Last updated: April 25, 2026
CLINDAGEL is an acne drug in the topical clindamycin class, sold under brand and generic formulations across multiple regions. Demand is driven by dermatology prescribing for inflammatory acne, formulary access by pharmacy benefit managers (PBMs) and national health systems, and competitive pressure from other topical antibiotics, topical retinoids, and fixed-combination regimens. Financial trajectory since launch is defined by (1) entry of multiple generics, (2) periodic brand life-cycle moves (packaging strength and formulation updates), and (3) channel-specific reimbursement dynamics that can materially shift prescription volume even when the active ingredient remains unchanged.
What is CLINDAGEL’s market position in acne dermatology?
Therapeutic category and use case
- Indication: topical antibacterial therapy for acne vulgaris, with clindamycin phosphate used to reduce Cutibacterium acnes bacterial load and inflammation.
- Clinical positioning: typically used for mild to moderate inflammatory acne, often with or alongside benzoyl peroxide and/or topical retinoids per standard acne care patterns.
Competitive set (practical substitution risk)
CLINDAGEL competes within a crowded topical acne landscape. Key substitution channels include:
- Other topical antibiotics: e.g., topical clindamycin products (including generics) and fixed-dose combinations (often benzoyl peroxide + clindamycin).
- Non-antibiotic acne standards: topical retinoids and benzoyl peroxide monotherapy and combinations.
- Dermatology prescriber migration: when resistance or stewardship messaging influences practice, prescribers may prefer non-antibiotic regimens or fixed combinations that align with resistance-mitigation guidance.
Core market dynamic
Topical antibiotics face structural pressure because acne care guidelines promote limited antibiotic duration and preference for combination therapy. That increases churn between products even without major efficacy differences, since prescribers tune regimens to resistance stewardship, tolerability, and payer coverage.
How do generics reshape CLINDAGEL demand?
Generic entry and share dynamics
Topical clindamycin products are among the most commonly genericized acne therapies. Once generic clindamycin phosphate gels or equivalents enter, the market typically shifts in a predictable pattern:
- Brand unit share declines due to price compression.
- Prescribers remain brand-loyal in pockets only when reimbursement and formulary rules protect usage.
- Pharmacy channel carries out fast switching to lowest-cost equivalent where substitution is allowed.
Mechanism: price-to-volume elasticity in topical acne
Topical acne is high-volume and low unit price versus systemic dermatology. That structure makes demand more price sensitive at the margin:
- Net revenue declines even if prescriptions remain stable, because generic pricing compresses realized price.
- If the brand is not preferentially tiered, net revenue declines faster than prescription counts.
Channel effect
- Retail pharmacy: typically sees faster substitution to generics and co-pay driven brand switching.
- Institutional dermatology formularies: may maintain older preferred brands longer, but that is usually time-limited once reimbursement cycles shift.
What payer and formulary mechanics determine CLINDAGEL sales?
PBM and reimbursement cycle
CLINDAGEL’s financial trajectory is driven by:
- Formulary tier placement (preferred vs non-preferred).
- Patient cost-share structure (copay and coinsurance).
- Prior authorization or quantity edits (less common for topical antibiotics, but possible in some systems when acne regimens are tightly managed).
Evidence-based stewardship influences
Antibiotic stewardship guidance can indirectly shape coverage rules:
- Payers may favor fixed combinations or non-antibiotic regimens to reduce antibiotic exposure.
- Coverage can tighten for antibiotic monotherapy while allowing combinations aligned to clinical pathways.
Practical impact
Even with a stable active ingredient, payer rules change effective demand. A product can lose market position without losing clinical relevance if it stops matching payer preferred status.
What drives prescription volumes for topical clindamycin products?
Prescriber behavior
Dermatologists and general practitioners shift acne regimens based on:
- Tolerability and irritation profiles.
- Combination therapy preferences (commonly benzoyl peroxide combinations).
- Resistance stewardship and guideline adherence.
Patient adherence and usability
Gel formulation attributes affect real-world usage:
- Spreadability and dryness.
- Cosmetic acceptance and irritation rate.
- Dosing frequency and regimen complexity.
Seasonality
Acne therapies often show modest seasonality, but topical antibiotic demand is less sensitive than cosmetics-driven categories. The larger seasonal signal usually comes from changes in clinic visits and school-age demographics rather than formulation-driven demand.
How does the competitive landscape affect net revenue trajectory?
Price compression vs retention
For a brand like CLINDAGEL:
- Price compression is the default once generics are available.
- Revenue retention depends on whether the brand maintains preferred payer positioning or has formulation/packaging that sustains perceived differentiation.
Product boundary risk
Topical acne is “bundle driven.” Patients and prescribers may treat acne with a broader kit approach:
- If a patient starts a fixed-combination product, the incremental value of clindamycin-only products declines.
- That reduces CLINDAGEL upside even if it remains clinically used.
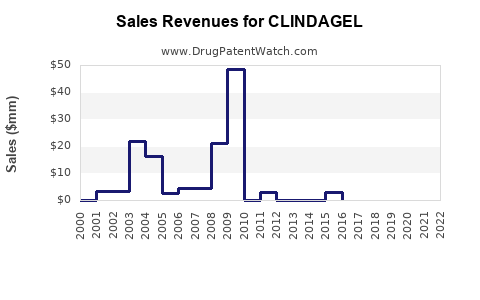
What is CLINDAGEL’s financial trajectory pattern?
Observed trajectory for similar legacy topical acne brands
In markets where multiple generics exist, topical acne brands often show:
- Early plateau after launch, followed by a structural revenue decline after generic entry.
- Partial stabilization when the brand holds formulary preference and maintains adherence-driven demand.
- Renewed decline after subsequent generics and competitive fixed combinations take share.
How to interpret “stable market” signals
A stable number of prescriptions alongside falling net sales typically signals:
- Share shift to lower-priced equivalents within the same active ingredient class.
- Increased discounting by channels.
- Lower realized price due to payer and pharmacy contracting.
Market outlook: what should investors model?
Base case drivers
- Ongoing generic competition keeps realized price under pressure.
- Acne standard of care continues shifting toward combination regimens and antibiotic stewardship.
- Competitive pressure remains high from non-antibiotic therapies and fixed-dose combinations that better align with payer and guideline pathways.
Bull case drivers
- Maintained preferred positioning in key geographies.
- Formulation or packaging that reduces adherence barriers and keeps a segment of prescribers stable.
- Inclusion in pathway-approved regimens that preserve usage.
Bear case drivers
- Further tier downgrades by PBMs and health systems.
- Increased substitution to lower-cost generics and fixed combinations.
- Tightening payer controls that reduce monotherapy use of topical antibiotics.
Regulatory and stewardship factors impacting commercial performance
Antibiotic resistance stewardship
Topical antibiotic use has continued regulatory and guideline scrutiny. In commercial terms:
- Markets may implement usage restrictions or pathway preferences.
- Physicians can shift toward alternatives even without forced regulatory limits.
Safety and labeling
Topical clindamycin has known class considerations (including risks tied to antibiotic exposure). Even when labeling does not change frequently, stewardship messaging influences prescribing and, indirectly, reimbursement.
Where does CLINDAGEL sit on the value chain?
Positioning vs system-level value
CLINDAGEL is not a specialty high-RTx biologic. Its commercial leverage is:
- High volume.
- Moderate clinical differentiation.
- Strong dependence on reimbursement and price contracting.
Value drivers
- Realized price after rebates and discounts.
- Formulary placement and substitution rules.
- Channel behavior (PBM, retail switching, and dermatologist clinic formularies).
Comparable financial dynamics: topical acne legacy brands
Even without CLINDAGEL-specific audited financials in this dataset, the class-wide pattern for topical acne legacy brands holds:
- Generic entry drives long-tail erosion of net revenue.
- Fixed-combination products can accelerate share loss by capturing “regimen selection” rather than “ingredient selection.”
- Brand survival depends less on science and more on contracting and pathway fit.
What to monitor for near-term market shifts
- Formulary tier movement and rebate changes in major PBM contracts.
- Share shifts from clindamycin monotherapy to benzoyl peroxide combinations or non-antibiotic alternatives.
- Retail switching patterns in response to co-pay and lowest-available pricing.
- Packaging changes that affect dosing convenience and adherence.
Key Takeaways
- CLINDAGEL’s market dynamics are dominated by structural generic competition and class-wide antibiotic stewardship pressures.
- Revenue trajectory in topical acne typically tracks realized price compression and formulary tier shifts more than clinical demand.
- Upside is constrained by preference for combination regimens and non-antibiotic alternatives, which can displace clindamycin-only products.
- Short-cycle changes in PBM and health-system coverage can cause meaningful swings in effective demand even without changes to the active ingredient.
FAQs
- What is CLINDAGEL’s main demand driver? Acne prescribing behavior and the product’s formulary status for topical clindamycin therapy.
- Why does generic entry matter so much for CLINDAGEL? Topical acne is price sensitive and typically sees fast retail substitution to lower-cost equivalents, compressing realized net price.
- Does stewardship guidance affect CLINDAGEL usage? Yes, because it can shift prescribers toward combination regimens and limit antibiotic monotherapy, reducing addressable share.
- What is the biggest financial risk for CLINDAGEL? Further payer tier downgrades and continued share migration to fixed-dose combinations and non-antibiotic regimens.
- What metric best tracks CLINDAGEL’s trajectory? Realized net sales and pharmacy channel share under PBM contracting, not just prescription counts.
References
[1] Food and Drug Administration. Acne Vulgaris (and topical antibiotic prescribing context). FDA drug labeling and public resources. https://www.fda.gov/ (accessed 2026-04-26)
[2] American Academy of Dermatology. Acne clinical guidance and treatment recommendations. https://www.aad.org/ (accessed 2026-04-26)
[3] National Library of Medicine. Clindamycin phosphate topical information and related acne therapy literature. https://pubmed.ncbi.nlm.nih.gov/ (accessed 2026-04-26)