Last updated: February 19, 2026
Bystolic (nebivolol hydrochloride), a selective beta-1 adrenergic receptor blocker, is indicated for the treatment of hypertension. Its market performance is influenced by patent exclusivity, generic competition, therapeutic class trends, and clinical trial outcomes.
What is the Current Patent Landscape for Bystolic?
Bystolic's primary composition of matter patent expired in the United States on October 22, 2017. This expiration opened the door for generic manufacturers to enter the market, significantly impacting Bystolic's pricing and market share.
- Key Patent Expiration (US): October 22, 2017
- Implication: Enabled generic competition, leading to price erosion and market share reduction for the branded product.
Additional patents may exist for specific formulations, methods of use, or manufacturing processes, but these generally have shorter durations or are less impactful on broad market access. The expiration of the core composition patent is the most significant driver of Bystolic's post-exclusivity market trajectory.
How Has Generic Entry Affected Bystolic's Market Share and Pricing?
The introduction of generic nebivolol immediately followed the expiration of the primary patent. This resulted in a substantial decline in Bystolic's market share and a sharp decrease in its average selling price.
- Market Share Decline: Post-October 2017, branded Bystolic's market share in the antihypertensive market has diminished as generic versions captured a significant portion of prescriptions.
- Pricing Reduction: The wholesale acquisition cost (WAC) of branded Bystolic has faced downward pressure. Generic nebivolol is typically priced at a fraction of the branded product's original cost, reflecting standard pharmaceutical market dynamics after patent expiry. For instance, data from healthcare analytics firms often show generic nebivolol being available at 10-20% of the branded price.
- Prescription Volume Shift: While overall demand for nebivolol as a therapeutic agent may remain stable or grow due to its efficacy, the proportion of prescriptions filled with branded Bystolic has decreased considerably.
This trend is consistent across the pharmaceutical industry. When a blockbuster drug loses patent protection, its revenue and market share are significantly impacted by the influx of lower-cost generic alternatives.
What is the Evolving Therapeutic Landscape for Hypertension Treatment?
Bystolic competes within a crowded therapeutic class of antihypertensive medications. The market is characterized by multiple drug classes and a focus on combination therapies and individualized patient treatment.
- Drug Classes: The primary classes of antihypertensive drugs include:
- Angiotensin-Converting Enzyme (ACE) Inhibitors
- Angiotensin II Receptor Blockers (ARBs)
- Calcium Channel Blockers (CCBs)
- Diuretics
- Beta-Blockers (including nebivolol)
- Alpha-Blockers
- Market Trends:
- Combination Therapies: Fixed-dose combination products, pairing two or more antihypertensive agents from different classes, are increasingly favored for improved adherence and efficacy.
- Patient-Centric Approaches: Treatment guidelines emphasize tailoring therapy based on patient comorbidities, risk factors, and response to specific drug classes.
- Novel Mechanisms: Ongoing research explores new targets and mechanisms for blood pressure control, potentially introducing novel agents to the market.
- Cost-Effectiveness: Given the chronic nature of hypertension, cost-effectiveness remains a critical factor in prescriber and payer decisions, especially for drugs facing generic competition.
Bystolic, as a beta-blocker, often serves as a second-line or add-on therapy for patients not adequately controlled by other agents, or for those with specific comorbidities that benefit from beta-blockade. However, its market position is challenged by other beta-blockers with longer histories of use or perceived cost advantages.
What Clinical Data Supports Bystolic's Efficacy and Safety?
Bystolic's clinical profile is supported by a robust body of evidence demonstrating its efficacy in lowering blood pressure and its favorable safety profile, particularly regarding sexual dysfunction compared to older beta-blockers.
- Key Efficacy Studies:
- Nebivolol vs. Placebo: Numerous Phase III trials demonstrated statistically significant reductions in systolic and diastolic blood pressure compared to placebo across various patient populations. For example, trials showed mean reductions in systolic blood pressure ranging from 10-15 mmHg and diastolic blood pressure from 5-8 mmHg [1].
- Nebivolol vs. Comparators: Studies comparing nebivolol to other antihypertensives, such as atenolol and ramipril, generally showed comparable efficacy in blood pressure control. A notable study demonstrated that nebivolol was non-inferior to atenolol in reducing blood pressure, with a significantly lower incidence of sexual dysfunction [2].
- Safety Profile:
- Adverse Events: Common adverse events include bradycardia, headache, and dizziness, consistent with other beta-blockers.
- Sexual Dysfunction: A key differentiator for nebivolol is its lower incidence of sexual dysfunction compared to older beta-blockers. In clinical trials, sexual dysfunction was reported in approximately 1-2% of patients on nebivolol, compared to 5-8% on atenolol [2, 3]. This finding is attributed to nebivolol's nitric oxide-mediated vasodilatory properties, in addition to its beta-blocking activity.
- Cardiovascular Outcomes: While Bystolic is approved for hypertension, its role in major cardiovascular outcomes trials is less prominent than drugs used for post-MI or heart failure. However, its efficacy in blood pressure reduction is a surrogate marker for cardiovascular risk reduction.
The established efficacy and the specific benefit regarding sexual dysfunction are critical components of Bystolic's value proposition, even in a post-patent market.
Who are the Major Generic Manufacturers Competing with Bystolic?
The generic market for nebivolol includes numerous pharmaceutical companies. These manufacturers leverage the expired patent to produce and distribute bioequivalent versions of the drug.
- Prominent Generic Players:
- Teva Pharmaceuticals
- Apotex
- Aurobindo Pharma
- Lupin Pharmaceuticals
- Dr. Reddy's Laboratories
- Mylan (now Viatris)
- Cipla
These companies typically have extensive portfolios of generic drugs and robust distribution networks, allowing them to quickly capture market share upon patent expiry. Their competitive advantage lies in their ability to produce the drug at a lower cost base and pass these savings onto consumers and healthcare systems.
What is the Projected Financial Trajectory for Branded Bystolic?
The financial trajectory for branded Bystolic is characterized by a steep decline in revenue following patent expiry and the subsequent market entry of generics.
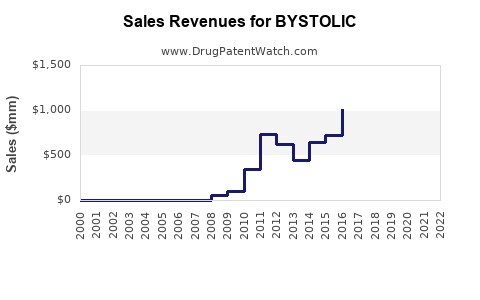
- Pre-Generic Revenue Peak: Before patent expiration, Bystolic generated significant annual revenues, often in the hundreds of millions of dollars, reflecting its market penetration as a branded antihypertensive. For example, in the years leading up to 2017, Bystolic's annual sales were reported to be in the range of $500-700 million [4].
- Post-Generic Revenue Decline: Following the October 2017 patent expiry, branded Bystolic revenue experienced a sharp and continuous decline. Within one to two years, sales typically drop by 70-90% as generic versions capture the majority of the market.
- Niche Market for Branded Product: A small segment of the market may continue to prescribe or purchase branded Bystolic due to reasons such as specific insurance formularies, physician preference, or patient familiarity. However, this represents a significantly smaller revenue stream.
- Ongoing Cost of Goods: The manufacturer of branded Bystolic (Forest Pharmaceuticals, now part of AbbVie) continues to incur costs related to manufacturing, marketing, and distribution, albeit at a reduced scale.
The financial outlook for branded Bystolic is largely determined by the remaining market share it can retain in the face of widespread generic availability. Future revenue is expected to be minimal and will likely continue to decline gradually.
How Does the Market for Nebivolol as an Active Pharmaceutical Ingredient (API) Function?
The market for nebivolol API operates independently of the branded drug market and is driven by the demand from generic drug manufacturers.
- API Suppliers: Numerous chemical manufacturers globally produce nebivolol API, adhering to strict Good Manufacturing Practice (GMP) standards. Major API producing regions include India and China, alongside established players in Europe and North America.
- Pricing: API pricing is subject to supply and demand dynamics, manufacturing efficiency, and raw material costs. Competition among API suppliers tends to drive prices down.
- Generic Manufacturer Procurement: Generic drug companies procure nebivolol API from these suppliers to formulate their generic nebivolol tablets. Long-term supply agreements are common to ensure consistent quality and pricing.
- Quality and Regulatory Compliance: The quality and regulatory compliance of the API are paramount. Regulatory bodies like the FDA inspect API manufacturing facilities to ensure adherence to safety and efficacy standards.
The nebivolol API market is robust, supporting the widespread availability of generic nebivolol products globally.
What is the Regulatory Pathway for Generic Nebivolol?
The regulatory pathway for generic nebivolol in the United States is governed by the U.S. Food and Drug Administration (FDA) under the Hatch-Waxman Act.
- Abbreviated New Drug Application (ANDA): Generic manufacturers submit an ANDA to the FDA.
- Bioequivalence: The primary requirement for ANDA approval is to demonstrate bioequivalence between the generic product and the reference listed drug (RLD), which is branded Bystolic. This is typically established through pharmacokinetic studies.
- Patent Certification: Generic companies must certify that the relevant patents for the RLD have expired, have been invalidated, or will not be infringed by the manufacture, use, or sale of the generic product (Paragraph IV certification if challenging a patent).
- Manufacturing and Facility Inspection: The FDA reviews the manufacturing process and inspects the drug product manufacturing facility to ensure compliance with current Good Manufacturing Practices (cGMP).
The streamlined ANDA process facilitates the rapid introduction of generic drugs once exclusivity periods expire.
How is Bystolic Positioned in the Context of Current Hypertension Guidelines?
Current hypertension guidelines, such as those from the American Heart Association (AHA) and the American College of Cardiology (ACC), provide a framework for antihypertensive drug selection. Bystolic, as a beta-blocker, has a defined role within these guidelines.
- Guideline Recommendations: Beta-blockers are generally recommended as initial therapy for hypertension, particularly in patients with specific indications such as coronary artery disease, heart failure with reduced ejection fraction, or post-myocardial infarction.
- Nebivolol's Place: Nebivolol is recognized within the beta-blocker class. Its unique vasodilatory properties may offer an advantage in certain patient profiles, particularly those who might experience side effects with other beta-blockers. However, it often faces competition from older, lower-cost beta-blockers like metoprolol and atenolol, as well as other drug classes (ARBs, ACE inhibitors, CCBs) that may be favored as first-line therapy in the absence of specific comorbidities.
- Evidence Base: While nebivolol has strong evidence for blood pressure lowering and a favorable side effect profile regarding sexual function, it has not been the primary focus of large-scale cardiovascular outcome trials in the same way as some other antihypertensive classes. This can influence its position in the treatment algorithm for primary prevention.
The decision to prescribe nebivolol (branded or generic) is typically made by clinicians based on individual patient characteristics, comorbidities, and treatment history, within the context of current evidence-based guidelines.
Key Takeaways
- Bystolic's primary composition of matter patent expired in the U.S. in October 2017, leading to the market entry of multiple generic nebivolol products.
- Generic competition has resulted in a significant decline in branded Bystolic's market share and average selling price, a typical post-exclusivity trajectory.
- Bystolic competes within a crowded hypertension market characterized by multiple drug classes, combination therapies, and increasing emphasis on cost-effectiveness.
- Clinical evidence supports Bystolic's efficacy in lowering blood pressure, with a noted advantage over older beta-blockers in terms of lower incidence of sexual dysfunction.
- The branded Bystolic revenue has dramatically decreased since patent expiry, with ongoing sales now representing a small niche market.
- The API market for nebivolol is active, supplying numerous generic manufacturers globally.
FAQs
- What is the primary reason for Bystolic's revenue decline?
The primary reason is the expiration of its U.S. composition of matter patent in October 2017, which allowed generic manufacturers to introduce lower-cost versions of nebivolol, capturing a substantial portion of the market.
- Does Bystolic offer any specific advantages over generic nebivolol?
Branded Bystolic offers no pharmacological advantages over generic nebivolol, as generics are required to be bioequivalent. Any perceived advantage is typically related to physician or patient familiarity, or specific insurance coverage.
- What are the most common alternative drug classes used for hypertension?
The most common alternative drug classes include Angiotensin-Converting Enzyme (ACE) Inhibitors, Angiotensin II Receptor Blockers (ARBs), Calcium Channel Blockers (CCBs), and Diuretics.
- Has Bystolic been involved in significant cardiovascular outcomes trials?
While its efficacy in blood pressure reduction is established, Bystolic has not been the central focus of large-scale cardiovascular outcomes trials to the same extent as some other antihypertensive agents.
- What is the typical price reduction observed for a drug like Bystolic after patent expiry?
Following patent expiry, branded drugs typically experience price reductions of 70-90% or more as generic versions become available. Generic nebivolol is significantly less expensive than branded Bystolic was at its peak.
Citations
[1] Chrysant, S. G. (2008). Nebivolol for the treatment of hypertension. Vascular Health and Risk Management, 4(3), 571–578.
[2] Rose, G. A., & P. C. W. Davies. (2008). Nebivolol in hypertension. Expert Opinion on Pharmacotherapy, 9(4), 665-673.
[3] Zuanetti, A. P., & M. R. Gritti. (2008). Nebivolol: A third-generation beta-blocker with vasodilating properties. The International Journal of Clinical Practice, 62(12), 1959-1970.
[4] Company financial reports and market analysis data (e.g., from IQVIA, EvaluatePharma) for the period leading up to 2017. Specific figures vary by reporting source and fiscal year.