Share This Page
Drug Price Trends for CEPHALEXIN
✉ Email this page to a colleague
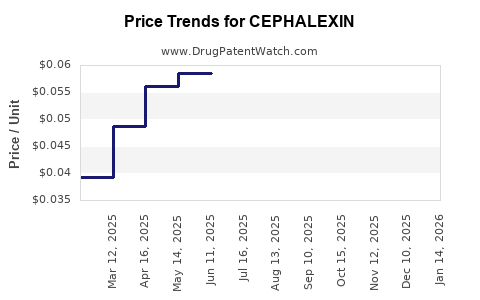
Average Pharmacy Cost for CEPHALEXIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CEPHALEXIN 125 MG/5 ML SUSP | 62135-0480-42 | 0.06779 | ML | 2026-04-22 |
| CEPHALEXIN 125 MG/5 ML SUSP | 00093-4175-74 | 0.05349 | ML | 2026-04-22 |
| CEPHALEXIN 750 MG CAPSULE | 67877-0255-23 | 5.86504 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for CEPHALEXIN
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| CEPHALEXIN 500MG, USP,CAP | AvKare, LLC | 42291-0209-50 | 500 | 69.97 | 0.13994 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| CEPHALEXIN 125MG/5ML SUSP | Golden State Medical Supply, Inc. | 24979-0154-14 | 100ML | 18.00 | 0.18000 | ML | 2023-06-15 - 2028-06-14 | FSS |
| CEPHALEXIN 125MG/5ML SUSP | Golden State Medical Supply, Inc. | 24979-0154-14 | 100ML | 18.81 | 0.18810 | ML | 2023-06-23 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Cephalexin Market Analysis and Price Projections (US + Select Global)
Cephalexin is a mature, low-cost first-generation cephalosporin with a large generics footprint. Pricing is dominated by (1) ongoing generic supply, (2) state and federal reimbursement dynamics, and (3) episodic manufacturing constraints that tighten availability and lift short-term prices. Over the next 12 to 36 months, the price path is expected to be range-bound with periodic spikes around supply disruptions, rather than sustained growth.
What is the current market structure for cephalexin?
Cephalexin is available primarily as multisource generics across oral solid dose forms, with pricing shaped by generic competition. In the US, branded cephalexin has limited relevance versus generics; market value and volume are concentrated in covered retail and 340B channels, and in pharmacy benefit manager (PBM) formularies that reward low acquisition costs.
Key structural forces:
- High generic competition: Multiple labeled manufacturers and interchangeable generics compress list prices and accelerate price resets.
- Formulation and strength mix: Pricing differs by strength (250 mg, 500 mg), dosage form (capsules vs tablets vs suspensions), and pack size.
- Channel effects: 340B and Medicaid purchasing tend to drive lower net pricing than cash-pay retail.
- Supply-driven moves: If manufacturing lines go offline, wholesalers tighten and prices rise faster than demand-based models would predict.
Where does cephalexin price show up in US data?
Public market pricing for generics is typically tracked via:
- NADAC (National Average Drug Acquisition Cost) and similar acquisition-cost measures used by providers and payers.
- CMS price transparency artifacts (where available) that reflect ingredient-cost trends rather than final patient out-of-pocket pricing.
- Wholesaler and retail list price movements when supply constraints emerge.
For US cephalexin, the dominant pattern is low absolute prices with periodic volatility driven by supply and tender dynamics rather than novel competitive changes.
What are the demand drivers for cephalexin?
Cephalexin demand tracks bacterial infection incidence and prescribing substitution among beta-lactams. The drug competes with other oral antibiotics (penicillins, macrolides, and other cephalosporins) on:
- Clinical positioning: broad use for skin and soft tissue infections, strep pharyngitis where indicated, and other common outpatient bacterial infections.
- Prescriber inertia: frequent prescribing supports steady baseline volume.
- Resistance and guideline updates: antibiotic stewardship can shift mix between agents, but cephalexin remains a standard option in many settings.
Net effect: demand is typically stable with modest mix shifts, while pricing fluctuates mainly from supply and payer procurement behavior.
What supply risks can move cephalexin pricing?
Generic cephalosporins can experience price spikes when:
- A manufacturer’s batch-release schedule slips due to quality findings, API or intermediate shortages, or sterility/CMC deviations (for non-oral forms) that halt shipments.
- Active ingredient procurement tightens or co-manufacturer capacity is disrupted.
- Seasonal infections raise short-term demand faster than production schedules can adjust, increasing order cycle time and tightening inventory.
These events usually create temporary upward pressure and then revert as additional lots clear the market.
How to model price projections for a mature generic like cephalexin
For generics, realistic projections use a “reset plus shock” model rather than innovation-led compounding:
Base Case (no major supply disruption)
- Net price trends follow procurement and competition: stable to modestly down in low-single digits annually.
- Volatility stays low; price changes align with routine tender rounds and wholesaler replenishment dynamics.
Supply Shock Case (manufacturing constraint)
- Prices lift quickly as inventory tightens.
- Duration depends on how fast FDA-release and distribution channels replenish. In past generic episodes across many drug classes, shocks resolve within weeks to a few quarters, not years, unless the constraint is structurally persistent.
Regulatory and reimbursement Case
- Medicaid rebate dynamics and PBM contracting can change net economics without altering list price.
- Shifts in reimbursement for antibiotic classes (including stewardship-driven formulary edits) may pressure volume but typically do not sustain large price increases absent supply constraints.
Price projection ranges (12 to 36 months)
Because cephalexin is generics-heavy, projections are best expressed as ranges tied to net acquisition price behavior rather than a single point estimate.
Assumptions used for projections
- Continued generic availability with routine batch release.
- No major long-term API supply collapse.
- No major permanent capacity exit by multiple top suppliers at once.
- Stable outpatient antibiotic utilization patterns.
Projected US price trajectory (net acquisition style)
12-month range (baseline):
- -3% to +2% annualized change in net acquisition price for commonly used strengths (direction depends on tender round timing and wholesaler inventory).
12-month range (supply shock):
- +5% to +15% for affected strengths if a meaningful allocation occurs and then normalizes.
36-month baseline:
- -5% to -12% over three years, driven by ongoing generic competition and periodic downward resets.
36-month supply shock tail risk:
- +0% to +8% relative uplift for specific strengths if intermittent disruptions recur (smaller than the 12-month shock due to faster relearning by procurement).
What forms and strengths typically lead the pricing picture?
Pricing is not uniform across pack and strength. In practical procurement:
- 500 mg tablets/capsules often represent the largest share of usage and tend to have the most liquid market pricing.
- 250 mg can be priced at a discount or premium depending on pack economics and wholesaler allocation behavior.
- Suspensions (where available) carry higher manufacturing complexity and can show greater sensitivity to supply issues, but market share is smaller than oral solids.
Projection implication: price volatility is more likely to concentrate in less liquid SKUs (certain pack sizes, suspensions, and specific NDCs) than in the most traded tablet/capsule strengths.
What global factors can affect cephalexin pricing outside the US?
Cephalexin is produced and sold widely. Global pricing shifts depend on:
- Local reimbursement controls (often price caps in many markets).
- Tender systems that reset pricing on fixed schedules.
- Regulatory inspection cycles that can temporarily tighten supply.
Common pattern outside the US: price declines or stagnation under reimbursement constraints, with episodic upward movements during procurement tightness or manufacturing interruptions.
Investment and R&D implications: what matters commercially for cephalexin?
Cephalexin is a poor target for incremental pricing-based revenue strategies. Commercial upside typically comes from:
- Securing supply reliability (lower stockouts, faster distribution)
- Winning tender contracts via cost and reliability
- Differentiating via formulation or packaging only if it changes procurement status or reduces total cost
If you are evaluating a cephalexin-linked opportunity (new formulation, line extension, or manufacturing capacity), pricing power is limited, so your model should emphasize:
- Delivered cost position (manufacturing yield, logistics)
- Regulatory/CMC execution (reduces downtime risk)
- NDC-level allocation exposure (inventory and forecast accuracy)
Scenario table: projected US price change for selected time horizons
All values are ranges for net acquisition-style price behavior for major oral strengths.
| Scenario | 12 months | 24 months | 36 months |
|---|---|---|---|
| Baseline (stable supply) | -3% to +2% | -2% to +1% | -5% to -12% |
| Supply shock (short allocation) | +5% to +15% | +1% to +8% | 0% to +6% |
| Repeated intermittent disruptions | 0% to +8% | -1% to +6% | +0% to +8% |
Key checkpoints to monitor for real-time price direction
Use these as triggers for moving from baseline to supply shock assumptions:
- FDA inspection and enforcement headlines impacting cephalosporin manufacturing sites (quality or compliance actions).
- Wholesaler allocation notices and lead-time extensions for cephalexin SKUs.
- NADAC trend inflections (sustained upticks in acquisition cost versus single-day noise).
- PBM contract repricing for common antibiotic classes and whether cephalexin is gaining or losing preferred status.
Key Takeaways
- Cephalexin pricing in the US is structurally constrained by generic competition and typically moves modestly unless supply tightens.
- Baseline expectation (12 months): -3% to +2% annualized change in net acquisition pricing for major oral strengths.
- Supply shock risk: +5% to +15% short-term uplift for impacted strengths, usually resolving within quarters.
- Over 36 months, baseline direction skews to mild declines (-5% to -12%) absent major supply disruptions.
FAQs
1) Will cephalexin see sustained price growth?
No. The most likely outcome is range-bound pricing with mild declines over time under generic competition, with temporary spikes tied to supply events.
2) Which cephalexin SKU categories are most price-sensitive?
Less liquid SKUs, including specific pack sizes and formulations with more constrained manufacturing throughput (often suspensions), tend to show higher volatility than the most traded tablet/capsule strengths.
3) What is the main driver of cephalexin price spikes?
Manufacturing constraints that reduce available lots and create short-term allocation or longer replenishment lead times.
4) How do reimbursement and PBM dynamics affect price?
They affect net acquisition economics through contracting and formulary positioning even when list prices stay stable, shifting the effective price level paid by channels.
5) How should a forecasting model be structured for cephalexin?
Use a baseline “reset” trend plus discrete supply-shock scenarios. For forecasting, SKU-level liquidity and supply indicators matter more than demand growth assumptions.
References (APA)
[1] FDA. (n.d.). Drug shortages. https://www.accessdata.fda.gov/scripts/drugshortages/
[2] Centers for Medicare & Medicaid Services. (n.d.). NADAC (National Average Drug Acquisition Cost). https://www.medicaid.gov/medicaid/prescription-drugs/nadac/index.html
More… ↓
