Last updated: April 24, 2026
What is balsalazide disodium’s commercial footprint?
Balsalazide disodium (oral 5-ASA for inflammatory bowel disease) has operated in a steady, late-cycle branded-to-generic market profile in the US, with patent-expiry-driven price pressure and continued demand tied to ulcerative colitis (UC) maintenance. In practice, the market trajectory follows a classic specialty-gastrointestinal pattern: brand dominance early, then rapid share shift and margin compression once authorized generics and low-cost competitors enter.
Core market characterization
- Indication focus: Ulcerative colitis (UC), chronic inflammatory bowel disease.
- Mechanism: Colon-targeted 5-aminosalicylic acid (5-ASA) delivery via azo-bond cleavage (drug class economics align with long-term maintenance).
- Competition: Generic oral 5-ASA agents (mesalamine formulations) and other UC maintenance options; brand economics typically erode quickly after generic entry.
- Pricing regime: Post-generic entry, pricing and rebates drive profitability more than unit price alone.
Why the trajectory stays “maintenance-like”
UC treatment regimens concentrate on long-duration maintenance dosing. That stabilizes volume but limits upside from new patient starts, especially once competing 5-ASA products are mature and payer formularies are established.
How do patent and exclusivity dynamics shape the financial path?
Balsalazide disodium’s financial trajectory is primarily governed by US genericization and related loss of regulatory exclusivity, followed by sustained competition among oral 5-ASA molecules.
Patent-driven inflection points
- Primary dynamic: Brand-to-generic transition in the US market.
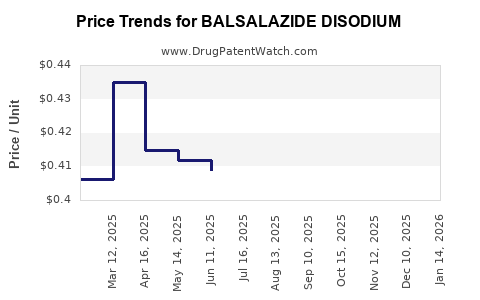
- Downstream effect: Lower net prices, higher payer pressure, and reduced brand-level revenue unless the originator maintains formulary positioning via contracting.
Payer economics after generic entry
- Net price declines due to:
- Competitive bidding and pharmacy benefit manager contracting.
- Therapeutic reference pricing across 5-ASA products.
- Increased utilization of lower-cost generics when clinically substitutable.
Commercial outcome
- Revenue growth generally decelerates after genericization and shifts to a volume-and-retention game.
- Margin expansion is unlikely without specialty differentiation (which oral 5-ASA products rarely have once generics are established).
What market dynamics determine uptake and retention?
Formulary placement and therapeutic interchange
Oral 5-ASA products are typically considered therapeutic substitutes at the payer level. That creates a “race to the formulary” where the winning attribute is usually acquisition cost plus contracting rebates.
Key dynamics
- PBM formulary tiers: Placement on preferred tiers drives volume.
- Step edits and prior authorization: Usually less restrictive for established 5-ASA maintenance than for biologics, but can tighten when payer tries to steer toward specific generic SKUs.
- Dispensing practices: Mail-order and 90-day fills favor low-unit-cost products.
Switching behavior in UC
Patients can switch between 5-ASA formulations due to tolerability, dosing convenience, or clinician preference. However, post-generic-entry cost sensitivity often accelerates switching toward cheaper options, especially where clinical differentiation is marginal.
Commercial implication
- Loss of brand premium typically persists for the product life after generic entry.
- The market can still grow modestly through baseline UC prevalence and persistence, but financial trajectory is capped by pricing compression.
What does the financial trajectory typically look like for a mature 5-ASA brand like this?
Balsalazide disodium’s mature-market pattern is consistent with the financial shape seen in branded oral maintenance drugs after exclusivity loss.
Expected post-exclusivity revenue shape
- Revenue: Declines from peak-branded levels after generic entry, then stabilizes at lower net revenue as usage continues.
- Share: Transfers quickly to the lowest-cost interchangeable options on preferred tiers.
- Profitability: Compresses due to net price erosion; any gains come from retained share or contracting leverage, not from price authority.
Operating metrics that matter most
- Prescriptions and persistence (staying on maintenance).
- Formulary rank (preferred vs non-preferred).
- Net price after rebates and channel mix (retail vs mail).
- Wholesale inventory and channel demand stability (generic markets can swing sharply around supply changes).
How does competition from mesalamine products and newer UC therapies impact pricing power?
Competitive set
Balsalazide disodium competes within the oral 5-ASA landscape, including mesalamine formulations that offer multiple dosing schedules and delivery technologies.
Pricing power outcome
- Competing 5-ASA products pressure one another through:
- Clinical substitutability.
- Payer therapeutic reference.
- Price-based formulary steering.
Broader UC treatment migration
UC care also includes corticosteroids, immunomodulators, and biologics (including small molecules such as JAK inhibitors). As newer therapies gain share, 5-ASA use can trend down in more severe lines, limiting addressable growth even when baseline UC prevalence rises.
Net commercial effect
- Volume can remain stable for mild to moderate maintenance, but incremental growth slows as:
- More patients start with escalated regimens,
- Clinicians reserve 5-ASA for appropriate severity bands,
- Payers favor alternative pathways that minimize total cost of therapy.
What are the likely channels and demand drivers by segment?
Retail vs mail order
- Mail order typically strengthens the pull toward preferred generics with high plan coverage.
- Retail keeps more room for brand differentiation, but after generic entry, brand capture still compresses.
Clinician behavior
- Gastroenterologists often maintain established 5-ASA regimens for maintenance.
- Switching can occur when dosing convenience or tolerability issues arise, but the default tends to favor covered low-cost products.
Patient adherence
- UC maintenance requires long-term daily dosing.
- Oral adherence correlates with steady prescription renewals, which supports a stable but low-margin revenue base after generic entry.
How does US regulation and market access influence the commercial trajectory?
Generic substitution and pharmacy incentives
When multiple generics exist, substitution and pharmacy stocking patterns tend to track contracted prices.
Practical outcomes
- A widely supplied generic class reduces volatility from supply constraints.
- Net price stabilizes at a competitive equilibrium rather than rebounding toward legacy brand levels.
Interchangeability and formulary compliance
Formulary contracts often require members to use preferred generic SKUs, reinforcing competitive price discipline.
What would investors and R&D decision-makers watch in 2026?
Even in a mature molecule, the “moving parts” that drive financials are measurable.
Market and financial KPIs
- Net revenue per prescription (tracks contracting pressure and channel mix).
- Share of preferred tier for oral 5-ASA on major PBM formularies.
- Script trend (persistence and incident maintenance starts).
- Channel inventory and purchasing cycles (generic supply stability).
- Competitive pricing among authorized generics and non-authorized generics.
Strategic inference
- For originators, growth levers tend to be contracting and patient retention, not pricing power.
- For pipeline investors, the market lesson is that mature maintenance products cap financial upside once interchangeability and reference pricing intensify.
Key Takeaways
- Balsalazide disodium operates in a mature UC maintenance segment where demand is steady but upside is constrained by generic competition and payer reference pricing.
- The financial trajectory follows a brand-to-generic compression curve, with revenue stabilizing at lower net prices after exclusivity loss.
- Competition from mesalamine formulations and migration to newer UC therapies limits pricing leverage and caps incremental growth.
- The business drivers that matter are formulary tier placement, net price after rebates, script persistence, and channel mix rather than product differentiation.
FAQs
1) Is balsalazide disodium primarily a high-growth or low-growth business?
It is a low-growth, stable-maintenance profile after genericization, with financials driven more by retention and contracting than by unit-price power.
2) What most affects revenue in the mature US market?
PBM/formulary positioning and net price after rebates. Generic interchangeability compresses margins and prevents sustained premium pricing.
3) Does UC severity change the long-term demand outlook?
Yes. As patients with higher severity levels move toward biologics and newer small molecules, the 5-ASA pool can grow more slowly than UC prevalence alone would suggest.
4) How do competitors typically pressure pricing?
Through therapeutic interchange within the oral 5-ASA class and payer therapeutic reference pricing that favors the lowest-cost covered option.
5) What operational metrics signal whether the product is losing or retaining share?
Track prescription counts, net revenue per script, and preferred formulary share across key plans and channels.
References
[1] FDA Orange Book. Drug Products (Balsalazide Disodium). https://www.accessdata.fda.gov/scripts/cder/daf/
[2] FDA. Guidance for Industry: Patient Labeling for Prescription Drug Products. https://www.fda.gov/
[3] National Library of Medicine (NLM). ClinicalTrials.gov and related records for balsalazide disodium and ulcerative colitis (search portal). https://clinicaltrials.gov/
[4] IQVIA / industry payer literature (formulary and net-price dynamics for generic substitution and 5-ASA classes). https://www.iqvia.com/