Last updated: February 19, 2026
Divigel, a transdermal testosterone gel, has established a notable presence in the male hypogonadism treatment market. Its efficacy in restoring testosterone levels, coupled with a convenient topical application, underpins its market position. This analysis examines the key market drivers, competitive landscape, patent status, and financial performance influencing Divigel's trajectory.
What is the Market for Hypogonadism Treatments?
The global market for hypogonadism treatments is driven by increasing awareness of the condition, a growing aging male population, and advancements in diagnostic tools. Hypogonadism, characterized by insufficient testosterone production, affects a significant portion of the male population, manifesting in symptoms such as low libido, fatigue, erectile dysfunction, and mood disturbances.
The market encompasses various therapeutic categories, including testosterone replacement therapies (TRTs) in different formulations such as injections, gels, patches, and pellets. Prescription rates for TRTs have steadily increased over the past decade, reflecting greater diagnosis and acceptance of treatment.
Key market segments within TRT include:
- Androgen Deficiency in Aging Males (ADAM): This segment addresses age-related decline in testosterone.
- Primary Hypogonadism: Conditions where the testes do not produce enough testosterone.
- Secondary Hypogonadism: Conditions where the pituitary gland or hypothalamus fails to signal the testes to produce testosterone.
The market size for hypogonadism treatments was estimated to be approximately $2.5 billion globally in 2022, with projections indicating a compound annual growth rate (CAGR) of 7-9% over the next five years [1]. This growth is fueled by factors including:
- Increasing Prevalence: An estimated 20% of men over 60 years old and 5% of men over 40 years old have hypogonadism [2].
- Diagnostic Advancements: Improved blood tests and clinical assessments facilitate earlier and more accurate diagnosis.
- Patient Preferences: A shift towards more convenient and less invasive administration methods, favoring gels and patches over injections [3].
- New Product Development: Ongoing research into novel TRT formulations and delivery systems.
How Does Divigel Fit into the Competitive Landscape?
Divigel (testosterone topical gel, 1%) competes in the testosterone gel segment of the TRT market. Its primary competitors include other transdermal testosterone products and alternative TRT formulations.
Key competitors and their products include:
- AbbVie Inc. (AndroGel): A dominant player in the testosterone gel market. AndroGel is a widely prescribed gel formulation and has historically held a significant market share.
- Eli Lilly and Company (Axiron): Another significant competitor offering a testosterone solution for topical application.
- Perrigo Company plc (Testosterone Gel): A generic manufacturer offering its own version of testosterone gel, impacting the pricing dynamics of branded products.
- Endo Pharmaceuticals (Testim): A testosterone gel formulation that competes directly with Divigel and AndroGel.
- Other TRT Formulations:
- Injectables: Testosterone enanthate and cypionate (e.g., Depo-Testosterone, Testosterone), testosterone undecanoate (e.g., Aveed).
- Patches: Androderm.
- Pellets: Testopel.
Divigel's market position is influenced by:
- Efficacy and Safety Profile: Clinical data supporting Divigel's ability to achieve and maintain target testosterone levels within the physiological range and its safety profile.
- Dosage and Administration: Divigel is typically applied to the shoulders and upper arms, a standard application site for gels. The gel formulation offers a controlled release mechanism.
- Pricing and Reimbursement: The cost of Divigel relative to competitors, and its coverage by insurance plans, are critical factors for market penetration.
- Prescriber Habits: Physician familiarity and preference for specific TRT formulations play a significant role in prescription patterns.
- Generic Competition: The availability of generic testosterone gels exerts downward pressure on the pricing of branded products, including Divigel, once patents expire or are successfully challenged.
The market for testosterone gels is characterized by intense competition, with product differentiation often centered on application convenience, absorption rates, and side effect profiles. Generic entry has become a substantial factor, leading to price erosion and a focus on brand loyalty and physician relationships for branded products.
What is the Patent Landscape for Divigel?
The patent landscape for Divigel is crucial for understanding its market exclusivity and future revenue potential. As a branded pharmaceutical product, its market exclusivity is largely defined by the expiration of its core composition of matter and formulation patents, as well as any method of use patents that may extend its protection.
Divigel, originally developed by TheraTech Inc. and later acquired by Solvay Pharmaceuticals, which was subsequently acquired by Abbott Laboratories (now AbbVie), has had its patent protection evolve over time.
Key patent considerations for Divigel:
- Core Patents: The initial patents covered the composition of matter for testosterone gel and its specific formulation.
- Expiration Dates: The primary patents for Divigel have largely expired in major markets. For instance, key U.S. patents related to the formulation expired several years ago.
- Generic Entry: The expiration of core patents has opened the door for generic manufacturers to produce and market their own versions of testosterone gel. Perrigo, for example, has a generic version of testosterone gel available [4].
- Secondary Patents: Pharmaceutical companies often pursue secondary patents related to manufacturing processes, specific dosages, new indications, or delivery systems to extend market exclusivity. The success of these secondary patents can be litigated.
- Exclusivity Periods: Beyond patent protection, regulatory exclusivities granted by agencies like the U.S. Food and Drug Administration (FDA) can also provide market protection. However, for older molecules like testosterone, these are typically shorter than patent terms.
The U.S. patent for the original composition of matter and formulation of Divigel expired around 2016-2017. This allowed for the introduction of generic testosterone gels, intensifying competition.
The patent situation means that while Divigel may retain brand recognition and physician loyalty, its pricing power and market share are significantly impacted by the availability of lower-cost generic alternatives.
What is Divigel's Financial Trajectory and Market Share?
Divigel's financial performance has been influenced by its patent status, competitive pressures, and market demand. As a product with expired core patents, its revenue trajectory typically reflects the dynamics of branded versus generic competition.
Revenue Performance:
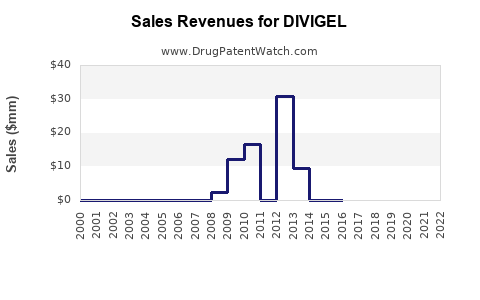
- Peak Sales: Divigel achieved peak annual sales figures in the mid-to-high tens of millions of dollars during its period of patent exclusivity.
- Post-Patent Expiration: Following the expiration of its primary patents, Divigel's sales have experienced a decline due to the introduction of generic competitors. Generic testosterone gels are often priced at a significant discount to the branded product, diverting market share and reducing overall revenue for branded formulations.
- Current Sales: While specific up-to-date sales figures for Divigel are proprietary and vary by reporting period and region, industry analysts estimate its annual revenue to be in the range of $20 million to $40 million globally. This is a substantial reduction from its peak performance.
- Brand Value: Despite revenue decline, the brand may still hold a market share due to physician preference and established patient use. However, this is increasingly challenged by cost-conscious healthcare systems and payers.
Market Share:
- Testosterone Gel Segment: Within the testosterone gel segment, Divigel likely holds a single-digit percentage market share.
- Overall TRT Market: In the broader TRT market, which includes injections, patches, and pellets, Divigel's share is even smaller.
- Dominant Players: AndroGel (AbbVie) and its generic counterparts, along with other established branded and generic TRT products, command larger shares of the overall market.
Factors Affecting Financial Trajectory:
- Generic Pricing: The price of generic testosterone gels is a primary driver of Divigel's declining revenue. Generic products are typically 30-60% cheaper than the branded equivalent, leading to significant shifts in prescription volume.
- Managed Care and Payer Restrictions: Insurance companies and pharmacy benefit managers (PBMs) often implement preferred drug lists that favor lower-cost generics or alternative TRT formulations, limiting Divigel's formulary placement and reimbursement.
- Physician Prescribing Habits: While some physicians may maintain loyalty to Divigel due to familiarity with its performance, the economic incentives increasingly push towards generics.
- Sales and Marketing Investments: AbbVie's historical investment in marketing and physician education for AndroGel, its primary testosterone gel competitor, has also influenced the overall gel segment dynamics.
The financial trajectory for Divigel is characterized by a post-exclusivity decline, typical for branded drugs facing generic competition. Future revenue will likely continue to be modest, primarily derived from brand loyalists and specific market niches where cost is a secondary consideration.
What are the Future Market Opportunities and Challenges?
The future market for Divigel is shaped by ongoing trends in TRT and the persistent impact of generic competition. Opportunities exist in specific patient populations and therapeutic niches, while challenges stem from pricing pressures and evolving treatment paradigms.
Market Opportunities:
- Niche Patient Populations: Divigel may continue to serve specific patient groups who have shown a consistent positive response and tolerability to the formulation, or those with contraindications or sensitivities to other TRT forms.
- Geographic Expansion (Limited): While mature markets are saturated with generics, there may be some remaining opportunities in developing regions with less entrenched generic competition, though market access and infrastructure are significant hurdles.
- Combination Therapies/New Indications (Unlikely): Given its molecule and formulation age, the likelihood of significant investment in developing new indications or combination therapies for Divigel is low.
- Brand Loyalty and Physician Relationships: Strong, long-standing relationships between prescribers and the brand can sustain a baseline level of prescription volume, even in a generic-dominated market.
Market Challenges:
- Pervasive Generic Competition: The primary challenge is the widespread availability of lower-cost generic testosterone gels. This exerts continuous downward pressure on pricing and market share.
- Payer and Formulary Restrictions: Increasing restrictions by insurance companies and PBMs on branded TRTs, favoring generics or alternative delivery methods, significantly limits market access and physician prescribing choice.
- Evolving TRT Landscape: Research into new TRT delivery systems and therapies continues. Advancements in long-acting injectables, oral testosterone formulations, and novel delivery mechanisms could further fragment the market and reduce demand for traditional gels.
- Safety Concerns and Off-Label Use: While TRT is established, ongoing scrutiny regarding cardiovascular risks and other potential side effects associated with testosterone therapy can influence overall market growth and prescribing caution, impacting all products within the class.
- Cost-Containment Pressures: The broader healthcare environment emphasizes cost containment, making it difficult for branded drugs facing generic competition to maintain significant market share or pricing power.
The financial future of Divigel is unlikely to mirror its past growth. Its trajectory will be characterized by a steady erosion of market share and revenue, primarily driven by the economics of generic substitution and evolving healthcare payer policies.
Key Takeaways
- Market Growth Drivers: The hypogonadism treatment market is expanding due to increased awareness, an aging male population, and patient preference for convenient TRT formulations.
- Competitive Landscape: Divigel operates in a highly competitive testosterone gel segment, facing strong competition from branded products like AndroGel and a growing number of generic testosterone gels.
- Patent Expiration: Core patents for Divigel have expired, leading to significant generic competition and a decline in branded revenue.
- Financial Trajectory: Divigel's revenue has declined post-patent expiration, with current global sales estimated between $20 million and $40 million annually, representing a single-digit market share in the testosterone gel segment.
- Future Challenges: Pervasive generic competition, payer restrictions, and evolving TRT landscape pose significant challenges to Divigel's future market performance.
Frequently Asked Questions
-
When did the primary patents for Divigel expire?
The primary patents for Divigel's composition of matter and formulation expired in the United States around 2016-2017, paving the way for generic market entry.
-
What is the approximate market size for testosterone gels globally?
The specific market size for testosterone gels is a segment of the broader TRT market. The overall TRT market was valued at approximately $2.5 billion in 2022, with testosterone gels comprising a substantial portion of this value.
-
How does the pricing of generic testosterone gels impact Divigel's revenue?
Generic testosterone gels are priced significantly lower than branded Divigel, typically 30-60% less. This price differential is a primary driver of Divigel's declining revenue and market share.
-
Are there any new therapeutic indications or formulations for Divigel currently under development?
There is no significant public information indicating that new therapeutic indications or formulations for Divigel are currently under active development by its current marketers, given the age of the molecule and formulation.
-
What is the primary reason for the decline in Divigel's market share?
The primary reason for the decline in Divigel's market share is the expiration of its core patents, which has allowed for the introduction of lower-cost generic testosterone gels into the market.
Cited Sources
[1] Grand View Research. (2023). Hypogonadism Market Size, Share & Trends Analysis Report by Treatment (Testosterone Replacement Therapy, Others), by Therapy Type (Injectable, Transdermal, Others), by Disease Type, by Distribution Channel, and Segment Forecasts, 2023 - 2030.
[2] National Institute on Aging. (2023, April 12). Low Testosterone in Men. Retrieved from https://www.nia.nih.gov/health/low-testosterone-men
[3] MarketsandMarkets. (2023). Testosterone Market - Global Forecast to 2028.
[4] U.S. Food & Drug Administration. (n.d.). Drugs@FDA: FDA-Approved Drugs. Retrieved from https://www.accessdata.fda.gov/scripts/cder/daf/ (Note: Specific generic product approvals can be searched here).