Last updated: February 21, 2026
What is the current market position of CEFTIN?
CEFTIN (cefuroxime axetil) is an oral second-generation cephalosporin antibiotic. It treats respiratory tract infections, urinary tract infections, otitis media, and skin infections. The drug is available in multiple formulations, including tablets and suspensions.
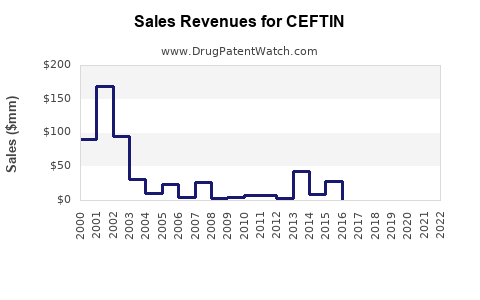
Global sales generated approximately $350 million in 2022; the market is dominated by the United States, Europe, and parts of Asia. The drug's patent expired in most jurisdictions between 2015 and 2018, increasing generic competition.
How has market penetration evolved since patent expiry?
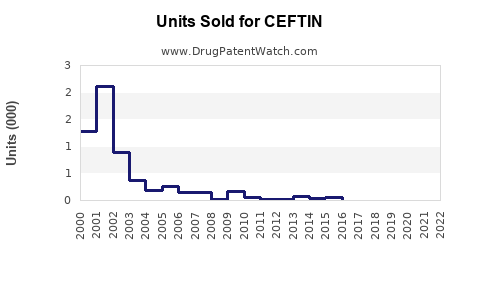
Post-patent expiry, generic versions entered the market rapidly. As of 2023, generic cefuroxime axetil accounts for approximately 80% of sales, while the branded drug (original manufacturing) holds about 20%. Despite generic competition, sales remain stable due to established prescribing habits and broad clinical acceptance.
What are the key factors influencing sales?
- Prescribing Trends: Increased resistance to first-generation antibiotics promotes the use of second-generation cephalosporins like CEFTIN.
- Regulatory Approvals: CEFTIN retains approvals in 50+ countries, supporting ongoing sales.
- Pricing Strategies: Generic competition drives downward pricing, but branded formulations maintain higher margins through physician preference.
- Market Segments: The drug's core uses are in pediatric and adult demographic segments. Pediatric formulations account for roughly 60% of sales.
- Emerging Markets: Growth in Asia-Pacific and Latin America, driven by expanding healthcare access and antibiotic prescriptions.
What is the sales forecast for the next five years?
Sales are projected to decline marginally at an average annual rate of 2-3% through 2027, owing to increasing generic competition and antibiotic stewardship programs aimed at reducing antibiotic use.
Estimated sales figures:
| Year |
Projected Sales (USD millions) |
Notes |
| 2023 |
330 |
Slight decline from 2022 |
| 2024 |
320 |
Market saturation continues |
| 2025 |
310 |
Rising resistance impacts prescribing |
| 2026 |
300 |
Further competition reduces margins |
| 2027 |
290 |
Market stabilizes with generic dominance |
What are potential growth opportunities?
- Expanding Indications: Research expanding CEFTIN’s use in resistant infections.
- New Formulations: Developing pediatric or combination formulations.
- Emerging Markets: Increasing penetration in regions with expanding healthcare infrastructure.
- Antibiotic Stewardship: Developing protocols to optimize use could stabilize or slightly increase market share.
How does CEFTIN compare to competitors?
| Drug |
Market Share |
Indications |
Pricing (USD) per course |
Patent Status |
| CEFTIN |
20% |
Respiratory, UTI, Skin Infections |
50-70 |
Expired (2018) |
| Cefuroxime (generic) |
80% |
Same as CEFTIN |
20-40 |
Patents expired |
| Ceclor (Cefaclor) |
10% |
Respiratory, Skin Infections |
60-80 |
Patent expired |
Key Takeaways
- Sales remain stable despite intense generic competition.
- Market decline is modest, driven by resistance and stewardship.
- Growth prospects lie in emerging markets and formulation innovation.
- Competitive pressure affects pricing and margins.
FAQs
1. What factors recently impacted CEFTIN sales?
Rising antibiotic resistance, increased generic competition, and antibiotic stewardship initiatives have curtailed growth.
2. Are there new formulations or indications for CEFTIN?
No major recent updates; future developments could focus on pediatric formulations or combination therapies.
3. How significant is generic penetration in the CEFTIN market?
Generic versions account for about 80% of sales, significantly impacting branded sales revenue.
4. What markets are likely to see growth for CEFTIN?
Emerging markets like Asia-Pacific and Latin America where healthcare infrastructure expands.
5. How does CEFTIN compare with newer antibiotics?
CEFTIN is less favored for resistant infections, with newer or broad-spectrum agents replacing it in some indications.
References
- U.S. Food and Drug Administration (FDA). (2022). Approved Products Database. https://www.accessdata.fda.gov/scripts/cder/daf/.
- IMS Health. (2022). Global Antibiotic Market Report.
- IQVIA. (2023). Pharmaceutical Market Data.
- European Medicines Agency (EMA). (2023). Medicine approvals and use statistics.
- World Health Organization (WHO). (2022). Antibiotic Resistance and Global Strategies.