ERLEADA Drug Patent Profile
✉ Email this page to a colleague
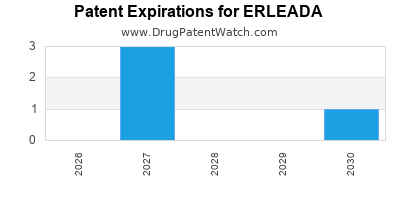
When do Erleada patents expire, and when can generic versions of Erleada launch?
Erleada is a drug marketed by Janssen Biotech and is included in one NDA. There are fourteen patents protecting this drug and two Paragraph IV challenges.
This drug has three hundred and fifty-seven patent family members in forty-nine countries.
The generic ingredient in ERLEADA is apalutamide. One supplier is listed for this compound. Additional details are available on the apalutamide profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Erleada
A generic version of ERLEADA was approved as apalutamide by ZYDUS on March 17th, 2025.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for ERLEADA?
- What are the global sales for ERLEADA?
- What is Average Wholesale Price for ERLEADA?
Summary for ERLEADA
| International Patents: | 357 |
| US Patents: | 14 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 74 |
| Clinical Trials: | 22 |
| Patent Applications: | 1,693 |
| Drug Prices: | Drug price information for ERLEADA |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for ERLEADA |
| What excipients (inactive ingredients) are in ERLEADA? | ERLEADA excipients list |
| DailyMed Link: | ERLEADA at DailyMed |
Recent Clinical Trials for ERLEADA
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| University of Washington | Phase 2 |
| Jonsson Comprehensive Cancer Center | Phase 2 |
| National Cancer Institute (NCI) | Phase 1 |
Paragraph IV (Patent) Challenges for ERLEADA
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| ERLEADA | Tablets | apalutamide | 240 mg | 210951 | 1 | 2025-03-20 |
| ERLEADA | Tablets | apalutamide | 60 mg | 210951 | 5 | 2022-02-14 |
US Patents and Regulatory Information for ERLEADA
ERLEADA is protected by eighteen US patents.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Janssen Biotech | ERLEADA | apalutamide | TABLET;ORAL | 210951-002 | Feb 17, 2023 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Janssen Biotech | ERLEADA | apalutamide | TABLET;ORAL | 210951-002 | Feb 17, 2023 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Janssen Biotech | ERLEADA | apalutamide | TABLET;ORAL | 210951-002 | Feb 17, 2023 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Janssen Biotech | ERLEADA | apalutamide | TABLET;ORAL | 210951-001 | Feb 14, 2018 | AB | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Janssen Biotech | ERLEADA | apalutamide | TABLET;ORAL | 210951-001 | Feb 14, 2018 | AB | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | Y | ⤷ Start Trial | |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for ERLEADA
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Janssen-Cilag International NV | Erleada | apalutamide | EMEA/H/C/004452Erleada is indicated:in adult men for the treatment of non metastatic castration resistant prostate cancer (nmCRPC) who are at high risk of developing metastatic disease.in adult men for the treatment of metastatic hormone-sensitive prostate cancer (mHSPC) in combination with androgen deprivation therapy (ADT). | Authorised | no | no | no | 2019-01-14 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for ERLEADA
When does loss-of-exclusivity occur for ERLEADA?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Australia
Patent: 13271751
Estimated Expiration: ⤷ Start Trial
Patent: 17200298
Estimated Expiration: ⤷ Start Trial
Patent: 17279807
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 2014030678
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 75767
Estimated Expiration: ⤷ Start Trial
Patent: 08345
Estimated Expiration: ⤷ Start Trial
Patent: 55660
Estimated Expiration: ⤷ Start Trial
Patent: 14726
Estimated Expiration: ⤷ Start Trial
Chile
Patent: 14003331
Estimated Expiration: ⤷ Start Trial
China
Patent: 4619692
Estimated Expiration: ⤷ Start Trial
Patent: 5693692
Estimated Expiration: ⤷ Start Trial
Patent: 3135892
Estimated Expiration: ⤷ Start Trial
Colombia
Patent: 40407
Estimated Expiration: ⤷ Start Trial
Costa Rica
Patent: 140549
Estimated Expiration: ⤷ Start Trial
Patent: 190331
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0180902
Estimated Expiration: ⤷ Start Trial
Patent: 0201387
Estimated Expiration: ⤷ Start Trial
Patent: 0210909
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 20393
Estimated Expiration: ⤷ Start Trial
Patent: 23427
Estimated Expiration: ⤷ Start Trial
Patent: 24831
Estimated Expiration: ⤷ Start Trial
Patent: 21032
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 58985
Estimated Expiration: ⤷ Start Trial
Patent: 48553
Estimated Expiration: ⤷ Start Trial
Patent: 33792
Estimated Expiration: ⤷ Start Trial
Ecuador
Patent: 14030098
Estimated Expiration: ⤷ Start Trial
Eurasian Patent Organization
Patent: 8791
Estimated Expiration: ⤷ Start Trial
Patent: 3956
Estimated Expiration: ⤷ Start Trial
Patent: 1492272
Estimated Expiration: ⤷ Start Trial
Patent: 1791592
Estimated Expiration: ⤷ Start Trial
Patent: 1992010
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 58985
Estimated Expiration: ⤷ Start Trial
Patent: 48553
Estimated Expiration: ⤷ Start Trial
Patent: 33792
Estimated Expiration: ⤷ Start Trial
Patent: 22629
Estimated Expiration: ⤷ Start Trial
France
Patent: C1050
Estimated Expiration: ⤷ Start Trial
Guatemala
Patent: 1400283
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 10175
Estimated Expiration: ⤷ Start Trial
Patent: 26066
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 38082
Estimated Expiration: ⤷ Start Trial
Patent: 50357
Estimated Expiration: ⤷ Start Trial
Patent: 54595
Estimated Expiration: ⤷ Start Trial
Patent: 100047
Estimated Expiration: ⤷ Start Trial
India
Patent: 084DEN2014
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 9738
Estimated Expiration: ⤷ Start Trial
Patent: 7608
Estimated Expiration: ⤷ Start Trial
Patent: 5413
Estimated Expiration: ⤷ Start Trial
Patent: 0522
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 82209
Estimated Expiration: ⤷ Start Trial
Patent: 45821
Estimated Expiration: ⤷ Start Trial
Patent: 15518890
Estimated Expiration: ⤷ Start Trial
Patent: 17178923
Estimated Expiration: ⤷ Start Trial
Patent: 18141009
Estimated Expiration: ⤷ Start Trial
Lithuania
Patent: 58985
Estimated Expiration: ⤷ Start Trial
Patent: 48553
Estimated Expiration: ⤷ Start Trial
Patent: 33792
Estimated Expiration: ⤷ Start Trial
Patent: 533792
Estimated Expiration: ⤷ Start Trial
Patent: 2021525
Estimated Expiration: ⤷ Start Trial
Luxembourg
Patent: 0236
Estimated Expiration: ⤷ Start Trial
Malaysia
Patent: 7500
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 6754
Estimated Expiration: ⤷ Start Trial
Patent: 14015005
Estimated Expiration: ⤷ Start Trial
Montenegro
Patent: 081
Estimated Expiration: ⤷ Start Trial
Patent: 815
Estimated Expiration: ⤷ Start Trial
Netherlands
Patent: 1144
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 2203
Estimated Expiration: ⤷ Start Trial
Patent: 7683
Estimated Expiration: ⤷ Start Trial
Nicaragua
Patent: 1400142
Estimated Expiration: ⤷ Start Trial
Norway
Patent: 21046
Estimated Expiration: ⤷ Start Trial
Peru
Patent: 150631
Estimated Expiration: ⤷ Start Trial
Patent: 200725
Estimated Expiration: ⤷ Start Trial
Patent: 200795
Estimated Expiration: ⤷ Start Trial
Philippines
Patent: 014502714
Estimated Expiration: ⤷ Start Trial
Patent: 016501470
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 58985
Estimated Expiration: ⤷ Start Trial
Patent: 48553
Estimated Expiration: ⤷ Start Trial
Patent: 33792
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 58985
Estimated Expiration: ⤷ Start Trial
Patent: 48553
Estimated Expiration: ⤷ Start Trial
Patent: 33792
Estimated Expiration: ⤷ Start Trial
San Marino
Patent: 01800311
Estimated Expiration: ⤷ Start Trial
Patent: 02000496
Estimated Expiration: ⤷ Start Trial
Patent: 02100355
Estimated Expiration: ⤷ Start Trial
Serbia
Patent: 370
Patent: KRISTALNI OBLICI MODULATORA RECEPTORA ANDROGENA (CRYSTALLINE FORMS OF AN ANDROGEN RECEPTOR MODULATOR)
Estimated Expiration: ⤷ Start Trial
Patent: 617
Patent: KRISTALNI OBLICI MODULATORA RECEPTORA ANDROGENA (CRYSTALLINE FORMS OF AN ANDROGEN RECEPTOR MODULATOR)
Estimated Expiration: ⤷ Start Trial
Patent: 988
Patent: KRISTALNI OBLICI MODULATORA RECEPTORA ANDROGENA (CRYSTALLINE FORMS OF AN ANDROGEN RECEPTOR MODULATOR)
Estimated Expiration: ⤷ Start Trial
Singapore
Patent: 201610248S
Patent: CRYSTALLINE FORMS OF AN ANDROGEN RECEPTOR MODULATOR
Estimated Expiration: ⤷ Start Trial
Patent: 201610249T
Patent: CRYSTALLINE FORMS OF AN ANDROGEN RECEPTOR MODULATOR
Estimated Expiration: ⤷ Start Trial
Patent: 201408140Q
Patent: CRYSTALLINE FORMS OF AN ANDROGEN RECEPTOR MODULATOR
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 58985
Estimated Expiration: ⤷ Start Trial
Patent: 48553
Estimated Expiration: ⤷ Start Trial
Patent: 33792
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 1500076
Patent: CRYSTALLINE FORMS OF ANDROGEN RECEPTOR MODULATOR
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 2062024
Estimated Expiration: ⤷ Start Trial
Patent: 2195916
Estimated Expiration: ⤷ Start Trial
Patent: 150021993
Estimated Expiration: ⤷ Start Trial
Patent: 190132543
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 70683
Estimated Expiration: ⤷ Start Trial
Patent: 09738
Estimated Expiration: ⤷ Start Trial
Patent: 75932
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 1402561
Patent: Crystalline forms of an androgen receptor modulator
Estimated Expiration: ⤷ Start Trial
Patent: 32732
Estimated Expiration: ⤷ Start Trial
Turkey
Patent: 1808939
Estimated Expiration: ⤷ Start Trial
Ukraine
Patent: 5665
Patent: КРИСТАЛІЧНІ ФОРМИ МОДУЛЯТОРА АНДРОГЕННОГО РЕЦЕПТОРА (CRYSTALLINE FORMS OF AN ANDROGEN RECEPTOR MODULATOR)
Estimated Expiration: ⤷ Start Trial
Patent: 3142
Patent: КРИСТАЛІЧНІ ФОРМИ МОДУЛЯТОРА АНДРОГЕННОГО РЕЦЕПТОРА
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering ERLEADA around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Taiwan | I844512 | ⤷ Start Trial | |
| South Korea | 20220025231 | 비전이성 거세 저항성 전립선암 치료용 항안드로겐 (ANTI-ANDROGENS FOR THE TREATMENT OF NON-METASTATIC CASTRATE-RESISTANT PROSTATE CANCER) | ⤷ Start Trial |
| Japan | 2012092149 | ⤷ Start Trial | |
| Japan | 2021130718 | 非転移性去勢抵抗性前立腺癌の治療のための抗アンドロゲン剤 (ANTI-ANDROGENS FOR TREATING NON-METASTATIC CASTRATION-RESISTANT PROSTATE CANCER) | ⤷ Start Trial |
| Lithuania | PA2019512 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for ERLEADA
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 3533792 | PA2021525,C3533792 | Lithuania | ⤷ Start Trial | PRODUCT NAME: APALUTAMIDAS; REGISTRATION NO/DATE: EU/1/18/1342 20190114 |
| 2368550 | 2019C/529 | Belgium | ⤷ Start Trial | PRODUCT NAME: APALUTAMIDE OU UN SEL PHARMACOLOGIQUEMENT ADMISSIBLE DE CELLES-CI; AUTHORISATION NUMBER AND DATE: EU/1/18/1342 20190116 |
| 3533792 | 202140035 | Slovenia | ⤷ Start Trial | PRODUCT NAME: APALUTAMID; NATIONAL AUTHORISATION NUMBER: EU/1/18/1342; DATE OF NATIONAL AUTHORISATION: 20190114; AUTHORITY FOR NATIONAL AUTHORISATION: EU |
| 3533792 | CR 2021 00041 | Denmark | ⤷ Start Trial | PRODUCT NAME: APALUTAMID; REG. NO/DATE: EU/1/18/1342 20190116 |
| 2368550 | PA2019512 | Lithuania | ⤷ Start Trial | PRODUCT NAME: APALUTAMIDAS ARBA JO FARMACINIU POZIURIU PRIIMTINA DRUSKA; REGISTRATION NO/DATE: EU/1/18/1342 20190114 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for ERLEADA (apalutamide)
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.