Last updated: February 19, 2026
What is the Current Market Size and Growth Rate?
Dihydroergotamine mesylate (DHE) is a serotonin receptor agonist primarily used to treat migraines and cluster headaches. The global market for migraine drugs, including DHE, is valued at approximately $4.2 billion as of 2022. The compound's market share within this segment remains significant due to its efficacy in acute migraine management.
The compound is marketed mainly by Pfizer under the brand DHE 45 and by other generic producers. The market is projected to grow at a Compound Annual Growth Rate (CAGR) of about 4.3% from 2023 to 2030, driven by increasing awareness and migraine prevalence.
What Are Key Market Drivers?
-
Growing Prevalence of Migraines: Annually, around 1 billion people worldwide experience migraines, with a substantial portion seeking acute treatment. The rising prevalence accelerates demand for effective therapies like DHE.
-
Limited Alternative Treatments: While triptans dominate the migraine market, some patient groups have contraindications or inadequate responses. DHE remains a vital alternative for intractable cases or those contraindicated for triptans.
-
Regulatory Endorsements: Certain health authorities recognize DHE's efficacy, which encourages formulary inclusion and prescription frequency, thus stabilizing its demand.
-
Formulation Innovations: Development of new delivery methods, such as nasal sprays and auto-injectors, improves accessibility and patient compliance, contributing to market expansion.
What Challenges Affect Market and Financial Outlook?
-
Generic Competition: Entry of lower-cost generics post patent expiration depresses prices and reduces profitability for branded formulations.
-
Limited Patent Protections: DHE lacks recent patent protection, leading to price erosion and intensified generic target competition.
-
Pricing Pressures: Healthcare systems worldwide face cost containment measures, pressuring prices for DHE formulations.
-
Alternative Therapies: The advent of new migraine drugs, including CGRP antagonists (e.g., erenumab), capture some market share, especially for prophylactic use, indirectly affecting DHE's revenue stream.
What Are the Revenue Trends and Profitabilities?
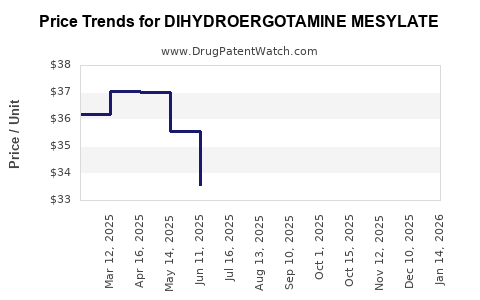
Pfizer's sales of DHE formulations have remained steady, with approximate revenues of $150 million in 2022. Generic manufacturers report margins of 25-30% on DHE products, but overall revenues are declining due to volume pressures.
Projections indicate that unless innovational formulations or new indications emerge, revenues could decrease by 10-15% annually over the next five years post-major generic entry.
How Does the Regulatory Environment Impact Financial Trajectory?
Regulatory authorities in the U.S. (FDA) and Europe (EMA) approve formulations primarily for acute migraine management. Limited indications restrict potential revenue expansion.
However, off-label uses and new delivery system approvals could open additional revenue streams. For instance, nasal spray formulations gain FDA approval, potentially increasing patient accessibility and sales.
What Are Key Strategic Opportunities?
-
Formulation Innovation: Developing new, patient-friendly delivery methods can expand market share.
-
New Indications: Exploring potential use in other vasospastic or neurological conditions might diversify revenue.
-
Combination Therapies: Pairing DHE with other agents could improve efficacy and create premium offerings.
-
Generic Differentiation: Patent-protected differentiations or proprietary formulations can mitigate generic price erosion.
What Is the Future Financial Outlook?
DHE's revenue is expected to decline gradually unless strategic innovations or expanding indications are pursued. Market participants with proprietary formulations or delivery systems could outpace generic competitors and sustain topline growth.
Summary Table: Dihydroergotamine Mesylate Market Metrics
| Aspect |
Key Details |
| Market Size (2022) |
$4.2 billion globally |
| CAGR (2023–2030) |
4.3% |
| Major Players |
Pfizer, Teva, Sandoz, Mylan |
| Revenue (2022) |
~$150 million (Pfizer) |
| Patent Status |
No recent patents; generic competition present |
| Pricing Margin (Generics) |
25–30% |
Key Takeaways
- The DHE market is stable but faces long-term decline pressures due to generics and emerging therapies.
- Market growth depends on innovations in delivery and expanded indications.
- Pricing pressures and generic entry impact profitability; strategic differentiation remains crucial.
- The sizeable migraine treatment market offers ongoing revenue opportunities but requires adaptation.
- Companies investing in formulation development or new patent protections can mitigate revenue erosion.
FAQs
Q1: How long has Dihydroergotamine mesylate been on the market?
A: Since the 1940s, with formulations approved for acute migraine treatment in the late 20th century.
Q2: What are the main competitors to DHE?
A: Triptans like sumatriptan, and newer CGRP antagonists such as erenumab, fremanezumab.
Q3: Can DHE be used for prophylactic migraine treatment?
A: No, it is primarily an acute treatment. Prophylaxis involves other medication classes.
Q4: What delivery forms are available for DHE?
A: Intravenous, intranasal spray, and autoinjector formulations.
Q5: Are there ongoing clinical developments related to DHE?
A: Research focuses on improving delivery methods and exploring additional neurological indications.
References
- MarketWatch. (2022). Global migraine drugs market size and forecast.
- Pfizer Inc. (2022). Annual report on marketed drugs.
- Grand View Research. (2022). Migraine therapeutics market analysis.
- U.S. Food and Drug Administration. (2020). Guidance on migraine treatment formulations.
- European Medicines Agency. (2021). Dihydroergotamine nasal spray approval.