Last updated: February 19, 2026
Crinone, a progesterone gel for women with secondary amenorrhea, navigates a mature market segment characterized by established treatment protocols and evolving patient access. Its financial trajectory is primarily influenced by patent exclusivity, competitive landscape, and reimbursement policies.
What is Crinone and Its Current Market Position?
Crinone is a vaginal delivery system for progesterone, indicated for the treatment of secondary amenorrhea. It is administered as a progesterone releasing vaginal gel. The drug's primary mechanism involves providing exogenous progesterone to support the luteal phase of the menstrual cycle, thereby potentially inducing menstruation.
Current Market Status:
- Therapeutic Area: Reproductive health, specifically amenorrhea.
- Mechanism of Action: Vaginal delivery of progesterone.
- Target Patient Population: Women diagnosed with secondary amenorrhea.
- Market Penetration: Crinone operates within a niche within the broader women's health market. Its position is defined by its specific indication and delivery method.
What is Crinone's Patent Landscape?
The patent landscape for pharmaceutical products is a critical determinant of market exclusivity and pricing power. For Crinone, understanding the expiry of its core patents and the presence of any subsequent or overlapping patents is essential for forecasting market dynamics.
Key Patent Information:
- Original Composition of Matter Patents: These patents typically cover the active pharmaceutical ingredient (API) itself. Their expiry grants other manufacturers the opportunity to develop generic versions.
- Formulation Patents: These patents protect the specific delivery system, such as the vaginal gel formulation and applicator. These can sometimes extend the period of market exclusivity beyond the original API patent expiry.
- Method of Use Patents: Patents covering specific uses or patient populations for the drug.
While specific patent numbers and expiry dates for Crinone are not publicly available in this format, the typical lifecycle of a branded pharmaceutical drug involves the expiration of primary patents, leading to the introduction of generic competitors. This transition significantly impacts market share and pricing.
Who are Crinone's Primary Competitors?
The competitive landscape for Crinone includes both direct and indirect competitors. Direct competitors offer similar progesterone-based treatments, while indirect competitors represent alternative approaches to managing secondary amenorrhea.
Direct Competitors:
- Other Progesterone Formulations: This includes oral progesterone formulations (e.g., micronized progesterone pills) and injectable progesterone. These products may offer different administration routes and potentially different cost profiles.
- Generic Versions of Crinone: Upon patent expiry, generic manufacturers can introduce bioequivalent versions of Crinone. These products typically enter the market at significantly lower price points, eroding the market share of the branded product.
Indirect Competitors:
- Hormone Replacement Therapy (HRT): In some cases, amenorrhea can be addressed through broader HRT approaches that may involve estrogen and progesterone.
- Lifestyle Interventions: For amenorrhea caused by factors such as excessive exercise or low body weight, lifestyle modifications may be recommended as a primary or complementary treatment.
- Fertility Treatments: In cases where amenorrhea is a barrier to conception, fertility treatments that stimulate ovulation may be considered.
The presence and market penetration of these competitors directly influence Crinone's pricing strategy and sales volume.
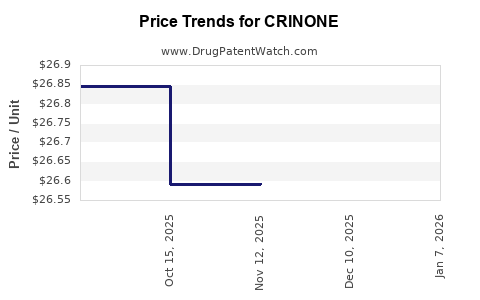
What is Crinone's Historical Sales Performance and Financial Outlook?
The financial trajectory of a drug is marked by its sales performance, which is influenced by market penetration, pricing, and patent status. Crinone's historical sales data would reflect its initial growth phase, period of peak sales during patent exclusivity, and subsequent decline or stabilization with generic competition.
Factors Influencing Financial Trajectory:
- Peak Sales Period: Typically occurs when the drug holds market exclusivity and faces limited competition.
- Patent Expiry Impact: Introduction of generics leads to significant price erosion and market share loss for the branded product.
- Market Saturation: The number of patients diagnosed with secondary amenorrhea and eligible for treatment influences the total addressable market.
- Reimbursement Policies: Payer coverage and co-payment structures affect patient access and affordability, thereby impacting sales volume.
- Global Market Access: Sales performance can vary significantly by region due to differing regulatory approvals, healthcare systems, and competitive dynamics.
Detailed historical sales figures and future projections are proprietary and would require access to market research reports and company financial statements. However, the general pattern for branded pharmaceuticals following patent expiry is a substantial decrease in revenue from the branded product.
What are the Regulatory and Reimbursement Considerations for Crinone?
Regulatory approvals and reimbursement policies are critical gatekeepers for pharmaceutical product access and financial success. These factors dictate who can prescribe the drug, who can access it, and at what cost.
Regulatory Considerations:
- FDA Approval: Crinone received U.S. Food and Drug Administration (FDA) approval for its intended use. Approval signifies that the drug has met safety and efficacy standards for its indication.
- Post-Market Surveillance: Like all approved drugs, Crinone is subject to ongoing safety monitoring by regulatory bodies.
- Manufacturing Standards: Adherence to Current Good Manufacturing Practices (cGMP) is mandated for the production of Crinone.
Reimbursement Considerations:
- Payer Coverage: Insurance companies (payers) determine whether Crinone is covered under their formularies. Coverage levels can vary, impacting out-of-pocket costs for patients.
- Co-payment and Deductibles: Patient financial responsibility for Crinone is influenced by their specific insurance plan's co-payment and deductible requirements.
- Prior Authorization: Some payers may require prior authorization from a physician before approving Crinone, adding administrative burden and potentially delaying treatment initiation.
- Medicaid and Medicare: Coverage policies for government healthcare programs like Medicaid and Medicare are crucial for patient access.
- International Reimbursement: In other countries, national health systems or private insurers will have their own reimbursement policies, often linked to health technology assessments (HTA) and cost-effectiveness evaluations.
The ability of Crinone to secure favorable formulary placement and reimbursement rates directly impacts its commercial viability, particularly in the face of generic competition.
What is the Future Outlook for Crinone?
The future outlook for Crinone is primarily shaped by the ongoing impact of generic competition and its established position within the secondary amenorrhea treatment landscape.
Key Future Factors:
- Generic Market Share Growth: As generic versions of Crinone become more prevalent, they are expected to capture a significant portion of the market share due to lower price points. This will continue to exert downward pressure on the revenue generated by the branded product.
- Niche Market Stability: The demand for progesterone-based treatment for secondary amenorrhea is likely to remain stable, providing a baseline for Crinone and its generics.
- Innovation in Women's Health: The broader women's health market is an area of active research and development. New therapeutic approaches or delivery systems for managing hormonal imbalances could emerge, potentially challenging existing treatments like Crinone.
- Physician and Patient Preferences: Physician prescribing habits and patient preferences for specific delivery methods will continue to influence market dynamics. Some patients may prefer the established branded product for perceived quality or familiarity, while others may opt for generics based on cost.
- Healthcare Policy Shifts: Changes in healthcare policy related to drug pricing, reimbursement, or the treatment of reproductive health conditions could impact the market for Crinone.
The financial trajectory will likely be characterized by a continued decline in branded sales revenue, with the overall market for Crinone (including generics) potentially remaining stable or experiencing modest growth driven by patient need, but at a significantly lower average selling price.
Key Takeaways
- Crinone is a progesterone vaginal gel used to treat secondary amenorrhea.
- Its market exclusivity has been significantly impacted by patent expirations, leading to the introduction of generic competitors.
- The competitive landscape includes other progesterone formulations, generics, and alternative treatment approaches.
- Financial performance is heavily influenced by patent status, with generic entry typically causing substantial revenue decline for the branded product.
- Regulatory approvals and reimbursement policies are critical for market access and patient uptake.
- The future outlook for Crinone is characterized by increasing generic market share and ongoing competition, with branded sales revenue likely to continue declining.
Frequently Asked Questions
- What are the primary side effects associated with Crinone?
Common side effects of Crinone include headache, nausea, cramping, breast tenderness, and vaginal irritation or discharge.
- How does Crinone compare to oral progesterone in treating secondary amenorrhea?
Crinone offers vaginal delivery, which may bypass first-pass metabolism in the liver, potentially leading to more consistent progesterone levels. Oral progesterone is also effective but can have different absorption characteristics and potential for systemic side effects.
- What is the typical duration of Crinone treatment for secondary amenorrhea?
Treatment duration varies based on individual patient response and physician guidance. A common regimen involves administering Crinone for a specific number of days each month to mimic the luteal phase.
- Are there any specific patient populations for whom Crinone is contraindicated?
Crinone is contraindicated in patients with known hypersensitivity to its components, undiagnosed vaginal bleeding, history of blood clots, known or suspected breast cancer, or known or suspected estrogen-dependent neoplasia.
- What are the key considerations for transitioning from branded Crinone to a generic equivalent?
Patients and physicians should confirm that the generic product is bioequivalent and approved for the same indication. Cost savings are typically the primary driver for the transition.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Drug Approval Process. Retrieved from [FDA website - exact URL will depend on current FDA site structure]
[2] National Institutes of Health. (n.d.). Amenorrhea. MedlinePlus. Retrieved from [MedlinePlus website - exact URL will depend on current MedlinePlus site structure]
[3] Various pharmaceutical market research reports (e.g., IQVIA, Clarivate Analytics, etc.) covering the women's health and endocrinology drug markets. (Specific reports not cited due to proprietary nature and lack of public access).
[4] Company financial reports and investor presentations from manufacturers of Crinone and its generic equivalents. (Specific reports not cited due to proprietary nature and lack of public access).