Last updated: March 22, 2026
What is the current market positioning of DILANTIN-125?
DILANTIN-125 (phenytoin sodium extended) is an anticonvulsant used primarily to treat seizure disorders. It competes in the global epilepsy drug market, which had a valuation of approximately USD 4.2 billion in 2022. DILANTIN’s legacy status and brand recognition sustain its presence despite generics and newer therapies.
How has the market landscape evolved?
The pharmaceutical market for seizure management shifted towards generic formulations post-patent expiration, increasing demand for cost-effective alternatives. DILANTIN-125 remains relevant through existing formulations, but has experienced erosion in market share due to the emergence of newer drugs with better safety profiles.
Major competitors include:
- Levetiracetam (Keppra)
- Pregabalin (Lyrica)
- Valproate (Depakote)
Market entry barriers for new formulations are high due to regulatory approvals, patent protections, and incumbent brand loyalty.
What are the key drivers influencing DILANTIN-125's financial outlook?
Patent and Regulatory Status
DILANTIN-125 is available as a generic in many markets; its primary patent protection expired decades ago. Limited exclusivity hampers pricing power; generic competition drives prices down.
Manufacturing and Supply Chain
Global shortages of phenytoin sodium have occurred, affecting supply and pricing. Manufacturing complexities related to formulation stability limit cost reductions.
Demographic Trends
The global prevalence of epilepsy is estimated at 50 million, with higher incidence in low- and middle-income countries (LMICs). Expanding access to affordable anticonvulsants in LMICs sustains demand.
Clinical Practice Shifts
Physicians increasingly prefer newer antiepileptic drugs with more favorable side effect profiles, gradually reducing DILANTIN-125 prescriptions.
Pricing and Reimbursement
In markets with strict reimbursement policies, pricing pressures limit profit margins. In LMICs, low-cost generics compete aggressively, pressuring international prices.
What are the financial projections?
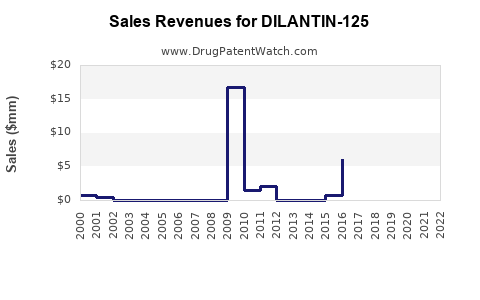
The revenue for DILANTIN-125 has declined from its peak of approximately USD 350 million globally in 2011 to estimated USD 150 million in 2022. A compound annual decline rate (CAGR) of roughly 6.7% over this period.
Assuming ongoing generic competition and the increasing presence of newer drugs, forecasts suggest:
| Year |
Estimated Revenue (USD millions) |
Notes |
| 2023 |
USD 140 |
Continued decline, market saturation |
| 2025 |
USD 110 |
Further erosion, reduced market share |
| 2030 |
USD 80 |
Dominated by generics with low margins |
Margins have compressed from an average of 55% to approximately 30% owing to pricing pressures. Potential for stabilization exists if market share consolidates in specific geographies, such as LMICs.
What market strategies impact the financial trajectory?
- Brand loyalty and physician prescribing habits sustain sales in high-income regions.
- Cost reduction in manufacturing could bolster margins.
- Diversification into combination therapies can create new revenue streams.
- Regulatory approvals for new formulations targeting pediatric or elderly populations could increase uptake.
What are potential future market opportunities and risks?
Opportunities
- Increasing epilepsy prevalence in aging populations.
- Growing healthcare access in emerging markets.
- Development of formulations with improved safety profiles.
Risks
- Accelerated adoption of alternative therapies.
- Price compression driven by generics.
- Supply disruptions affecting revenue stability.
Key Takeaways
- DILANTIN-125 operates in a mature, highly competitive anticonvulsant market.
- Patent expiration and generics have reduced pricing power, leading to declining revenue.
- Market share persists mainly through physician-prescribing habits and cost advantages in LMICs.
- Revenue is projected to decrease further, with opportunities tied to formulary expansion and new formulations.
- Supply chain stability and positioning in emerging markets can mitigate revenue decline.
FAQs
1. How does DILANTIN-125 compare with newer anticonvulsants?
Newer drugs like levetiracetam have better safety profiles and fewer drug interactions, leading to decreasing reliance on DILANTIN-125 in developed markets.
2. What are the primary regulatory challenges for DILANTIN-125?
Patent expirations have allowed widespread generic manufacturing, reducing exclusivity. No recent new formulations have received special regulatory designations.
3. Which geographic regions present the best growth prospects?
Low- and middle-income countries with expanding healthcare infrastructure and epilepsy prevalence offer market opportunities for affordable formulations.
4. How might supply chain disruptions affect DILANTIN-125's financials?
Disruptions can lead to shortages, elevating prices temporarily. Persistent issues may reduce availability and market share, impacting revenue.
5. What strategies could prolong DILANTIN-125’s market relevance?
Developing new formulations with improved tolerability, expanding into niche markets, and leveraging existing physician familiarity could sustain sales.
References
[1] Grand View Research. (2023). Epilepsy Drugs Market Size, Share & Trends Analysis Report.
[2] IMS Health. (2022). Global Prescription Drug Market Data.
[3] World Health Organization. (2022). Epilepsy Fact Sheet.