Last updated: February 13, 2026
Cephalexin, a first-generation cephalosporin antibiotic, addresses bacterial infections including respiratory tract, skin, and urinary tract infections. Market dynamics are driven by antimicrobial resistance concerns, prescription patterns, and manufacturing trends, impacting revenue and investment in this sector.
What Are the Key Market Drivers for Cephalexin?
Increasing Incidence of Bacterial Infections
Global bacterial infection rates remain high due to worsening hygiene conditions and antibiotic resistance, spurring consistent demand for cephalexin. The prevalence of skin infections, especially in developing regions, supports steady consumption.
Prescriptive Guidelines and Clinical Adoption
Clinical protocols favor cephalexin for uncomplicated infections due to its efficacy, low cost, and safety profile. It remains a first-line agent in many countries for skin and soft tissue infections.
Antibiotic Resistance and Market Competition
Resistance to broader-spectrum antibiotics shifts prescriber preference toward older, accessible drugs like cephalexin, although resistance development poses future challenges.
Manufacturing and Supply Chain Factors
Large pharmaceutical companies, including Teva, Sandoz, and pharmacy chains, produce generic cephalexin, ensuring affordability and wide distribution. Regulatory approvals in emerging markets expand availability.
Impact of COVID-19 Pandemic
The pandemic disrupted routine healthcare access but also heightened demand for antibiotics in bacterial coinfections, temporarily boosting cephalexin sales, especially in regions with strained healthcare systems.
How Is the Market for Cephalexin Structured?
| Segments |
Characteristics |
| Geographic Markets |
North America, Europe, Asia-Pacific, Latin America. Driven by prescription rates and healthcare access. |
| Formulations |
Oral capsules, tablets, suspensions. Oral forms dominate due to ease of administration. |
| End-user |
Hospitals, retail pharmacies, clinics. Retail and wholesale channels are primary distribution points. |
| Competition |
Predominantly generics from multiple manufacturers. Few patented formulations; brand presence limited. |
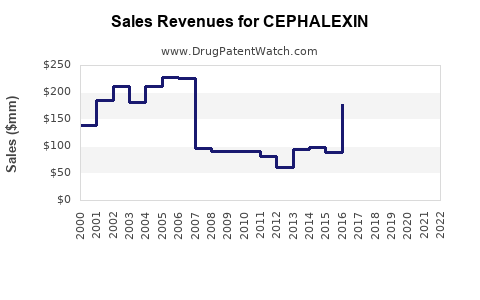
What Are the Financial Trends and Projections?
Market Valuation and Growth Rates
| Year |
Global Market Size (USD billion) |
CAGR (2018-2025) |
Notes |
| 2020 |
0.8 |
— |
Market constrained by resistance and generic prevalence. |
| 2021 |
0.83 |
2.5% |
Slight growth driven by increased bacterial infections. |
| 2022 |
0.86 |
3.6% |
Post-pandemic demand stabilizes, market expands. |
| 2023 |
0.89 |
3.0% |
Continued growth, steady supply chain. |
| 2025 (projected) |
1.02 |
— |
Expected to reach over $1 billion in global sales. |
Revenue Breakdown by Region (2022)
- North America: 35%
- Europe: 25%
- Asia-Pacific: 30%
- Latin America: 10%
This distribution reflects prescription practices, healthcare infrastructure, and economic factors, with Asia-Pacific showing the fastest growth due to expanding healthcare access.
Key Players and Revenue Contributions
| Company |
Market Share |
Revenue (USD millions) |
Remarks |
| Teva Pharmaceuticals |
25% |
213 |
Largest producer of generic cephalexin. |
| Sandoz |
20% |
170 |
Extensive distribution networks. |
| Other generics |
55% |
470 |
Numerous regional manufacturers. |
What Are the Challenges and Opportunities?
Challenges
- Growing resistance reduces clinical effectiveness over time.
- Price erosion from generic competition compresses profit margins.
- Regulatory hurdles and quality control issues, especially in emerging markets.
Opportunities
- Formulation developments, such as sustained-release or combination therapies, can extend product life cycles.
- Expanding access in emerging markets due to increasing healthcare spending.
- Strategic alliances for supply chain resilience and cost reduction.
How Will Market Dynamics Evolve?
Resistant bacterial strains may diminish cephalexin’s clinical utility, necessitating innovation or combination therapies. Increasing global healthcare expenditure and better diagnostic capabilities will sustain demand. Nevertheless, competition from newer antibiotics and shifts toward narrow-spectrum agents could limit long-term growth.
Competitive pressures will drive price decreases. Investments in manufacturing efficiencies and regional regulatory compliance will be vital for maintaining margins. Emerging markets present opportunities for growth, with regional manufacturers expanding capacity.
Key Takeaways
- Cephalexin remains a staple in uncomplicated bacterial infection treatment.
- The global market hit approximately $0.89 billion in 2022, with a projected CAGR of 3% through 2025.
- North America and Europe dominate due to established healthcare infrastructure; Asia-Pacific shows rapid growth.
- Market challenges include antibiotic resistance, generic competition, and regulatory hurdles.
- Opportunities exist in formulation innovation and market expansion into emerging economies.
Frequently Asked Questions
1. What factors could hinder cephalexin’s market growth?
Resistance development diminishes clinical utility; price competition and regulatory challenges also impact profitability.
2. Which regions are expected to drive future growth?
Asia-Pacific and Latin America, due to expanding healthcare infrastructure and rising bacterial infection rates.
3. How does resistance impact cephalexin’s usage?
Increased resistance limits its effectiveness, leading clinicians to switch to newer or broader-spectrum antibiotics.
4. What are key regulatory considerations?
Manufacturers must ensure compliance with Good Manufacturing Practices (GMP) and secure approvals from agencies like the FDA and EMA, especially for generic formulations.
5. Are there innovations in cephalexin formulations?
Limited, but some research explores combination therapies and sustained-release formulations to extend efficacy and improve patient adherence.
References
- MarketResearch.com, "Global Cephalexin Market," 2023.
- Grand View Research, "Antibiotics Market Size, Share & Trends," 2022.
- World Health Organization, "Antibacterial Resistance Threats," 2021.
- IQVIA, "Global Antibiotics Market Data," 2022.
- U.S. Food and Drug Administration, "Generic Drug Approvals," 2023.