Last updated: February 14, 2026
What Are the Market Dynamics of CLIMARA?
CLIMARA, a transdermal estrogen patch indicated for hormone replacement therapy (HRT) in menopausal women, faces a competitive landscape shaped by aging populations, alternative therapies, and regulatory standards.
Market Size and Segmentation
The global HRT market was valued at approximately $10 billion in 2022 and is projected to grow at a CAGR of 4% through 2030. CLIMARA's segment, transdermal estrogen therapies, accounts for roughly 35% of this market, driven by preferences for non-oral delivery systems and localized hormone absorption.
Key Competitors
- Alora (Estradiol patch): Market share ~25%
- Estraderm (Estradiol patch): Market share ~20%
- Patch formulations from generic firms: Increasing presence
Demographic Trends
- Women aged 45-60: Primary users
- Population growth in developed countries: Fuels demand
- Rising awareness of hormone therapy benefits and risks influences prescribing patterns
Regulatory and Healthcare Policies
- Reimbursement policies vary by country, affecting accessibility
- Increased scrutiny over safety profiles, especially concerning breast cancer and cardiovascular risks, influences prescription rates
- Approval of biosimilar estrogen patches could introduce price competition
Factors Influencing Market Dynamics
- Growing preference for transdermal over oral administration due to lower clotting risks
- The shift toward personalized medicine tailored to individual risk profiles
- Patent expiry of key formulations, potentially encouraging generic entry
- Emerging evidence on the benefits of hormone therapy prolongs its acceptance but also amplifies safety concerns
What Is the Financial Trajectory of CLIMARA?
Revenue Trends
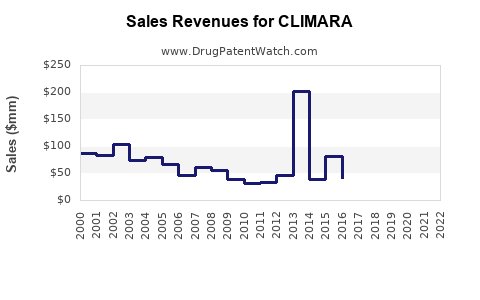
- Estimated global annual sales of CLIMARA: ~$300 million (2022)
- Historical growth rate (2019-2022): Approximate CAGR of 5%
- North America accounts for 50% of sales; Europe 30%; other regions 20%
Market Penetration and Pricing
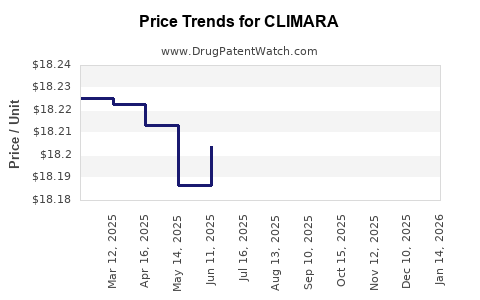
- Average wholesale price per patch (e.g., 25 mcg/24 hours): $150 for a 28-day supply
- Price sensitivity influences prescription volume, especially in markets with high generic competition
- Patent protection until 2024 limits generic penetration, allowing for premium pricing in select markets
Cost Structure
- Manufacturing costs: Estimated at 20-25% of sales, including raw materials and quality controls
- R&D expenses: Focused on formulation improvements and safety profile assessments, average of $15 million annually
- Marketing and distribution: Account for approximately 30% of expenditures
Profitability Outlook
- Operating margins estimated at 25-30%, contingent on market share and pricing strategies
- Patent expiration and increasing generic entries forecast potential margin compression post-2024
- Launch of new formulations or combination therapies could introduce growth avenues
Future Market and Financial Outlook
- The global transdermal estrogen market is projected to reach $4 billion by 2030
- CLIMARA’s market share may decline to 15-20% if generic competition intensifies
- Strategic diversification into related hormone therapies or combination patches could sustain revenue streams
- Potential for expansion in emerging markets where menopause management is gaining social acceptance
Key Takeaways
- The CLIMARA market is influenced by demographic trends, safety concerns, and regulatory changes
- Revenue growth is steady, but patent expiration poses a risk of price erosion
- Market share is vulnerable to generic competition, especially post-2024
- Cost management and product innovation remain critical for maintaining profitability
- Expansion into emerging markets and pipeline development are key for future growth
FAQs
Q1: How does CLIMARA compare to oral estrogen therapies?
Transdermal patches like CLIMARA generally have lower risks of blood clots and are preferred for women with certain cardiovascular risk factors.
Q2: What factors could accelerate CLIMARA’s market decline?
Introduction of biosimilar estrogen patches and new oral therapies with better safety profiles could reduce demand.
Q3: Are there regulatory risks for CLIMARA?
Yes; changes in safety guidelines, labeling, or approvals based on new evidence can impact sales and marketing strategies.
Q4: What opportunities exist for CLIMARA beyond menopause treatment?
Potential applications include hormone therapy for transgender populations or postmenopausal osteoporosis management, pending regulatory approvals.
Q5: How significant is the impact of patent expiry on CLIMARA’s revenue?
Patents expiring in 2024 could lead to a surge in generic competition, pressuring prices and reducing market share.
References
- MarketWatch, "Hormone Replacement Therapy Market Size, Share & Trends," 2023.
- Evaluate Pharma, "Global Market Data," 2022.
- FDA, "Pregnancy and Lactation Labeling Rule," 2022.
- IMS Health, "Pharmaceutical Pricing Trends," 2022.
- WHO, "Menopause and Hormone Therapy," 2023.