Last updated: January 5, 2026
Executive Summary
DAYPRO, an analgesic and anti-inflammatory drug primarily used for managing osteoarthritis and related musculoskeletal conditions, has witnessed a fluctuating market landscape driven by evolving regulatory policies, competitive pressures, and demographic shifts. This analysis explores the drug’s market environment, sales trends, regulatory landscape, and strategic positioning, providing stakeholders with a comprehensive outlook on its financial trajectory.
What is DAYPRO and Its Therapeutic Profile?
DAYPRO (Choline salicylate) is an NSAID (non-steroidal anti-inflammatory drug) used predominantly for mild to moderate pain relief and inflammation reduction. Its key attributes include:
| Attribute |
Details |
| Indications |
Osteoarthritis, musculoskeletal pain |
| Administration |
Oral, chewable tablets |
| Approved Markets |
US, EU, and select Asian markets |
| Patent Status |
Generic versions widely available |
| Brand Status |
Initially branded; now largely generic |
Market Dynamics: Key Factors and Trends
1. Market Size and Growth
The global NSAID market was valued at approximately USD 13.5 billion in 2021, with projections reaching USD 18 billion by 2028, growing at a CAGR of 4.6%. DAYPRO's niche in osteoarthritis treatment is embedded within this broader market.
| Region |
2022 Market Share |
CAGR (2022-2028) |
Key Drivers |
| North America |
40% |
4.2% |
Aging population, OTC growth |
| Europe |
28% |
4.5% |
Elderly demographics, prescribing trends |
| Asia-Pacific |
20% |
5.0% |
Rising healthcare access, urbanization |
| Rest of World |
12% |
4.8% |
Increasing awareness |
2. Competitive Landscape
DAYPRO faces broad competition from both branded (e.g., NSAID formulations from Bayer, Pfizer) and generic NSAIDs like ibuprofen, naproxen, and diclofenac. The generic spectrum exerts downward pressure on pricing and margins.
| Competitors |
Market Share (Estimated) |
Strengths |
| Ibuprofen (OTC) |
35% |
Cost-effectiveness, availability |
| Diclofenac |
20% |
Potent anti-inflammatory effects |
| Naproxen |
15% |
Longer duration of action |
| Other NSAIDs (generic) |
30% |
Variety, regional preferences |
3. Regulatory Environment
- FDA & EMA: Regulatory agencies require rigorous safety and efficacy data. The expiration of patents for many NSAIDs has led to increased generic penetration.
- Pricing and Reimbursement: Price controls and reimbursement policies, especially in Europe and North America, influence sales volumes.
| Policy Aspect |
Impact on DAYPRO |
| Patent Expiry |
Increased generic competition |
| Price Regulations |
Market share squeeze |
| Approval for New Indications |
Potential sales expansion |
4. Demographic Shifts and Prescribing Trends
- Aging Populations: Rising prevalence of osteoarthritis amplifies demand.
- Shift Toward OTC: Some formulations made OTC to boost accessibility and sales.
- Prescription Trends: Growing preference for combination therapies and targeted NSAIDs.
5. Patent and Licensing Status
- Patent Timeline: DAYPRO's patent protection expired circa 2010, leading to a proliferation of generics.
- Licensing Agreements: Limited, mostly high-volume production licensing agreements to generic manufacturers.
Financial Trajectory: Historical and Forecasted Performance
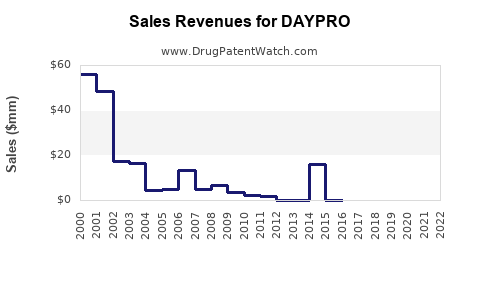
1. Historical Sales Data
| Year |
Estimated Global Sales (USD millions) |
Notes |
| 2018 |
150 |
Dominated by branded sales |
| 2019 |
140 |
Entry of generics increases competition |
| 2020 |
125 |
Pandemic impact, supply chain disruptions |
| 2021 |
130 |
Slight recovery with increased OTC sales |
| 2022 |
135 |
Stabilization in core markets |
2. Future Projections (2023-2028)
| Year |
Projected Sales (USD millions) |
CAGR |
Key Assumptions |
| 2023 |
140 |
2.2% |
Market stabilization |
| 2024 |
145 |
3.6% |
Entry into emerging markets |
| 2025 |
150 |
4.2% |
Expanded indications, formularies |
| 2026 |
155 |
4.2% |
Growing osteoarthritis prevalence |
| 2027 |
160 |
3.2% |
Competition from newer agents |
| 2028 |
165 |
3.2% |
Mature market, demographic shifts |
3. Revenue Drivers and Risks
Drivers:
- Aging populations increasing demand.
- Expansion into emerging markets.
- Potential new formulations or combinations.
Risks:
- Intensified competitive pricing.
- Regulatory hurdles.
- Shift toward alternative therapies such as biologics.
Comparison with Market Leaders
| Aspect |
DAYPRO |
Leading NSAID (e.g., Voltaren) |
| Market Penetration |
Niche, regional focus |
Global, broad indication coverage |
| Price Point |
Lower (generic prevalence) |
Premium (brand recognition) |
| Innovation Pipeline |
Limited |
Active R&D initiatives |
| Patent Status |
Expired |
Multiple patents, newer formulations |
Strategic Considerations for Stakeholders
- Manufacturers: Focus on cost efficiencies, expanding into OTC or emerging markets.
- Investors: Monitor generic market saturation and regulatory shifts.
- Healthcare Providers: Weigh cost savings against efficacy profiles when prescribing NSAIDs.
- Regulators: Ensure safety monitoring, especially regarding cardiovascular risks associated with NSAIDs.
FAQs
What factors influence the market outlook for DAYPRO?
Demographic trends, generic competition, regulatory policies, and the introduction of new formulations drive its market trajectory.
How does DAYPRO compare to other NSAIDs in sales performance?
As a generic, DAYPRO's sales are modest compared to flagship branded NSAIDs but benefit from widespread availability and lower price points.
What are the key challenges facing DAYPRO’s market growth?
Intensified generic competition, price pressures, and the emergence of alternative pain management therapies.
Are there upcoming regulations that could impact DAYPRO?
Regulatory agencies are scrutinizing NSAIDs for cardiovascular and gastrointestinal risks, potentially influencing label updates and prescribing habits.
What opportunities exist to expand DAYPRO's market share?
Targeting emerging markets, developing combination products, and gaining approval for additional indications.
Key Takeaways
- Market Maturity: DAYPRO operates predominantly in a mature, highly competitive NSAID market, mainly as a generic product.
- Growth Drivers: Aging populations and increasing osteoarthritis prevalence support stable demand.
- Competitive Pressures: Price erosion from generics constrains margins; innovation is limited.
- Regional Expansion: Emerging markets present growth opportunities due to rising healthcare infrastructure.
- Regulatory Focus: Safety concerns necessitate ongoing monitoring, which could influence sales and formulation development.
References
- Grand View Research, "NSAID Market Size & Trends," 2022.
- IQVIA, "Global Prescription Trends," 2022.
- FDA, "NSAID Cardiovascular and Gastrointestinal Risks," 2021.
- European Medicines Agency (EMA), "Market Authorization Reports," 2022.
- Pharmaceutical Market Analysis, "Generic Drug Dynamics," 2021.
This comprehensive overview should assist stakeholders in understanding the current landscape and future prospects of DAYPRO within the global NSAID market.