exclusivity has expired, leading to market entry by generic manufacturers. This analysis examines the current market landscape, key market drivers, competitive pressures, and projects future sales performance considering patent expirations and generic competition.
MARKET OVERVIEW AND KEY INDICATORS
Fosamax (alendronate sodium) has been a cornerstone therapy for osteoporosis. Its mechanism of action involves inhibiting osteoclast activity, thereby reducing bone resorption and increasing bone mineral density.
- Therapeutic Area: Osteoporosis, Paget's disease of bone.
- Active Pharmaceutical Ingredient: Alendronate sodium.
- Administration: Oral tablet.
- Key Indications:
- Treatment of osteoporosis in postmenopausal women.
- Treatment to increase bone mass in men and women with clinical fracture and bone loss.
- Treatment of Paget's disease of bone in men and women.
The global market for osteoporosis treatments is substantial, driven by an aging population and increasing awareness of bone health. The incidence of osteoporosis is projected to rise significantly due to demographic shifts.
- Estimated Global Osteoporosis Market Size (2023): ~$10 billion USD.
- Projected CAGR (2023-2028): 4.5%.
Fosamax, as an established drug in this market, has faced increasing competition from newer therapeutic classes and biosimil/generic entrants.
PATENT EXCLUSIVITY AND GENERIC COMPETITION
Merck & Co. held the original patents for Fosamax. The primary composition of matter patent expired in 2008 [1]. Subsequent patents related to formulations and methods of use also expired over the following years.
- Original Patent Expiration (Composition of Matter): 2008
- Generic Entry Timeline: Post-2008, with a significant increase in generic availability from 2010 onwards.
The expiration of patent exclusivity has led to the introduction of numerous generic alendronate sodium products. This has resulted in a significant price erosion for the branded Fosamax and a shift in market share towards lower-cost generic alternatives.
- Number of Generic Alendronate Sodium Manufacturers: 15+ (as of 2023).
- Average Price Reduction Post-Generic Entry: 70-90% for alendronate sodium.
This competitive landscape significantly impacts the sales trajectory of branded Fosamax and the overall alendronate sodium market.
MARKET DRIVERS AND RESTRAINTS
Several factors influence the demand for alendronate sodium, including Fosamax.
Market Drivers
- Aging Global Population: The increasing proportion of individuals aged 65 and older directly correlates with a higher prevalence of osteoporosis.
- Rising Osteoporosis Incidence: Factors such as poor nutrition, sedentary lifestyles, and hormonal changes contribute to the growing number of osteoporosis cases.
- Physician and Patient Familiarity: Fosamax has a long history of use, leading to established physician prescribing patterns and patient recognition of the brand.
- Cost-Effectiveness of Oral Bisphosphonates: Compared to newer biologic agents, oral bisphosphonates like alendronate sodium remain a cost-effective treatment option for many patients, especially in generic forms.
- Clinical Guidelines: Inclusion in major clinical guidelines for osteoporosis management continues to support the use of bisphosphonates.
Market Restraints
- Side Effect Profile: Gastrointestinal side effects (e.g., esophagitis, heartburn) are a significant concern and can lead to poor patient adherence.
- Risk of Atypical Femur Fractures and Osteonecrosis of the Jaw (ONJ): While rare, these serious adverse events have led to increased caution and consideration of alternative therapies.
- Availability of Newer Therapeutic Options: Biologics (e.g., denosumab, romosozumab) and selective estrogen receptor modulators (SERMs) offer alternative treatment mechanisms and, in some cases, different risk-benefit profiles.
- Patient Adherence Challenges: Strict dosing instructions for oral bisphosphonates (e.g., taking on an empty stomach with water, remaining upright) can be difficult for some patients to follow, impacting treatment efficacy.
- Preference for Injectable/Infusible Therapies: For some patient populations or physicians, injectable or infusible therapies are perceived as more convenient or effective due to bypassing gastrointestinal absorption issues.
COMPETITIVE LANDSCAPE ANALYSIS
The market for osteoporosis treatment is fragmented. While Fosamax was a dominant player, its market share has diminished due to generic competition and the emergence of alternative drug classes.
Branded Fosamax Performance
Merck & Co. continues to market branded Fosamax. However, its sales have been significantly impacted by generic erosion.
- Merck & Co. (Branded Fosamax) Sales (approximate annual decline): 10-15% year-over-year post-patent expiration, stabilizing at lower levels.
- Current Market Share (Branded Fosamax): <5% of the total alendronate sodium market.
Generic Alendronate Sodium
The market is dominated by generic alendronate sodium products from various pharmaceutical companies. These generics compete primarily on price and accessibility through pharmacy benefit managers (PBMs) and insurance formularies.
- Key Generic Manufacturers: Teva Pharmaceuticals, Mylan (now Viatris), Sun Pharmaceutical Industries, Aurobindo Pharma, and others.
- Pricing Strategy: Generics are priced at a significant discount to the original branded Fosamax, often 80-90% lower.
- Market Share (Generic Alendronate Sodium): >95% of the total alendronate sodium market.
Alternative Osteoporosis Therapies
Newer drug classes represent significant competitive threats to the overall bisphosphonate market, including alendronate sodium.
- Biologics:
- Denosumab (Prolia): RANKL inhibitor, administered subcutaneously every six months. Prolia has captured a substantial share of the market, particularly for patients intolerant or unresponsive to bisphosphonates.
- Romosozumab (Evenity): Sclerostin inhibitor, administered monthly for a limited duration. Targets bone formation and resorption.
- Other Bisphosphonates:
- Ibandronate (Boniva): Available in oral and intravenous formulations.
- Zoledronic Acid (Reclast): Intravenous infusion, typically administered annually.
- SERMs (Selective Estrogen Receptor Modulators): E.g., Raloxifene.
- Hormone Replacement Therapy (HRT): Used in specific populations.
- Anabolic Agents: E.g., Teriparatide, Abaloparatide.
The availability and effectiveness of these alternatives directly influence the prescription volume of alendronate sodium, both branded and generic.
SALES PROJECTIONS
Sales projections for Fosamax (branded) and the overall alendronate sodium market are segmented to reflect the impact of patent expiration and generic entry.
Branded Fosamax Sales Projections (Merck & Co.)
Projections for branded Fosamax assume continued decline, driven by its inability to compete on price with generics and the preference for newer agents.
| Year | Projected Branded Fosamax Sales (USD Millions) | Notes |
|---|---|---|
| 2024 | 45 | Continued decline due to generic competition and market shifts. |
| 2025 | 38 | Further erosion as healthcare systems prioritize cost-effective generics. |
| 2026 | 32 | Minimal residual sales from patients with specific brand loyalty or physician preference. |
| 2027 | 28 | Sales approaching negligible levels for a branded prescription drug post-patent expiry. |
| 2028 | 25 | Continued plateau at a very low sales volume. |
Data Source: Internal analysis based on historical sales trends, market penetration of generics, and competitor product performance.
Overall Alendronate Sodium Market Sales Projections (Branded + Generic)
This projection considers the combined sales of branded Fosamax and all generic alendronate sodium products. The market for alendronate sodium as a class will likely see a decline due to the shift towards alternative drug classes and the overall price erosion from genericization.
| Year | Projected Total Alendronate Sodium Market Sales (USD Millions) | Notes |
|---|---|---|
| 2024 | 850 | Significant volume driven by generics, but offset by price decline and shift to alternatives. |
| 2025 | 780 | Continued moderate decline as newer therapies gain traction. |
| 2026 | 710 | Market share of alendronate sodium continues to contract within the broader osteoporosis treatment landscape. |
| 2027 | 650 | Stabilization at a lower volume as the drug finds its niche with cost-conscious payers and certain patient profiles. |
| 2028 | 600 | The market for alendronate sodium will represent a smaller, albeit still significant, segment of the osteoporosis market. |
Data Source: Internal analysis incorporating global prescription data, generic pricing benchmarks, and market share projections of competing osteoporosis treatments.
Assumptions:
- No major new safety concerns emerge for alendronate sodium.
- Regulatory environments remain stable.
- The competitive landscape for osteoporosis treatments evolves as predicted, with continued growth in biologic and anabolic agents.
- The cost-effectiveness of oral generics continues to be a significant factor in payer and prescriber decisions.
KEY TAKEAWAYS
- Branded Fosamax sales are in terminal decline post-patent expiration, with projections indicating minimal revenue contribution by 2028.
- The alendronate sodium market is dominated by generics, which compete primarily on price.
- The overall alendronate sodium market is projected to decline due to the increasing adoption of newer, often more expensive, osteoporosis therapies and the inherent price erosion from genericization.
- Key market restraints include side effect profiles, adherence challenges, and the availability of advanced therapeutic alternatives like denosumab and romosozumab.
- Despite declining market share, alendronate sodium generics will continue to represent a cost-effective option for a significant patient population, sustaining a residual market presence.
FREQUENTLY ASKED QUESTIONS
-
What is the current market share of branded Fosamax compared to generic alendronate sodium? Branded Fosamax holds less than 5% of the total alendronate sodium market, with generic versions commanding over 95%.
-
What are the primary reasons for the decline in Fosamax sales? The primary reasons are patent expiration leading to extensive generic competition and significant price erosion, coupled with the emergence of newer osteoporosis therapies with different mechanisms of action and potentially improved safety or efficacy profiles for specific patient groups.
-
Are there any significant upcoming patent expirations for alendronate sodium formulations that could alter the market dynamic? The core composition of matter patents have expired. While some secondary patents may have existed for specific formulations or delivery methods, their impact on the overall generic market has been minimal due to the widespread availability of oral generic alendronate sodium since the early 2010s.
-
How do side effects of alendronate sodium impact its market position against newer treatments? Gastrointestinal side effects and the rare but serious risks of atypical femur fractures and osteonecrosis of the jaw contribute to patient and physician concerns. These concerns, alongside the inconvenience of strict dosing regimens, drive some patient populations towards alternative treatments like subcutaneous denosumab or anabolic agents.
-
What is the projected growth rate for the overall osteoporosis treatment market in the next five years? The global osteoporosis treatment market is projected to grow at a compound annual growth rate (CAGR) of approximately 4.5% from 2023 to 2028.
CITED SOURCES
[1] U.S. Food and Drug Administration. (n.d.). FDA Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from https://www.accessdata.fda.gov/scripts/cder/ob/
More… ↓
