CLOBETASOL Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Clobetasol, and when can generic versions of Clobetasol launch?
Clobetasol is a drug marketed by Alembic, Glenmark Pharms Ltd, Novast Labs, Padagis Israel, Taro, Xiromed, Amneal, Aurobindo Pharma Usa, Chartwell Rx, Cosette, Encube, Fougera Pharms Inc, Glenmark Speclt, Lupin Ltd, Pai Holdings Pharm, Rising, Sun Pharma Canada, Teva Pharms Usa, Torrent, Zydus Pharms, Fougera Pharms, Padagis Us, Actavis Mid Atlantic, Beach Prods, Epic Pharma Llc, Zydus Lifesciences, Macleods Pharms Ltd, Novel Labs Inc, Hikma, Prinston Inc, Quagen, Saptalis Pharms, Wockhardt Bio Ag, Apotex, Sciegen Pharms, and Ani Pharms. and is included in eighty-two NDAs.
The generic ingredient in CLOBETASOL is clobetasol propionate. There are fourteen drug master file entries for this compound. Forty-five suppliers are listed for this compound. Additional details are available on the clobetasol propionate profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Clobetasol
A generic version of CLOBETASOL was approved as clobetasol propionate by COSETTE on February 16th, 1994.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for CLOBETASOL?
- What are the global sales for CLOBETASOL?
- What is Average Wholesale Price for CLOBETASOL?
Summary for CLOBETASOL
| US Patents: | 0 |
| Applicants: | 36 |
| NDAs: | 82 |
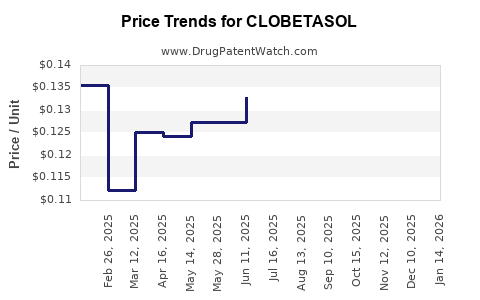
| Drug Prices: | Drug price information for CLOBETASOL |
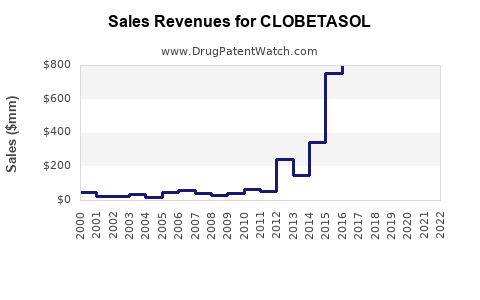
| Drug Sales Revenues: | Drug sales revenues for CLOBETASOL |
| DailyMed Link: | CLOBETASOL at DailyMed |
US Patents and Regulatory Information for CLOBETASOL
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Glenmark Speclt | CLOBETASOL PROPIONATE | clobetasol propionate | SPRAY;TOPICAL | 209004-001 | Mar 26, 2018 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Encube | CLOBETASOL PROPIONATE | clobetasol propionate | OINTMENT;TOPICAL | 211295-001 | Nov 15, 2019 | AB | RX | No | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Epic Pharma Llc | CLOBETASOL PROPIONATE | clobetasol propionate | LOTION;TOPICAL | 211348-001 | Oct 26, 2018 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Xiromed | CLOBETASOL PROPIONATE | clobetasol propionate | OINTMENT;TOPICAL | 209701-001 | Apr 17, 2018 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |