Share This Page
Drug Price Trends for DIVIGEL
✉ Email this page to a colleague
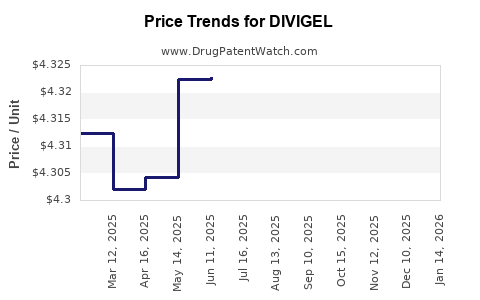
Average Pharmacy Cost for DIVIGEL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DIVIGEL 0.25 MG GEL PACKET | 68025-0065-30 | 5.39489 | EACH | 2026-04-22 |
| DIVIGEL 0.5 MG GEL PACKET | 68025-0066-30 | 5.39910 | EACH | 2026-04-22 |
| DIVIGEL 1 MG GEL PACKET | 68025-0067-30 | 5.38063 | GM | 2026-04-22 |
| DIVIGEL 0.25 MG GEL PACKET | 68025-0065-30 | 5.39967 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for DIVIGEL
What is DIVIGEL?
DIVIGEL is a transdermal estrogen therapy in gel form, containing estradiol, used primarily for hormone replacement therapy (HRT) in menopausal women. It is marketed by Noven Pharmaceuticals since FDA approval in 2014. The gel is designed for systemic hormone absorption via topical application, reducing risks associated with oral estrogen therapy.
Current Market Overview
Market Size
The global menopausal therapy market reached approximately $20 billion in 2022, with estrogen therapies accounting for about 80% of this figure. The U.S. dominates the market, representing roughly 40% of global sales.
Competition Landscape
Key competitors include:
- Estrogen patches: brands like Vivelle-Dot (Novartis), Estraderm (Novartis)
- Oral estrogens: Premarin, Estrace
- Other topical gels: Estradiol gel by progesterone companies, compounded formulations
DIVIGEL's direct competitors are mainly oral and patch-based HRT products. However, its transdermal gel format differentiates it distinctly as an option amid growing preference for non-oral delivery due to lower thrombosis risk.
Market Penetration
Since its launch, DIVIGEL has gained approximately 5-7% of the estrogen therapy segment in the U.S. among prescribed therapies for menopause. Its uptake remains moderate due to competition from established patches and oral formulations.
Regulatory and Reimbursement Environment
- FDA Approvals: Approved for in the U.S. for moderate to severe vasomotor symptoms related to menopause.
- Reimbursement: Covered broadly under Medicare and private insurance, with generic options providing price competition pressure.
Price Trends and Projections
Current Pricing
- DIVIGEL (0.25 mg/0.5 g): Wholesale acquisition cost (WAC) approximately $325 per 30-gram tube.
- Competitor gels: Range from $200 to $350 per month depending on the brand and dosage.
Factors Influencing Price
- Generic Entry: No generic version approved yet, sustaining high prices.
- Market Competition: Arrival of generics or biosimilars could reduce prices.
- Insurance Coverage: Widens access but compresses margins.
- Manufacturing Costs: Stable due to established production processes, but influenced by raw material prices especially estradiol bulk supply.
Price Projection (Next 5 Years)
| Year | Estimated Wholesale Price (Per 30g Tube) | Key Factors |
|---|---|---|
| 2023 | $325 | Stable, limited generic competition |
| 2024 | $310-$330 | Slight price pressure, patent exclusivity persists |
| 2025 | $300-$320 | Potential entry of generics, pricing competition |
| 2026 | $290-$310 | Increased generics, biosimilars entering |
| 2027 | $270-$295 | Market consolidation, cost reductions |
Prices assume no major policy shifts or new formulations.
Price Sensitivity
- The price of DIVIGEL is highly sensitive to generic competition.
- Price declines may be sharper if FDA approves generic estradiol gels.
- Insurance negotiations tend to maintain customer access but can shift profit margins.
Future Market Dynamics
- Growing demand: Older populations and rising menopause incidence will sustain growth.
- Innovation: New delivery systems or combination formulations could disrupt the market.
- Regulatory changes: US and EU policies on hormone therapy safety could modify prescribing patterns.
Key Insights Summary
- DIVIGEL holds a niche in transdermal estrogen therapy with an approximate 5-7% market share.
- Its current price premium stems from patent exclusivity and the absence of generic competition.
- Price projections indicate gradual declines with the potential for sharper drops if generics enter the market.
- Competitive pressure from patches, pills, and compounded therapies shapes fiscal and marketing strategies.
Key Takeaways
- DIVIGEL’s market is sizable but highly competitive, with slow price erosion anticipated.
- The absence of generics supports stable, high pricing through 2023 and possibly into 2024.
- Generic approval or biosimilar entry can halve prices within two to three years.
- The growth of hormone replacement therapy demand sustains revenue, despite competitive threats.
- Regulatory and reimbursement policies could significantly influence future price trends.
FAQs
1. When might generic versions of DIVIGEL become available?
No generic estradiol gel has received FDA approval as of early 2023. Approval timelines depend on patent status, ANDA submissions, and regulatory review processes, typically spanning 1-3 years after patent expiry.
2. How does DIVIGEL compare price-wise to other estrogen therapies?
DIVIGEL is priced higher than many patches and estradiol pills due to its formulation and patent protection. Patches typically cost $200-$250/month, while DIVIGEL averages $325 for a 30g tube.
3. What factors could accelerate price declines?
Approval of generic formulations, biosimilars, or biosimilar combination products. Market entry by competitors and institutional negotiations also contribute.
4. What regulatory changes could influence DIVIGEL’s market?
Stricter safety guidelines or new efficacy data could alter prescriptive patterns. Conversely, approvals of new delivery systems could expand options and reduce reliance on existing formulations.
5. What is the outlook for revenue growth?
Continued growth driven by aging populations and menopause prevalence supports revenue; however, price reductions due to generics are expected to offset volume gains over time.
References
- US Food and Drug Administration. (2014). Noven Pharmaceuticals receives FDA approval for DIVIGEL.
- MarketWatch. (2022). Global Menopausal Therapy Market size, share, trends.
- EvaluatePharma. (2022). Estrogen therapies market outlook.
- Noven Pharmaceuticals. (2023). DIVIGEL product information.
- IQVIA. (2022). US prescription drug sales figures.
*[1] FDA. (2014). Approval of DIVIGEL.***
More… ↓
