Last updated: April 25, 2026
What is DEPO-MEDROL in market terms?
DEPO-MEDROL is a branded corticosteroid product whose active ingredient is methylprednisolone acetate. It is marketed in the US as an injectable suspension for indications that include inflammatory and rheumatologic conditions (among others). Pricing, demand, and competitive pressure for DEPO-MEDROL track the broader US injectable steroid market, with added sensitivity to payer reimbursement and generic substitution.
How has the competitive landscape shaped pricing and volume?
DEPO-MEDROL’s financial trajectory is dominated by branded-versus-generic dynamics common to mature steroid injectables:
- Generic erosion risk: Methylprednisolone acetate injection products exist as multisource generics/authorized competitors in many markets once brand exclusivity ends. That typically compresses net pricing and shifts demand toward lower acquisition cost.
- Substitutability: The drug class is relatively substitutable for many use cases. When prescribers treat “a steroid injection” rather than a branded molecule-brand product, tendering and pharmacy benefit design favor generics.
- Channel mix: In hospital and clinic settings, formularies and group purchasing can drive faster switching than in ambulatory retail, accelerating branded volume declines after formulary loss.
What do historical prescribing and reimbursement patterns imply for DEPO-MEDROL revenue?
For established branded corticosteroids, revenue trajectories usually show three phases:
- Launch/build-out phase: Growth led by brand awareness and reimbursement acceptance.
- Maturity phase: Stable demand with gradual net price pressure.
- Post-exclusivity phase: Faster erosion of net price and branded volume as generics gain share; residual brand demand persists in pockets tied to specific billing history, device handling preferences, or supplier arrangements.
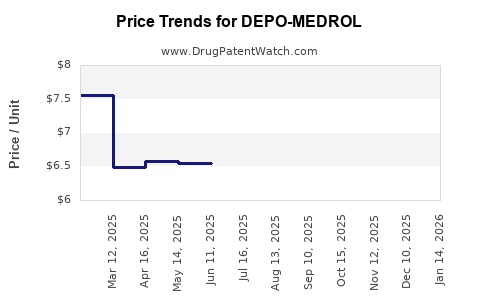
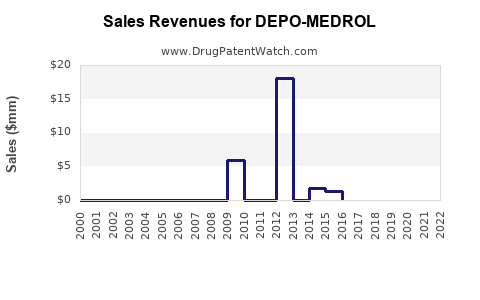
For DEPO-MEDROL, the most investable lens is the net sales versus net price mix shift: as generic availability expands, branded revenue declines often outpace pure volume changes because discounts and payer pressure rise while share falls.
Where does demand concentrate across settings?
DEPO-MEDROL is an injectable product, which concentrates demand in settings that include:
- Hospital outpatient: Higher sensitivity to contracting and formulary.
- Hospital inpatient: Use is often driven by clinical protocols and procurement contracts.
- Specialty clinics: Maintains steadier brand preference when treatment pathways standardize product choice.
Because institutional purchasing can switch quickly once alternatives are favored, DEPO-MEDROL’s market dynamics are tied to contract wins and formulary status.
What market dynamics affect supply and continuity?
Injectable steroid markets typically face demand volatility that is less about “therapeutic trend” and more about:
- Procurement timing: Contract cycles and inventory planning.
- Regulatory and manufacturing execution: Sterile injectables can be impacted by batch release schedules.
- Distribution incentives: Short-term promotions and stocking policies affect near-term shipment patterns.
For DEPO-MEDROL, any sustained revenue path depends on stable supply and consistent contracting, not on novel uptake.
How do financial trajectories generally present for mature steroid injectables?
A mature branded corticosteroid’s financial trajectory usually shows:
- Net sales decline as generics take incremental share.
- Net price erosion as rebates expand and acquisition cost comparisons force price reductions.
- Flatter operating leverage: gross margin can compress even when cost stays controlled, unless the company shifts mix, reduces commercial spend, or exits low-margin channels.
What is the revenue and profitability sensitivity to generic substitution?
DEPO-MEDROL’s class-level substitutability implies high sensitivity to:
- Payer formulary placement (preferred generic tiers)
- Administrative switching (pharmacy benefit edits that drive automatic substitution)
- Tender outcomes (institutional purchasing that awards lower-cost bids)
If branded product loses “preferred” status, the near-term effect is often a steep branded share decline, followed by a slower stabilization at a reduced baseline demand level.
Key market indicators to track for DEPO-MEDROL
For an investable monitor set, track the indicators below, because they typically precede changes in branded net sales:
| Indicator |
What it predicts |
Why it matters for DEPO-MEDROL |
| Formulary status changes |
Share shift |
Determines whether institutions can buy branded vs generic |
| Net price trends |
Margin compression |
Mature brands face discount pressure and rebate escalation |
| Generic competitor assortment |
Incremental substitution |
Multi-source availability increases switching velocity |
| Contract award cycle outcomes |
Shipment volatility |
Institutional purchasing drives short-cycle demand changes |
| Inventory/distribution pattern |
Temporary sales spikes or dips |
Sterile injectables show timing effects around replenishment |
Financial trajectory: expected shape (market-structure-driven)
DEPO-MEDROL’s financial trajectory is best characterized as structurally constrained by generic competition rather than by new clinical uptake curves.
Expected trajectory by phase:
- Downward slope in branded net sales after exclusivity periods, with declines driven by both volume erosion and net price compression.
- Margin pressure driven by higher rebates, channel mix deterioration, and competitive pricing.
- Stabilization at a smaller brand base where legacy prescribing patterns or contract specificity maintain minimal residual demand.
Decision-grade implications for R&D and investment
For investors
- Expect limited upside without a clear branded differentiation lever (device, formulation advantage, patient support program, or special contracting).
- Value analysis should emphasize cash yield from remaining branded base and risk-adjusted downside from further formulary tightening or additional multisource entry.
For product strategists
- If DEPO-MEDROL remains in scope for lifecycle management, the primary defense is commercial access: contracting strategy, payer negotiations, and supply assurance.
- R&D investment should be directed toward areas with defensible differentiation, because class-level substitutability limits brand resilience.
What does “market dynamics” mean for commercialization choices?
Commercial decisions for DEPO-MEDROL in mature conditions typically revolve around:
- Institutional contracting strategy (hospital systems and group purchasing organizations)
- Payer rebate engineering (maintain access in preferred tiers where possible)
- Distribution planning (avoid stockouts that can permanently shift prescribers to substitutes)
- Supply reliability (sterile injectables need stable manufacturing cadence)
Key Takeaways
- DEPO-MEDROL’s market dynamics are structurally shaped by generic substitution and institutional contracting, not by new therapeutic expansion.
- The financial trajectory is expected to show branded net sales decline via both volume erosion and net price compression, followed by stabilization at lower baseline demand.
- The most predictive leading indicators for trajectory changes are formulary status, net price, contract outcomes, and generic assortment availability.
- Commercial defensibility depends on continued institutional access and supply continuity, while profitability remains sensitive to rebate and discount pressure.
FAQs
-
Is DEPO-MEDROL primarily exposed to unit volume decline or net price erosion?
Both, but the dominant effect in mature injectables is typically net price erosion from payer discounts and rebate intensity once generics are entrenched.
-
What contracting events most affect DEPO-MEDROL performance?
Hospital formulary approvals/rejections, group purchasing organization awards, and preferred tier changes that force or incentivize substitution.
-
Does patient switching happen quickly for injectable steroids?
It can. When institutional protocols standardize on a non-branded alternative, switching can occur rapidly during procurement cycles.
-
What market signal best indicates future branded sales pressure?
Moves in formulary placement and net pricing behavior (rebates and discounts rising faster than volumes) are the most direct signals.
-
What would reverse DEPO-MEDROL’s negative trajectory?
A defensible access lever such as special contracting that keeps branded supply preferred, or a differentiation that reduces substitutability.
References
[1] DailyMed. “DEPO-MEDROL (methylprednisolone acetate) Injection.” U.S. National Library of Medicine.
[2] FDA Orange Book. “Methylprednisolone acetate injection” listings for approved products and application numbers.