Last updated: April 24, 2026
Cefuroxime axetil is a widely used oral second-generation cephalosporin antibiotic with mature, off-patent economics in most major geographies. Market dynamics are driven by (1) generic substitution in oral cephalosporins, (2) clinician and payer preferences for narrower formulary positioning and local resistance patterns, and (3) periodic supply and quality events that can temporarily shift share. Financial trajectory is dominated by low pricing power, high procurement sensitivity, and a shrinking addressable market relative to newer stewardship-concordant therapies, with value concentrated in volume markets where generics are cheapest and formularies are stable.
How big is the cefuroxime axetil market and what shapes demand?
Demand base: community-acquired infection coverage
Cefuroxime axetil is used for outpatient bacterial infections where oral therapy is appropriate, with labeled uses in many markets historically including respiratory and ENT infections, urinary tract infections, and skin and soft tissue infections (indication labeling varies by country). The commercial “engine” is not innovation demand but sustained prescribing for common bacterial syndromes and step-down therapy from parenteral cephalosporins.
Core demand shapers
- Stewardship and prescribing controls
- Antibiotic stewardship programs in Europe, the UK, and parts of Asia have tightened oral antibiotic prescribing, especially for viral or self-limited conditions.
- This shifts demand from broad “practice-style” prescribing to guideline-concordant, narrow-spectrum selection.
- Local resistance epidemiology
- Cefuroxime susceptibility patterns by region influence guideline inclusion and clinicians’ confidence in oral beta-lactams.
- Where resistance rises, clinicians often move to alternatives (or reserve cephalosporins for specific syndromes).
- Formulary access and procurement mechanisms
- Public tenders and hospital formularies in many countries emphasize price-per-treatment course.
- Oral antibiotics face strong generic procurement pressure, so net sales tend to track competitor pricing rather than brand premium.
Structural market reality: maturity and genericization
Cefuroxime axetil is largely off-patent, so the “market” behaves like a mature generic antibiotic segment:
- Prices are set by generic competition.
- Switching is easy when bioequivalence is demonstrated and regulatory interchangeability is accepted.
- New entrants rarely win share through differentiation alone.
What are the competitive dynamics and pricing drivers?
Competition: generics dominate, with frequent interchangeable substitutes
In most regions, cefuroxime axetil is supplied by multiple generic manufacturers. Competitive dynamics typically follow:
- Price compression as the number of suppliers rises.
- Share gains by procurement position where tender specifications and contract awards favor the lowest-cost compliant supply.
- Short-term share swings tied to availability and batch release performance.
Pricing drivers: net price erosion and channel mix
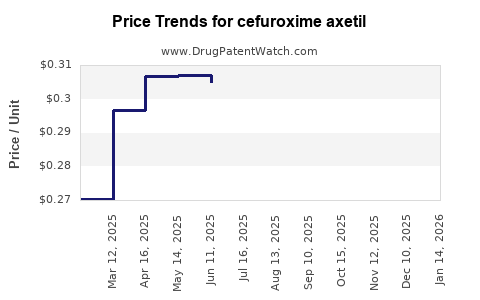
Cefuroxime axetil’s financial trajectory reflects persistent net price erosion:
- Wholesale and tender price pressure reduces gross-to-net spreads.
- Channel mix matters: outpatient retail tends to keep prices stable longer than tender-driven hospital outpatient procurement, but both segments are eventually pulled toward lowest available price.
Forecast implication
Without a proprietary product moat, the dominant levers for financial outcomes are:
- maintaining supply continuity,
- competing on lowest net price,
- and protecting formulary access at the local level.
How do generics and biosimilars influence cefuroxime axetil’s financial path?
Generic substitution is the primary mechanism
Unlike specialty pharma, cefuroxime axetil does not compete in a “me-too innovation” cycle. It competes through generic interchangeability:
- If a brand exists in a geography, it typically competes against multiple therapeutically substitutable oral antibiotics and low-cost generics.
- Generic entry triggers step-down in price and usually a share shift toward the lowest-cost supplier that meets procurement and quality requirements.
Margin structure: low operating leverage
For many manufacturers and distributors, economics tend to be:
- low gross margins,
- limited differentiation,
- and high working capital and compliance costs (antibiotic supply chain quality requirements).
Practical effect on trajectory
Financial performance typically shows:
- declining or flat unit revenues,
- declining net price,
- and volatility from supply and tender cycles.
What does the revenue and profitability trajectory look like over time?
Long-run pattern: revenue stability but margin contraction
For mature oral antibiotics like cefuroxime axetil, the typical trajectory is:
- Revenue: stable-to-declining as volume grows slowly or stays flat while price falls.
- Profitability: compresses as procurement price drops faster than variable costs can adjust.
- Cash flow: can improve temporarily if supply is stable and inventory turns are efficient, but working capital needs rise if distribution is disrupted.
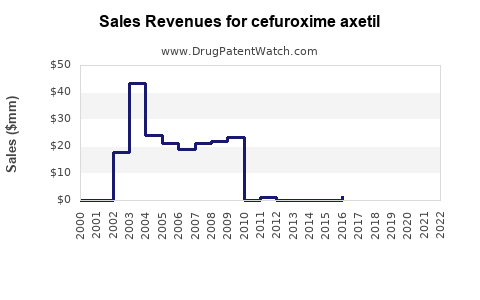
Short-run pattern: spikes from procurement and supply constraints
Net sales often show periodic step functions:
- winning a tender contract lifts volumes,
- losing a contract or supply disruption reduces volumes,
- quality or logistics events create temporary reallocation.
Investment and business implications
For business planners, cefuroxime axetil behaves more like an operational supply-and-procurement product than an R&D-led growth platform:
- successful outcomes come from consistent manufacturing and pricing discipline,
- and risk management centers on supply reliability and regulatory compliance.
What regulatory and lifecycle factors matter for the commercial outlook?
Antibiotic regulatory pressure and labeling constraints
Antibiotics face ongoing regulatory scrutiny around:
- appropriate use,
- stewardship alignment,
- safety monitoring,
- and post-market enforcement.
These factors rarely eliminate the product class outright but can reduce off-label volume and slow incremental uptake.
Lifecycle: mature status caps organic growth
Cefuroxime axetil’s lifecycle has limited catalysts:
- no patent-protected expansion implied for the base molecule in mature geographies,
- and growth depends on re-tendering cycles and stable guideline inclusion.
How do local guideline decisions and payer behavior translate into financial results?
Guideline inclusion depends on syndrome and local susceptibility
Where cefuroxime axetil is recommended for specific outpatient syndromes, physicians prescribe it within guideline frameworks. Where guidelines shift to alternatives due to resistance or spectrum positioning, uptake declines.
Payer behavior converts guideline text into market share
Even with guideline recommendations, reimbursement and procurement often decide actual share:
- If payer formularies restrict oral cephalosporins, demand falls even if clinical usage remains.
- If procurement contracts lock into a small set of lowest-cost SKUs, share concentrates among suppliers.
What market risks and opportunities could change the trajectory?
Main downside risks
- Rapid generic repricing
- A supplier that undercuts contract pricing can compress net revenue for all competitors.
- Stewardship tightening
- Continued tightening can reduce overall antibiotic prescribing volume.
- Supply disruption
- Any gap in manufacturing capacity or regulatory release can shift demand to substitutes and permanently reallocate prescriber and tender expectations.
Main upside opportunities
- Procurement wins in volume geographies
- Large outpatient and tender markets can create durable unit revenue stability if supplier performance is consistent.
- Narrow positioning in guideline-consistent syndromes
- When clinicians use cefuroxime axetil for specific indications, volume becomes less elastic to minor price changes.
Where does cefuroxime axetil sit versus other oral antibiotics economically?
Cefuroxime axetil competes in an oral antibiotic group where therapeutic substitutability is high. In such categories:
- payers and clinicians favor low-cost, guideline-concordant options,
- price tends to dominate market share,
- and innovation differentiation rarely supports sustained premium economics.
Compared with newer branded oral antibiotics in the same outpatient syndromes, cefuroxime axetil typically has:
- lower price,
- stable but capped growth,
- and lower margin potential,
- while benefiting from lower barriers to formulary inclusion due to mature acceptance.
What do the manufacturing and supply chain dynamics imply for financial performance?
Supply chain is the operational determinant
In mature generics, the biggest execution risks are:
- batch release delays,
- sterility and quality deviations,
- and manufacturing yield variability that affects cost of goods and ability to fulfill tenders.
Cost structure effects
- If API and excipient costs are stable, unit economics depend on throughput and yield.
- If costs rise, manufacturers either accept margin compression or exit tenders, leading to price and supply swings.
How can a financial model for cefuroxime axetil be structured for forecasting?
A practical forecast structure uses three drivers:
- Volume driver (scripts or treatment courses)
- proxy using market prescription trends and tender awards by country.
- Net price driver (weighted by channel)
- incorporate contract pricing and retail reimbursement pressure.
- Execution factor (share and supply continuity)
- include supplier-specific availability and quality performance.
A simplified decomposition:
- Net sales = Volume x Net price
- Operating margin = Net sales x gross margin - fixed compliance costs - logistics and regulatory costs
For cefuroxime axetil, changes in net price generally dominate financial outcomes because volume is relatively inelastic in mature antibiotic categories.
Market and Financial Trajectory Summary Table
| Dimension |
Typical trajectory for cefuroxime axetil |
Financial implication |
| Patent/IP |
Mature/off-patent economics |
No sustained premium pricing |
| Pricing |
Persistent compression from generic competition |
Net revenue declines per unit over time |
| Volume |
Stable-to-slow decline tied to stewardship |
Unit volumes do not offset price drops |
| Profitability |
Margin contraction with high compliance and low differentiation |
EBIT pressure; reliance on scale and execution |
| Volatility |
Tender cycles and supply events |
Step changes in quarterly sales |
Key Takeaways
- Cefuroxime axetil’s market is mature and generic-driven, so financial trajectory is dominated by pricing erosion and procurement dynamics rather than innovation.
- Demand is sustained by outpatient bacterial infection coverage, but stewardship and resistance patterns cap incremental growth and shift prescribing toward guideline-concordant alternatives.
- Profitability follows a low-margin, high-execution model: consistent supply, contract wins, and cost discipline matter more than differentiation.
- Forecasting should center on net price and volume-by-channel, with supplier availability and tender participation acting as the main variance sources.
FAQs
-
Is cefuroxime axetil a patent-protected product driving high pricing power?
No. In most major markets it functions as a mature, off-patent oral antibiotic where pricing is set by generic competition.
-
What is the biggest driver of net sales for cefuroxime axetil?
Weighted net price by channel (tenders and retail reimbursement), because generic substitution keeps volume relatively price-inelastic.
-
How do stewardship programs affect cefuroxime axetil?
They reduce inappropriate or broad antibiotic prescribing and shift demand toward more guideline-concordant choices, which can constrain growth.
-
What factors cause quarterly sales volatility?
Tender award timing, contract re-bids, and supply continuity or batch release performance.
-
Where can growth still occur for cefuroxime axetil manufacturers?
In volume procurement markets where contracts are awarded to reliable suppliers and local formulary positioning keeps cefuroxime axetil within guideline-consistent syndrome treatment.
References
[1] FDA. Cefuroxime Axetil (drug labels and prescribing information for listed products). U.S. Food and Drug Administration. https://www.accessdata.fda.gov/
[2] EMA. Cefuroxime and related product information (where applicable in European public sources). European Medicines Agency. https://www.ema.europa.eu/
[3] World Health Organization. Antimicrobial resistance and stewardship guidance and reporting. World Health Organization. https://www.who.int/
[4] IQVIA. Global antibiotic market and stewardship-related analytics (industry reports as published by IQVIA). IQVIA. https://www.iqvia.com/