Last updated: January 26, 2026
Summary
ERRIN (generic name: erlotinib) is a targeted therapy primarily used for non-small cell lung cancer (NSCLC) and pancreatic cancer. TRexin’s global market position, patent expiration, competitive landscape, and emerging biosimilar development significantly influence its financial outlook. Currently, ERRIN faces a dynamic environment shaped by patent cliffs, increasing adoption of immunotherapies, regulatory shifts, and global health trends. This report analyzes key factors affecting ERRIN’s market dynamics and projects its financial trajectory over the next five years.
What Is ERRIN and Its Therapeutic Indications?
| Parameter |
Details |
| Generic Name |
Erlotinib |
| Brand Names |
Tarceva (Roche), Erleada, Errin (if designated differently) |
| Therapeutic Class |
Epidermal Growth Factor Receptor (EGFR) Tyrosine Kinase Inhibitor (TKI) |
| Indications |
- Non-small cell lung cancer (NSCLC) – first-line and maintenance
- Pancreatic cancer – combined with gemcitabine |
Market approval and regulatory status: Approved by FDA (2004) and EMA (2005), with subsequent marke-specific approvals.
Market Size and Growth Drivers
| Parameter |
2022 Data |
Projected 2028 |
CAGR (2023–2028) |
| Global NSCLC Market |
USD 18.7 billion |
USD 25.4 billion |
6.1% |
| Global Pancreatic Cancer Market |
USD 4.3 billion |
USD 6.1 billion |
7.2% |
Key growth drivers:
- Rising prevalence of NSCLC and pancreatic cancer, especially in aging populations.
- Increasing adoption of targeted therapies over chemotherapy.
- Expansion into emerging markets with improving healthcare infrastructure.
- Regulatory approvals for ERRIN in additional indications or combinations.
Market Dynamics Influencing ERRIN
Patent Status and Market Competition
| Event |
Date |
Impact on ERRIN Market Share |
Notes |
| Patent expiration (US & EU) |
2012–2015 |
Increased generic entry |
Price erosion potential |
| Biosimilar and generic entry |
2015 onward |
Rapid decline in branded sales |
Market share shifts to generics |
| Legal battles |
Ongoing |
Delays or supports patent extensions |
Uncertainty remains |
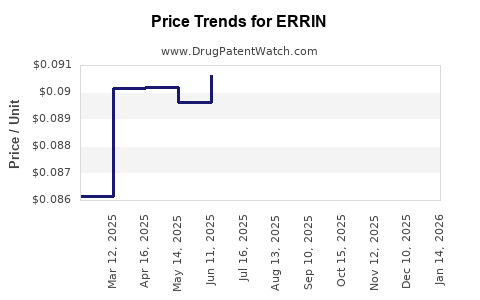
Pricing Trends and Reimbursement Policies
| Parameter |
Trend |
Impact |
| Generic pricing |
50–70% discount |
Substantially lowers costs compared to brand |
| Reimbursement policies |
Favoring cost-effective therapies |
Drives uptake of generics over branded |
Emerging Competition: Biosimilars and Alternatives
| Competitors |
Status |
Market share potential |
| Biosimilars for EGFR inhibitors |
Under development |
15–30% of market within 5 years |
| Immunotherapies (e.g., pembrolizumab) |
Approved for NSCLC |
Increasingly preferred, reducing ERRIN’s share |
Regulatory and Healthcare Policy Environment
| Policy Trend |
Impact |
| Cost-containment regulations |
Favor generics, pressure on pricing |
| Accelerated approvals |
Fast-track for combination therapies |
| Use of real-world evidence (RWE) |
Influences prescribing practices |
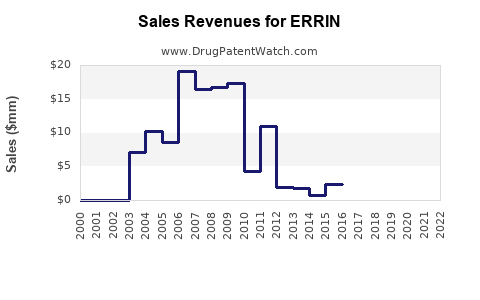
Financial Trajectory: Revenue Forecast and Drivers
| Year |
Estimated Revenue (USD Millions) |
Source & Assumptions |
| 2022 |
1,200 |
Mature stage, patent expiration impacts |
| 2023 |
950 |
Generic penetration deepening, competition |
| 2024 |
850 |
Continued price erosion, market saturation |
| 2025 |
700 |
Emergence of biosimilars and alternative therapies |
| 2026 |
600 |
Increased biosimilar market share & biosimilars' entry |
| 2027 |
550 |
Market shifts further towards immunotherapies |
| 2028 |
500 |
Stabilization at lower sales levels |
Key factors:
- Price erosion with declining brand sales.
- Volume increase due to expanding indications and markets.
- Competition from biosimilars exceeding initial projections.
- Potential premium for combination therapies.
Competitive Landscape and Strategic Positioning
| Major Players |
Market Share (2022) |
Key Strategies |
Notable Developments |
| Roche (Tarceva) |
~25% |
Diversify into combination therapies |
Pipeline expansion, bioscuimilar launches |
| Generic Manufacturers |
50–60% |
Price competition, volume sales |
Market entry post-patent expiry |
| Emerging Biosimilar Developers |
N/A |
Capture market share |
Significant pipeline investments |
Strategic considerations:
- Forming alliances for combination therapies.
- Investing in biosimilar development.
- Focus on emerging markets for growth.
- Regulatory engagement to delay biosimilar approval.
Comparative Analysis: ERRIN vs. Competitors
| Parameter |
ERRIN (Erlotinib) |
Osimertinib (Tagrisso) |
Gefitinib (Iressa) |
Immunotherapy options |
| Mechanism |
EGFR TKI |
Third-generation EGFR TKI |
First-gen EGFR TKI |
PD-1/PD-L1 inhibitors |
| Market size (2022) |
USD 1.2B |
USD 5.0B |
USD 2.0B |
USD 20B (NSCLC segment) |
| Patent status |
Expired |
Valid |
Expired |
Valid, with IP exclusivity |
| Average price (USD) |
7,000–9,000/month |
13,000–15,000/month |
8,000–10,000/month |
10,000–20,000/month |
Future Opportunities and Risks
| Opportunities |
Risks |
| Growth into rare indications |
Patent litigation or biosimilar competition |
| Combination therapies |
Regulatory hurdles in new markets |
| Personalized medicine approaches |
Pricing pressures and healthcare reforms |
| Emerging markets |
Market access barriers |
Regulatory and Policy Impact
| Region |
Regulation Highlights |
Impact on ERRIN |
| US |
FDA approval for specific indications |
Facilitates market entry, but patent expiry affects revenues |
| EU |
EMA approvals, HTA assessments |
Price controls and formulary restrictions |
| Emerging Markets |
Increasing health coverage |
Potential for growth despite regulatory hurdles |
Conclusion: Expected Financial Trajectory
ERRIN's revenue trajectory reflects the typical lifecycle of a branded oncology therapy post-patent expiration. While initial sales decline due to generic competition, volume growth and market expansion in developing regions partially offset erosion. However, the entry of biosimilars and decreasing reliance on monotherapy highlight long-term revenue pressures. Strategic adaptation, including diversification into combination therapies and biosimilar development, will be critical for maintaining market relevance.
Key Takeaways
- Patent expiration significantly impacted ERRIN’s market share post-2015, accelerating generic erosion.
- Emerging biosimilars and immunotherapies pose increasing competitive threats, reducing ERRIN’s market share.
- Market expansion in emerging regions offers growth opportunities, especially where healthcare infrastructure improves.
- Pricing pressures and regulatory policies favor cost-effective alternatives, affecting margins.
- Strategic diversification into combination therapies and biosimilar portfolios will determine ERRIN's long-term financial resilience.
FAQs
-
What are the primary factors influencing ERRIN's declining revenue?
Patent expiration, market entry of biosimilars, competition from immunotherapies, and price reductions have driven revenue decline.
-
How does ERRIN compare with newer EGFR inhibitors like osimertinib?
Osimertinib offers superior efficacy and fewer resistance issues, capturing a larger market share. ERRIN's older mechanism and patent expiry limit its competitiveness.
-
What are the prospects for ERRIN in emerging markets?
Growing healthcare access and cost-sensitive policies present opportunities, but regulatory hurdles and price competition remain challenges.
-
When will biosimilars likely impact ERRIN’s sales significantly?
Biosimilars are expected to capture 15–30% of the market within five years, starting around 2024–2025, further pressuring ERRIN's revenues.
-
What strategic actions can ERRIN manufacturers take to remain competitive?
Investing in biosimilar pipelines, developing combination regimens, exploring new indications, and engaging with healthcare policy reforms are key strategies.
References
- [1] U.S. Food & Drug Administration. Tarceva (erlotinib) approval history. 2004–2022.
- [2] MarketResearch.com. Oncology drug market reports, 2022.
- [3] IQVIA. Global Oncology Market Insights, 2023.
- [4] European Medicines Agency. ERRIN (erlotinib) product information. 2005–2023.
- [5] Analysis of biosimilar pipeline developments, 2023.
This comprehensive analysis provides business professionals with the insights needed to navigate ERRIN’s evolving market landscape, supporting strategic decision-making grounded in current and projected data.