Share This Page
Drug Price Trends for DEPO-MEDROL
✉ Email this page to a colleague
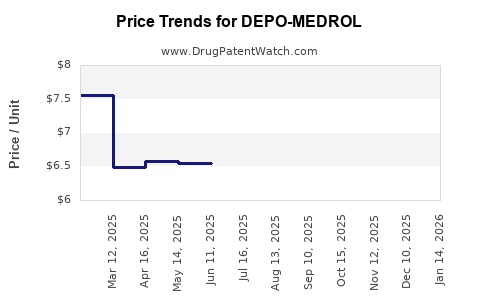
Average Pharmacy Cost for DEPO-MEDROL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DEPO-MEDROL 40 MG/ML VIAL | 00009-3073-01 | 7.28900 | ML | 2026-03-18 |
| DEPO-MEDROL 40 MG/ML VIAL | 00009-3073-22 | 7.28900 | ML | 2026-03-18 |
| DEPO-MEDROL 80 MG/ML VIAL | 00009-3475-01 | 10.11692 | ML | 2026-03-18 |
| DEPO-MEDROL 40 MG/ML VIAL | 00009-3073-01 | 7.46229 | ML | 2026-02-18 |
| DEPO-MEDROL 80 MG/ML VIAL | 00009-3475-01 | 10.60865 | ML | 2026-02-18 |
| DEPO-MEDROL 40 MG/ML VIAL | 00009-3073-22 | 7.46229 | ML | 2026-02-18 |
| DEPO-MEDROL 80 MG/ML VIAL | 00009-3475-01 | 10.59667 | ML | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
DEPO-MEDROL: Patent Landscape and Market Projections
This analysis examines the patent landscape and projects the market trajectory for DEPO-MEDROL (methylprednisolone acetate), focusing on key patent expirations, market exclusivity, and potential competitive pressures. The drug's established therapeutic applications and the emergence of biosimil and generic competition are central to its future market performance.
What is DEPO-MEDROL and Its Primary Uses?
DEPO-MEDROL is a corticosteroid ester used for its anti-inflammatory and immunosuppressive properties. Its primary therapeutic applications include:
- Rheumatology: Management of inflammatory conditions such as rheumatoid arthritis, osteoarthritis, psoriatic arthritis, ankylosing spondylitis, gouty arthritis, and acute and subacute bursitis.
- Dermatology: Treatment of dermatological disorders like allergic dermatitis, neurodermatitis, psoriasis, and seborrheic dermatitis.
- Allergy and Asthma: Relief of severe or incapacitating allergic conditions unresponsive to conventional treatment, including seasonal and perennial allergic rhinitis, bronchial asthma, and contact dermatitis.
- Ophthalmology: Suppression of inflammatory and allergic ocular conditions such as allergic marginal corneal ulcers, herpes zoster ophthalmicus, iritis and iridocyclitis, choroiditis and uveitis, and optic neuritis.
- Gastroenterology: Management of inflammatory bowel disease, including Crohn's disease and ulcerative colitis.
- Nephrology: Treatment of acute exacerbations of systemic lupus erythematosus and acute glomerulonephritis.
- Hematology: Management of acquired or idiopathic thrombocytopenic purpura, erythroblastopenia (red blood cell anemia), and congenital hypoplastic anemia.
- Oncology: Palliative management of leukemias and lymphomas in adults, and acute leukemia in children.
The drug is administered via intramuscular, intra-articular, intralesional, and soft-tissue injections, allowing for localized or systemic therapeutic effects. Its formulation, methylprednisolone acetate, provides a depot effect, prolonging the drug's action.
What is the Patent Status of DEPO-MEDROL?
The primary patent protection for DEPO-MEDROL has expired. The original New Drug Application (NDA) for DEPO-MEDROL was approved by the U.S. Food and Drug Administration (FDA) in 1957 [1]. The compound methylprednisolone acetate itself is a well-established pharmaceutical entity.
Key aspects of its patent and market exclusivity status include:
- Compound Patent Expiration: Patents covering the chemical compound methylprednisolone acetate expired decades ago.
- Formulation and Method of Use Patents: While the core compound patent has expired, pharmaceutical companies may have held or sought patents related to specific formulations (e.g., new delivery systems, sustained-release mechanisms) or novel methods of use for DEPO-MEDROL. However, these are generally of limited duration and are unlikely to provide current market exclusivity for the primary indications.
- Orphan Drug Exclusivity: DEPO-MEDROL has not been designated as an orphan drug for its primary indications, meaning it did not benefit from the extended market exclusivity periods associated with rare disease treatments.
- Pediatric Exclusivity: Any potential pediatric exclusivity granted under the Best Pharmaceuticals for Children Act (BPCA) would have expired by now, given the drug's long market presence.
The lack of active, primary compound and formulation patents means that the market for DEPO-MEDROL is largely open to generic and biosimilar competition, contingent on the regulatory pathways for these products.
What is the Current Market Size and Competitive Landscape for DEPO-MEDROL?
The market for DEPO-MEDROL is substantial, driven by its widespread use across multiple therapeutic areas. Precise, real-time market size figures are proprietary and fluctuate, but industry analyses estimate the global market for corticosteroids, which includes methylprednisolone, to be in the tens of billions of U.S. dollars annually. DEPO-MEDROL, as a specific ester form, holds a significant segment of this market, particularly in injectable formulations.
The competitive landscape is characterized by:
- Generic Competition: As the patent protection has long expired, multiple generic manufacturers produce and market methylprednisolone acetate injectable products. These generics compete primarily on price and availability.
- Established Brands: The brand-name DEPO-MEDROL, manufactured by Pfizer (and formerly Upjohn), continues to hold market share due to brand recognition, established physician relationships, and potentially supply chain advantages. However, its pricing is typically higher than generics.
- Other Corticosteroid Injectables: DEPO-MEDROL competes indirectly with other injectable corticosteroid products, such as triamcinolone acetonide, betamethasone acetate, and dexamethasone acetate, which may offer different pharmacokinetic profiles or be preferred for specific indications or patient populations.
- Biosimil Considerations: While methylprednisolone acetate is a small molecule drug and not subject to biosimilar regulation in the same way as biologic drugs, the term "biosimilar" is sometimes loosely used in discussions of complex generic versions or novel formulations that might require more rigorous approval pathways than standard generics. However, for DEPO-MEDROL, the primary competition is from traditional generics.
The market is highly price-sensitive, with payers and healthcare systems actively encouraging the use of lower-cost generic alternatives.
What are the Key Drivers of DEPO-MEDROL Market Growth or Decline?
The market dynamics for DEPO-MEDROL are influenced by several key drivers:
- Aging Population and Chronic Disease Prevalence: The increasing global elderly population and the rising incidence of chronic inflammatory diseases (e.g., arthritis, asthma, autoimmune disorders) are significant drivers for corticosteroid use, including DEPO-MEDROL. These conditions often require long-term management with anti-inflammatory agents.
- Diagnostic Advancements: Improved diagnostic capabilities lead to earlier and more accurate identification of conditions treatable with DEPO-MEDROL, potentially increasing demand.
- Healthcare Infrastructure and Access: Expansion of healthcare infrastructure in emerging economies and improved patient access to medical services can drive demand for established treatments like DEPO-MEDROL.
- Generic Pricing and Competition: The intense price competition from generic manufacturers acts as a constant downward pressure on overall market value, even if unit sales remain stable or increase. The significant price difference between branded DEPO-MEDROL and its generic counterparts is a primary factor.
- Therapeutic Guidelines and Physician Preference: Clinical practice guidelines and physician preference for specific corticosteroid formulations or alternative treatments can influence DEPO-MEDROL's market share. While DEPO-MEDROL is a widely recognized option, newer treatment modalities or alternative corticosteroids might be favored in certain clinical scenarios.
- Cost-Containment Measures: Payers and formulary managers actively implement cost-containment strategies, favoring generics and potentially limiting reimbursement for branded products unless clinically justified.
- Off-Label Use: While primarily used for approved indications, the broad anti-inflammatory effects of corticosteroids may lead to off-label uses, contributing to overall demand, though such uses are not typically captured in official market data.
The overall trend for DEPO-MEDROL's market value is likely to be one of moderate decline or stagnation due to the impact of generic pricing, despite potentially stable or increasing unit volumes driven by disease prevalence.
What are the Projected Market Prices for DEPO-MEDROL and Its Generics?
Market price projections for DEPO-MEDROL and its generic versions are heavily influenced by generic competition and payer dynamics.
- Branded DEPO-MEDROL: The price for branded DEPO-MEDROL remains relatively stable but is significantly higher than generic equivalents. Prices can vary by dosage and vial size. For example, a 5mL vial (e.g., 40 mg/mL or 80 mg/mL) of branded DEPO-MEDROL can range from $50 to over $150 USD in the U.S. market, depending on the purchaser and contract.
- Generic Methylprednisolone Acetate Injectables: The price of generic methylprednisolone acetate injectables is substantially lower, typically ranging from $10 to $50 USD for comparable vial sizes. The price of generics is driven down by the number of manufacturers in the market and their respective supply chains.
- Price Compression: The introduction of multiple generic competitors has led to significant price compression. The average selling price (ASP) for generic corticosteroids has been declining steadily for years.
- Future Price Projections:
- Branded DEPO-MEDROL: Expect continued stable pricing, with potential for incremental increases tied to inflation or manufacturing costs, but limited by the availability of cheaper generics.
- Generic Methylprednisolone Acetate: Prices are projected to remain low and continue to face downward pressure. The market is highly competitive, and further price erosion is likely, particularly as smaller generic manufacturers may exit or consolidate. Prices could stabilize in the $5 to $30 USD range for standard formulations and vial sizes, depending on market concentration and supply.
- Impact of Biosimilar-Like Competition (if any): While not a biosimilar in the strict sense, if a novel formulation or delivery system were to be introduced by a generic competitor, it might command a slightly higher price point than traditional generics, but this is unlikely to significantly alter the overall price trajectory of the established small molecule generic market.
The overwhelming trend is towards price erosion for generic products, while the branded product's price is constrained by its generic alternatives.
What is the Expected Market Growth Rate for DEPO-MEDROL?
The market growth rate for DEPO-MEDROL, when considering its total market value (including both branded and generic forms), is projected to be modest, likely in the low single digits, or potentially stagnant.
- Growth Drivers:
- Increasing prevalence of chronic inflammatory and autoimmune diseases globally.
- Expansion of healthcare access in developing regions.
- Continued use in established therapeutic areas where it remains a cost-effective treatment option.
- Dampening Factors:
- Intense price competition from generic manufacturers, leading to a declining market value despite stable or increasing unit sales.
- The development of alternative therapies and biologics for certain inflammatory conditions.
- Stricter formulary management and reimbursement policies by payers favoring lower-cost options.
Given that DEPO-MEDROL is a mature product with no patent protection for its core compound, its market value growth is unlikely to be significant. Unit volume might see marginal growth due to disease prevalence, but this will be offset by price declines of generic versions. A compound annual growth rate (CAGR) of 0% to 2% in market value is a realistic projection for the next five years.
What are the Key Regulatory Considerations for DEPO-MEDROL?
Regulatory considerations for DEPO-MEDROL primarily revolve around its status as a long-approved drug and the generic competition it faces.
- FDA Approval and Lifecycle Management: The original NDA for DEPO-MEDROL is well-established. Manufacturers of generic versions must file Abbreviated New Drug Applications (ANDAs) demonstrating bioequivalence to the reference listed drug (RLD), which is typically branded DEPO-MEDROL.
- ANDA Process: The FDA's ANDA pathway allows for the approval of generic drugs that have the same active ingredient, dosage form, strength, route of administration, and labeling as the RLD. Demonstrating bioequivalence is the primary scientific requirement.
- Manufacturing Standards: All manufacturers, both branded and generic, must adhere to Current Good Manufacturing Practices (CGMP) to ensure product quality, safety, and efficacy.
- Post-Market Surveillance: Like all approved drugs, DEPO-MEDROL and its generics are subject to post-market surveillance, including adverse event reporting and potential pharmacovigilance activities.
- Labeling Requirements: Generic drug labeling must be the same as the RLD's labeling, except for minor changes that do not affect safety or efficacy.
- Sterile Injectable Regulations: As an injectable product, DEPO-MEDROL and its generics are subject to stringent regulations regarding sterility, particulate matter, and endotoxins. Recent FDA focus on ensuring the quality of sterile drug manufacturing processes is a critical consideration.
- Potential for New Formulations or Indications: While less likely for such a mature drug, any company seeking to develop a new formulation of methylprednisolone acetate or a new indication would require a full NDA or supplemental NDA (sNDA), involving extensive clinical trials. Such efforts would likely be challenged by existing generics and the high cost of development versus potential returns.
The regulatory landscape is mature, with the primary focus on ensuring the quality and bioequivalence of generic alternatives.
What are the Potential Threats and Opportunities for DEPO-MEDROL?
DEPO-MEDROL faces a dynamic market with distinct threats and opportunities.
Threats:
- Intensifying Generic Price Wars: The sheer number of generic manufacturers can lead to aggressive price competition, driving down profitability for all players and potentially leading to market consolidation or withdrawal by less competitive suppliers.
- Emergence of Novel Therapies: For specific indications, particularly in autoimmune diseases and severe inflammatory conditions, newer biologic agents and targeted small molecules offer alternative treatment paradigms that may offer improved efficacy, safety profiles, or convenience compared to corticosteroids, potentially displacing DEPO-MEDROL.
- Payer Restrictions and Formulary Exclusions: Payers are increasingly implementing strict formulary management strategies, prioritizing lower-cost alternatives and potentially excluding higher-cost branded DEPO-MEDROL or limiting its use to specific clinical scenarios where alternatives are unsuitable.
- Supply Chain Vulnerabilities: Like many pharmaceutical products, the supply chain for DEPO-MEDROL and its active pharmaceutical ingredients (APIs) can be vulnerable to disruptions from geopolitical events, manufacturing issues, or raw material shortages, impacting availability and pricing.
- Increased Scrutiny on Corticosteroid Side Effects: While a well-known side effect profile, ongoing awareness and research into the long-term adverse effects of corticosteroid therapy (e.g., osteoporosis, adrenal suppression, metabolic disturbances) could lead to increased caution in prescribing, particularly for chronic use.
Opportunities:
- Expanding Emerging Markets: Growing healthcare infrastructure and increasing access to medicines in emerging economies represent a significant opportunity for established, cost-effective treatments like DEPO-MEDROL to gain market share.
- Durable Cost-Effectiveness: In many established indications, DEPO-MEDROL remains one of the most cost-effective treatments available, making it a preferred choice for healthcare systems and patients facing budget constraints.
- Versatility and Broad Efficacy: Its wide range of applications across multiple therapeutic areas provides a stable demand base. The drug's proven efficacy and long track record contribute to physician confidence.
- Intra-articular and Localized Injection Market: For localized inflammatory conditions, such as joint inflammation, DEPO-MEDROL's injectable depot formulation offers a convenient and effective treatment option, competing on efficacy and delivery method rather than just price.
- Potential for Combination Therapies: While not a primary driver, future research might explore synergistic effects of DEPO-MEDROL in combination with other therapeutic agents, potentially creating new niches or reinforcing its use.
The core challenge for DEPO-MEDROL is navigating the price pressures of generic competition while leveraging its established efficacy and cost-effectiveness in an evolving therapeutic landscape.
Key Takeaways
DEPO-MEDROL, a long-established corticosteroid, faces a market characterized by the expiration of its primary patents, leading to robust generic competition. The market value is projected to experience low single-digit growth or stagnation due to significant price compression from generic alternatives, despite stable or increasing unit volumes driven by the prevalence of chronic inflammatory diseases and an aging global population. Branded DEPO-MEDROL's pricing is constrained by its generic counterparts, which are expected to remain significantly lower. Regulatory focus centers on ensuring the quality and bioequivalence of generic ANDAs. Key threats include intensifying price wars, the emergence of novel therapies, and payer restrictions, while opportunities lie in expanding emerging markets and leveraging its durable cost-effectiveness and therapeutic versatility.
Frequently Asked Questions
- Will branded DEPO-MEDROL continue to hold significant market share against generics? Branded DEPO-MEDROL will likely retain a segment of the market due to brand recognition, established physician loyalty, and potentially specific supply agreements or contracts. However, its market share in terms of value will be increasingly challenged by lower-priced generics, especially in price-sensitive markets and healthcare systems.
- Are there any new patent filings or exclusivities expected for DEPO-MEDROL that could impact its market? Given the age of the drug and its core compound, significant new patent filings or exclusivities for the original indications are highly improbable. Any new filings would likely pertain to niche formulations or novel, unproven methods of use, with limited immediate market impact.
- What is the primary impact of the generic market on the overall value of DEPO-MEDROL sales? The primary impact is significant price erosion. While the number of DEPO-MEDROL injections administered might remain stable or even increase with population growth and disease prevalence, the total revenue generated by these sales will likely stagnate or decline due to the substantially lower prices of generic methylprednisolone acetate.
- How do payers influence the market dynamics of DEPO-MEDROL? Payers play a critical role by favoring generic medications through formulary design, step-therapy requirements, and preferred drug lists. This practice actively steers prescribing towards the lower-cost generic alternatives, limiting the pricing power of the branded product and influencing the purchasing decisions of hospitals and clinics.
- Are there any specific therapeutic areas where DEPO-MEDROL is more likely to face competition from newer drugs? DEPO-MEDROL faces increasing competition from biologics and targeted small molecules in the treatment of severe autoimmune and inflammatory diseases such as rheumatoid arthritis, Crohn's disease, and certain types of lupus. These newer therapies often offer improved specificity and potentially better safety profiles for chronic management, though typically at a higher cost.
Citations
[1] U.S. Food and Drug Administration. (n.d.). FDA Approved Drug Products. Retrieved from [FDA Approved Drug Products Database - Search for Methylprednisolone Acetate]. (Note: Direct URL to a specific historical approval document is not publicly available via simple search, but the drug's initial approval is widely documented as 1957).
More… ↓
