BRAFTOVI Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Braftovi, and what generic alternatives are available?
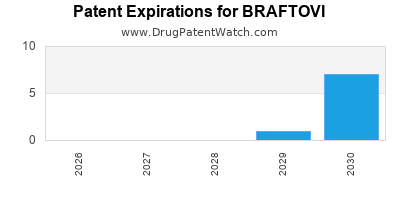
Braftovi is a drug marketed by Array Biopharma Inc and is included in one NDA. There are thirteen patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and ninety-nine patent family members in fifty-three countries.
The generic ingredient in BRAFTOVI is encorafenib. One supplier is listed for this compound. Additional details are available on the encorafenib profile page.
DrugPatentWatch® Generic Entry Outlook for Braftovi
Braftovi was eligible for patent challenges on June 27, 2022.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be November 21, 2032. This may change due to patent challenges or generic licensing.
There have been ten patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for BRAFTOVI?
- What are the global sales for BRAFTOVI?
- What is Average Wholesale Price for BRAFTOVI?
Summary for BRAFTOVI
| International Patents: | 199 |
| US Patents: | 13 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 59 |
| Clinical Trials: | 20 |
| Patent Applications: | 2,502 |
| Drug Prices: | Drug price information for BRAFTOVI |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for BRAFTOVI |
| What excipients (inactive ingredients) are in BRAFTOVI? | BRAFTOVI excipients list |
| DailyMed Link: | BRAFTOVI at DailyMed |
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for BRAFTOVI
Generic Entry Date for BRAFTOVI*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
CAPSULE;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for BRAFTOVI
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| National Cancer Institute (NCI) | Phase 1 |
| Deciphera Pharmaceuticals LLC | Phase 1/Phase 2 |
| Pfizer | Phase 2 |
Paragraph IV (Patent) Challenges for BRAFTOVI
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| BRAFTOVI | Capsules | encorafenib | 75 mg | 210496 | 3 | 2022-06-27 |
US Patents and Regulatory Information for BRAFTOVI
BRAFTOVI is protected by thirty-seven US patents and three FDA Regulatory Exclusivities.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of BRAFTOVI is ⤷ Start Trial.
This potential generic entry date is based on patent 9,387,208.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Array Biopharma Inc | BRAFTOVI | encorafenib | CAPSULE;ORAL | 210496-002 | Jun 27, 2018 | RX | Yes | Yes | RE49556 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Array Biopharma Inc | BRAFTOVI | encorafenib | CAPSULE;ORAL | 210496-002 | Jun 27, 2018 | RX | Yes | Yes | 9,593,100 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Array Biopharma Inc | BRAFTOVI | encorafenib | CAPSULE;ORAL | 210496-001 | Jun 27, 2018 | DISCN | Yes | No | 9,314,464 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Array Biopharma Inc | BRAFTOVI | encorafenib | CAPSULE;ORAL | 210496-002 | Jun 27, 2018 | RX | Yes | Yes | 8,541,575 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for BRAFTOVI
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Pierre Fabre Medicament | Braftovi | encorafenib | EMEA/H/C/004580Encorafenib is indicated:in combination with binimetinib is indicated for the treatment of adult patients with unresectable or metastatic melanoma with a BRAF V600 mutationin combination with cetuximab, for the treatment of adult patients with metastatic colorectal cancer (CRC) with a BRAF V600E mutation, who have received prior systemic therapy | Authorised | no | no | no | 2018-09-19 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for BRAFTOVI
When does loss-of-exclusivity occur for BRAFTOVI?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 8936
Patent: FORMULACIONES FARMACEUTICAS
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 12340759
Patent: Pharmaceutical formulations
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 2014011981
Patent: formulações farmacêuticas
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 56406
Patent: FORMULATIONS PHARMACEUTIQUES (PHARMACEUTICAL FORMULATIONS)
Estimated Expiration: ⤷ Start Trial
Chile
Patent: 14001337
Patent: Formulacion farmaceutica oral solida que comprende una dispersion solida que comprende a su vez un compuesto especifico derivado de 4-(1-isopropil-1h-pirazol-4-il)-pirimidina y su uso en el tratamiento de una enfermedad proliferativa.
Estimated Expiration: ⤷ Start Trial
China
Patent: 3945831
Patent: Pharmaceutical formulations
Estimated Expiration: ⤷ Start Trial
Patent: 5708819
Patent: 医药制剂 (PHARMACEUTICAL FORMULATIONS)
Estimated Expiration: ⤷ Start Trial
Colombia
Patent: 40426
Patent: Formulaciones farmacéuticas
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0181896
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 21421
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 82557
Estimated Expiration: ⤷ Start Trial
Ecuador
Patent: 23034537
Patent: FORMULACIONES FARMACEUTICAS
Estimated Expiration: ⤷ Start Trial
Eurasian Patent Organization
Patent: 5389
Patent: ЛЕКАРСТВЕННЫЕ СОСТАВЫ (PHARMACEUTICAL FORMULATIONS)
Estimated Expiration: ⤷ Start Trial
Patent: 1491007
Patent: ЛЕКАРСТВЕННЫЕ СОСТАВЫ
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 82557
Patent: FORMULATIONS PHARMACEUTIQUES (PHARMACEUTICAL FORMULATIONS)
Estimated Expiration: ⤷ Start Trial
Patent: 49911
Patent: FORMULATIONS PHARMACEUTIQUES (PHARMACEUTICAL FORMULATIONS)
Estimated Expiration: ⤷ Start Trial
Guatemala
Patent: 1400100
Patent: FORMULACIONES FARMACÉUTICAS
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 97024
Patent: 醫藥製劑 (PHARMACEUTICAL FORMULATIONS)
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 40370
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 2305
Patent: תכשירי רוקחות (Pharmaceutical formulations)
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 16325
Estimated Expiration: ⤷ Start Trial
Patent: 15501808
Patent: 医薬製剤
Estimated Expiration: ⤷ Start Trial
Patent: 18035171
Patent: 医薬製剤 (PHARMACEUTICAL FORMULATION)
Estimated Expiration: ⤷ Start Trial
Jordan
Patent: 93
Patent: صيغ صيدلانية (PHARMACEUTICAL FORMULATIONS)
Estimated Expiration: ⤷ Start Trial
Lithuania
Patent: 82557
Estimated Expiration: ⤷ Start Trial
Malaysia
Patent: 2729
Patent: PHARMACEUTICAL FORMULATIONS
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 3446
Patent: FORMULACIONES FARMACEUTICAS. (PHARMACEUTICAL FORMULATIONS.)
Estimated Expiration: ⤷ Start Trial
Patent: 14006278
Patent: FORMULACIONES FARMACEUTICAS. (PHARMACEUTICAL FORMULATIONS.)
Estimated Expiration: ⤷ Start Trial
Morocco
Patent: 716
Patent: Formulations pharmaceutiques
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 3628
Patent: Solid oral pharmaceutical formulations comprising amorphous (s)-methyl (1- ((4-(3-(5-chloro-2-fluoro-3-(methylsulfonamido)phenyl)-1-isopropy1-1h-pyrazo1-4-yl)pyrimidin-2-yl)amino)propan-2-yl)carbamate (compound a)
Estimated Expiration: ⤷ Start Trial
Peru
Patent: 141994
Patent: FORMULACIONES FARMACEUTICAS
Estimated Expiration: ⤷ Start Trial
Philippines
Patent: 014501157
Patent: PHARMACEUTICAL FORMULATIONS
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 82557
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 82557
Estimated Expiration: ⤷ Start Trial
San Marino
Patent: 01800595
Estimated Expiration: ⤷ Start Trial
Serbia
Patent: 048
Patent: FARMACEUTSKE FORMULACIJE (PHARMACEUTICAL FORMULATIONS)
Estimated Expiration: ⤷ Start Trial
Singapore
Patent: 201401260Q
Patent: PHARMACEUTICAL FORMULATIONS
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 82557
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 1402418
Patent: PHARMACEUTICAL FORMULATIONS
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 2091295
Estimated Expiration: ⤷ Start Trial
Patent: 140095500
Patent: PHARMACEUTICAL FORMULATIONS
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 95099
Estimated Expiration: ⤷ Start Trial
Patent: 37966
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 1328722
Patent: Pharmaceutical formulations
Estimated Expiration: ⤷ Start Trial
Patent: 49098
Estimated Expiration: ⤷ Start Trial
Tunisia
Patent: 14000145
Patent: PHARMACEUTICAL FORMULATIONS
Estimated Expiration: ⤷ Start Trial
Ukraine
Patent: 5039
Patent: ФАРМАЦЕВТИЧНІ КОМПОЗИЦІЇ (PHARMACEUTICAL FORMULATIONS)
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering BRAFTOVI around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Portugal | 2470526 | ⤷ Start Trial | |
| Serbia | 53489 | JEDINJENJA I PREPARATI PROTEIN KINAZNIH INHIBITORA (COMPOUNDS AND COMPOSITIONS AS PROTEIN KINASE INHIBITORS) | ⤷ Start Trial |
| Dominican Republic | P2012000051 | COMPUESTOS Y COMPOSICIONES COMO INHIBIDORES DE CINASA DE PROTEINA | ⤷ Start Trial |
| Hong Kong | 1167390 | 蛋白激酶抑制劑的化合物和組合物 (COMPOUNDS AND COMPOSITIONS AS PROTEIN KINASE INHIBITORS) | ⤷ Start Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for BRAFTOVI
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2727918 | LUC00102 | Luxembourg | ⤷ Start Trial | PRODUCT NAME: COMBINAISON DE BINIMETINIB ET D'ENCORAFENIB, CHACUN SOUS TOUTES SES FORMES TELLES QUE PROTEGEES PAR LE BREVET DE BASE; AUTHORISATION NUMBER AND DATE: EU/1/18/1314 20180924 |
| 2470526 | LUC00101 | Luxembourg | ⤷ Start Trial | PRODUCT NAME: ENCORAFENIB SOUS TOUTES SES FORMES TELLES QUE PROTEGEES PAR LE BREVET DE BASE; AUTHORISATION NUMBER AND DATE: EU/1/18/1314 20180924 |
| 2727918 | 2019C/509 | Belgium | ⤷ Start Trial | DETAILS ASSIGNMENT: CHANGE OF OWNER(S), MERGE |
| 2727918 | 201940012 | Slovenia | ⤷ Start Trial | PRODUCT NAME: COMBINATION OF BINIMETINIB AND ENCORAFENIB, EACH OF WHICH IS OPTIONAL IN THE FORM OF PHARMACEUTICALLY ACCEPTABLE SALT OR SOLVATE; NATIONAL AUTHORISATION NUMBER: EU/1/18/1314/001-002, EU/1/18/1315/001; DATE OF NATIONAL AUTHORISATION: 20180920; AUTHORITY FOR NATIONAL AUTHORISATION: EU |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for BRAFTOVI
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.