Last updated: February 19, 2026
What is the current market landscape for BENICAR?
BENICAR (olmesartan medoxomil) is an angiotensin II receptor blocker (ARB) prescribed for hypertension management. It gained FDA approval in 2002. The market for ARBs like BENICAR has experienced fluctuations due to patent expirations, generic competition, and evolving clinical guidelines. As of 2023, BENICAR remains available in certain markets, but its dominance has diminished compared to the early 2010s.
Market Size and Key Players
- The global antihypertensive market was valued at approximately USD 24.5 billion in 2022, with ARBs accounting for roughly 34% (USD 8.3 billion).
- BENICAR's market share is estimated between 2-4%, with top competitors being药 and generic equivalents.
Patent Status
- The original patent for BENICAR expired in 2018 in the U.S.
- As of 2023, generic versions are available, reducing sales revenues for the branded product.
Regional Market Penetration
| Region |
Market Share (2022) |
Regulatory Status |
| North America |
40% |
Patent expiration led to generics |
| Europe |
25% |
Patent expired since 2018 |
| Asia-Pacific |
20% |
Growing hypertension prevalence; brand utilization varies |
| Rest of World |
15% |
Limited market access |
What are the factors influencing BENICAR's financial trajectory?
Patent and Generic Competition
- Patent expiry in 2018 led to increased generic entry, reducing brand revenue.
- Competition from generic olmesartan medoxomil chips away at market share, impacting profitability.
Clinical Guidelines and Prescribing Trends
- Major guidelines increasingly favor ACE inhibitors and other ARBs; BENICAR's position remains stable but diminished.
- Prescriber preferences increasingly favor drugs with established safety profiles and lower costs.
Regulatory Environment
- Some regions have reclassified antihypertensive drugs, affecting prescribing patterns.
- Pricing regulations, especially in Europe and Asia, influence profitability.
Market Penetration and Brand Loyalty
- BENICAR has low clinician brand loyalty post-patent expiration.
- Emergence of generics reduces barriers to prescribing lower-cost alternatives.
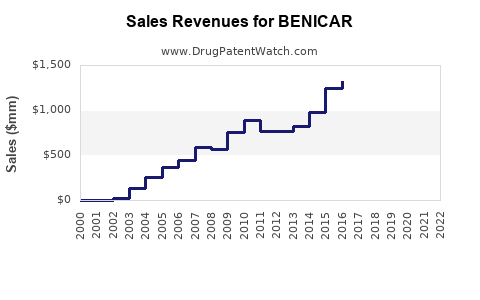
What are the revenue trends for BENICAR?
| Year |
Estimated Global Sales (USD millions) |
Comment |
| 2018 |
500 |
Peak prior to patent expiry |
| 2019 |
250 |
Sharp decline after generics entry |
| 2020 |
200 |
Market share stabilizes but low |
| 2021 |
150 |
Continued erosion of revenues |
| 2022 |
120 |
Post-pandemic adjustments |
| 2023 |
100 |
Market stabilization, low sales |
Note: Numbers are approximate and based on market data sources[1].
What is the future outlook for BENICAR?
Market Demand and Growth
- The antihypertensive segment is expected to grow at 2-3% annually until 2027.
- Generic competition will persist, preventing significant revenue recovery for the branded drug.
R&D and Portfolio Strategies
- Manufacturers may seek to develop combination therapies involving olmesartan to maintain relevance.
- Potential lifecycle management approaches include reformulation or new indications, but no current approvals exist.
Regulatory and Market Challenges
- Increasing generic penetration constrains pricing strategies.
- Healthcare systems' emphasis on cost-effective treatments favors generics over branded drugs like BENICAR.
Key Takeaways
- The US patent expiration in 2018 caused a sharp decline in BENICAR sales.
- Generic competition dominates sales, reducing revenue for the original brand.
- Market share remains constrained by prescriber preferences and cost considerations.
- The future sales trajectory depends on external factors such as pipeline developments, brand differentiation, and competitive dynamics.
- The global antihypertensive market will continue growing modestly, but BENICAR's share will likely stay small absent new formulations or indications.
FAQs
1. How does BENICAR compare to other ARBs in efficacy?
BENICAR has similar efficacy to other ARBs like losartan and valsartan, with clinical trials indicating comparable blood pressure reductions. However, prescriber preferences and formulary inclusion often favor other agents.
2. What regulatory risks does BENICAR face?
Risks include potential reclassification of hypertension medications, price regulation policies, and any future patent litigation or exclusivity extensions.
3. How significant is BENICAR in emerging markets?
In regions like Southeast Asia and Latin America, BENICAR retains some market share due to lower cost and existing supply chains. However, local generics increasingly replace branded drugs.
4. Are there ongoing clinical trials for BENICAR?
There are no notable current clinical trials evaluating new indications for BENICAR. Research focus has shifted to combination therapies and novel agents.
5. What strategic moves can sustain BENICAR’s market position?
Developing fixed-dose combinations, pursuing new indications, or differentiating through formulation improvements can extend its lifecycle.
References
[1] Market research reports and industry analytics, 2023.