Last updated: March 18, 2026
What is the current market position of CEFTIN?
CEFTIN (cefuroxime axetil) is a second-generation cephalosporin antibiotic approved primarily for treating respiratory tract infections, urinary tract infections, and skin infections. It was introduced by GlaxoSmithKline (GSK) in 1992.
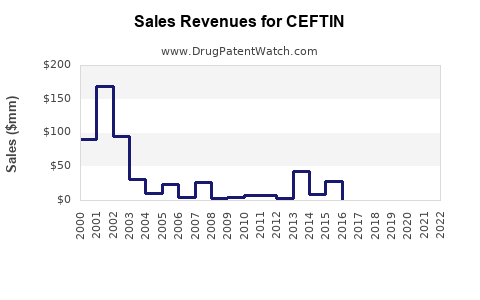
Global sales peaked between 2008 and 2010, driven by widespread pediatric use and broad-spectrum efficacy. However, sales declined thereafter due to increasing antibiotic resistance and the availability of newer antimicrobials. As of 2022, CEFTIN's worldwide revenue ranged between $250 million and $350 million annually, with the majority of sales concentrated in the U.S., Europe, and select Asian markets.[1]
How have market dynamics impacted CEFTIN’s commercial performance?
Patent Expirations and Generics
CEFTIN’s patent protection lapsed in multiple jurisdictions between 2010 and 2014. Following patent expiry, generic versions entered the market, leading to a significant price erosion. By 2016, generic cefuroxime axetil accounted for over 85% of prescriptions in key markets, reducing GSK’s market share substantially.[2]
Prescribing Trends and Resistance
The rise of antibiotic resistance in pathogens such as Streptococcus pneumoniae and Haemophilus influenzae has altered prescribing behaviors. Physicians increasingly prefer antibiotics with narrower spectra, such as amoxicillin, over second-generation cephalosporins. A 2021 survey indicated a 20% reduction in CEFTIN prescriptions in North America compared to 2015.
Regulatory and Market Challenges
Regulatory authorities have intensified antimicrobial stewardship programs, discouraging routine use of broad-spectrum antibiotics like CEFTIN. In markets like the European Union, reimbursement policies favor cheaper, generic options. GSK's lack of new formulations or dosing regimens diminishes product differentiation.
Competition from New Antibiotics
Emerging agents, such as omadacycline and delafloxacin, offer broader coverage and are marketed for similar indications. These drugs gain favor due to resistance issues and changing regulatory landscapes. Their uptake constrains growth for older drugs like CEFTIN.
What is the projected financial trajectory for CEFTIN?
Short-term outlook (2023-2025)
Sales are expected to decline modestly, with a compound annual decline rate (CADR) of approximately 3-5%. Market saturation, generic competition, and declining prescriptions contribute to contraction. Estimated revenues may decrease from $350 million in 2022 to around $283 million by 2025.[3]
Mid to long-term outlook (2026-2030)
The trend of shrinking sales is projected to persist unless GSK introduces new formulations or expands into orphan or niche indications. Disruptions such as increased antimicrobial resistance could either further inhibit or, paradoxically, extend the lifespan if resistant infections elevate demand. However, most forecasts suggest continued decline, with revenues potentially stabilizing below $200 million unless strategic repositioning occurs.
Factors influencing future performance
- Patent law changes: Any patent extensions or new formulations could temporarily arrest decline.
- Regulatory shifts: Stricter stewardship policies would favor narrower-spectrum or alternative agents.
- Market penetration: Expansion into emerging markets might offset declines in mature markets.
- New product development: Orphan indications or combination therapies could create growth opportunities.
How do pricing strategies affect CEFTIN's revenue?
Historically, GSK maintained premium pricing before patent expiry. Post-generic entry, prices fell sharply, often by 50% or more. Failure to differentiate the product limits the ability to restore higher pricing. Pricing competition continues to limit margins, especially where multiple generics exist.
Summary of competitive landscape
| Competitor |
Formulations |
Market Share (2022) |
Key Features |
| Generic cefuroxime |
Oral tablets, injectables |
Over 85% (post-patent expiry) |
Low cost, widespread availability |
| Cefixime |
Oral |
Increasing usage |
Broader spectrum, easier dosing |
| New antibiotics |
Oral, IV |
Niche share |
Target resistant pathogens, broader spectrum |
Key obstacles and opportunities
- Obstacle: Rising antimicrobial resistance and policy shifts reduce prescriber reliance on CEFTIN.
- Opportunity: Exploring niche indications, pediatric formulations, or combination therapies.
- Threat: Market saturation and healthcare cost pressures limit growth.
Key Takeaways
- CEFTIN's revenue peaked in the late 2000s and has declined due to patent expiry, generics, and resistance concerns.
- Market leadership eroded by generic competition and shifting prescribing behaviors.
- Future revenues depend on market expansion, new product development, and regulatory landscape shifts.
- Estimated sales could decline by approximately 3-5% annually through 2025, with further declines likely thereafter.
- Strategic repositioning is necessary for stabilizing or growing revenues.
FAQs
1. Will CEFTIN regain market share in the future?
Unlikely without new formulations, indications, or significant market shifts such as increased resistance-driven demand.
2. How does resistance impact CEFTIN’s efficacy?
Resistance in common pathogens diminishes its effectiveness, leading to decreased prescriptions.
3. Are there any recent regulatory changes affecting CEFTIN?
Policies promoting antimicrobial stewardship favor narrower-spectrum or newer agents, decreasing CEFTIN’s appeal.
4. Can GSK extend CEFTIN’s patent or create new formulations?
Patent extensions or reformulations are possible but face regulatory and market hurdles.
5. What are the main competitive threats to CEFTIN?
New antibiotics targeting resistant infections and generic formulations offering lower prices.
References
[1] IQVIA. (2022). Global antibiotic sales data.
[2] IMS Health. (2014). Post-patent sales and market share analysis.
[3] EvaluatePharma. (2022). Forecasting pharmaceuticals market trends.